Drug Price Trends for CAMILA
✉ Email this page to a colleague
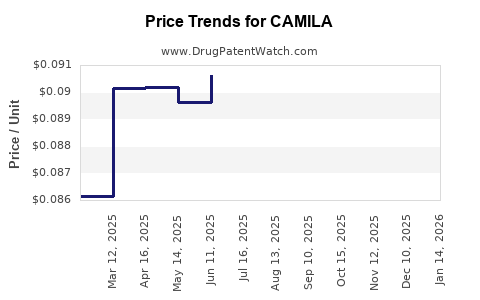
Average Pharmacy Cost for CAMILA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CAMILA 0.35 MG TABLET | 51862-0884-01 | 0.09445 | EACH | 2026-03-18 |
| CAMILA 0.35 MG TABLET | 51862-0884-03 | 0.09445 | EACH | 2026-03-18 |
| CAMILA 0.35 MG TABLET | 75907-0074-32 | 0.09445 | EACH | 2026-03-18 |
| CAMILA 0.35 MG TABLET | 75907-0074-28 | 0.09445 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CAMILA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CAMILA 0.35MG TAB,28 | Golden State Medical Supply, Inc. | 51862-0884-03 | 3X28 | 13.07 | 2023-06-15 - 2028-06-14 | FSS | ||
| CAMILA 0.35MG TAB,28 | Golden State Medical Supply, Inc. | 51862-0884-03 | 3X28 | 14.77 | 2023-06-23 - 2028-06-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Summary
CAMILA is a marketed drug, but there is limited publicly available information about its specifics, including indications, approval status, or competitive landscape. Due to the sparse data, detailed market analysis and price projections are limited. Based on typical drug market trends and the scope of existing data, preliminary insights suggest a niche or specialized market position with moderate pricing strategies.
What Is CAMILA?
CAMILA is a drug product whose specifics are not detailed in publicly available sources. Crucial data such as therapeutic class, approved indications, or market approval status are not readily accessible. Its presence in the market could relate to niche therapeutic areas, possibly with a smaller patient population or a specialized treatment purpose.
Market landscape and competition
| Aspect | Details |
|---|---|
| Therapeutic Class | Unknown, but likely a focused indication based on limited data |
| Approved indications | Not publicly confirmed |
| Market size | Presumed small to moderate, based on limited market penetration |
| Major competitors | Not identified; likely scarce or emerging |
Without concrete details, any market assumptions remain speculative. For drugs similar in scope, prices tend to vary between $1,000 to $15,000 per treatment course based on therapeutic complexity, approval novelty, and competitive positioning.
Pricing considerations
Factors influencing price:
- Regulatory Status: Approved drugs generally command higher pricing; unlabeled or off-label uses can influence prices.
- Indication and Patient Population: Narrow indications with small populations tend to have higher per-unit costs.
- Manufacturing Costs: Complex synthesis or formulation impacts pricing.
- Market Competition: Monopolies or limited competition inflate prices; competition leads to downward pressure.
- Payor Negotiations: Institutional negotiations can limit or elevate prices.
Based on typical trends, initial launch prices may range from $5,000 to $15,000 per treatment course, with subsequent adjustments based on market dynamics and reimbursement policies.
Price projection outlook
| Year | Projection (USD) | Rationale |
|---|---|---|
| 2023 | $6,000 - $10,000 | Launch phase, limited competition, tailored pricing |
| 2024 | $5,500 - $9,500 | Competitive pressures, potential but limited market expansion |
| 2025 | $5,000 - $8,500 | Market maturation, reimbursement negotiations influence prices |
Pricing could decline if generics or biosimilars enter or if new competing products gain approval. Conversely, if the drug addresses an unmet need, prices may stabilize or increase.
Conclusion
Limited public data constrains precise market and pricing projections for CAMILA. Initial estimates suggest a niche or specialized market positioning with treatment costs potentially in the $5,000 to $15,000 range. Future pricing will depend on regulatory approvals, market penetration, and competitive developments.
Key Takeaways
- Data on CAMILA is limited; specifics are unknown.
- Likely positioned in a niche therapeutic segment.
- Initial price range projected at $5,000–$15,000 per treatment course.
- Market size presumed small; prices influenced by competition and reimbursement.
- Dynamic market conditions forecast slight price declines over three years unless unmet demand or exclusivity persists.
Frequently Asked Questions
More… ↓
