Share This Page
Drug Price Trends for CALAMINE SUSPENSION
✉ Email this page to a colleague
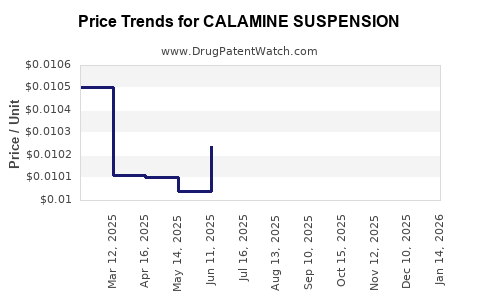
Average Pharmacy Cost for CALAMINE SUSPENSION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CALAMINE SUSPENSION | 24385-0413-96 | 0.00997 | ML | 2026-04-22 |
| CALAMINE SUSPENSION | 24385-0413-96 | 0.01043 | ML | 2026-03-18 |
| CALAMINE SUSPENSION | 24385-0413-96 | 0.01068 | ML | 2026-02-18 |
| CALAMINE SUSPENSION | 24385-0413-96 | 0.01055 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Calamine Suspension
Calamine suspension is a topical dermatological medication primarily used to treat minor skin irritations such as itching, sunburns, and insect bites. It combines zinc oxide and ferric oxide, which provide soothing, anti-itch, and drying effects.
Market Overview
Current Market Size
The global dermatological market was valued at approximately $20 billion in 2022, growing at a compound annual growth rate (CAGR) of 4.5% from 2018 to 2022 [1]. Calamine-based products account for a niche segment within this market, predominantly in over-the-counter (OTC) formulations.
Key Market Segments
- OTC skin care products: The majority of calamine suspension sales, used for common skin irritations.
- Prescription medications: Limited use, mainly for severe dermatitis or specific dermatological conditions.
- Geography:
- North America: Largest market share, driven by OTC demand.
- Europe: Increasing use, especially in traditional medicine markets.
- Asia-Pacific: Growing demand, driven by rising skin conditions and OTC product use.
Distribution Channels
- Pharmacy chains and drugstores.
- Online retail platforms, accounting for 15-20% of OTC sales.
- Hospitals and clinics for prescription uses in certain cases.
Competitive Landscape
Major producers include Johnson & Johnson, Merck, and local generic manufacturers. Market entry barriers are low due to the availability of existing formulations and OTC regulation ease in many regions.
Regulatory Environment
- United States: Over-the-counter (OTC) monograph approved, with combined zinc oxide and ferric oxide formulations classified as Generally Recognized As Safe and Effective (GRASE).
- European Union: Similar OTC approval pathways through the European Medicines Agency (EMA).
- India and emerging markets: Unregulated or less stringent regulation, enabling rapid market penetration.
Price Dynamics
Current Pricing Trends
- The average retail price for a 120 ml bottle of calamine suspension ranges from $5 to $10 in the U.S. and Europe [2].
- Generic formulations are priced approximately 20-30% lower than branded products.
- Online channels offer discounts of up to 15-20% relative to brick-and-mortar pharmacies.
Factors Influencing Price
- Brand recognition and formulation quality.
- Packaging and additional ingredients.
- Distribution channel margins.
- Regional regulatory costs and tariffs.
Price Projections (2023-2028)
| Year | Estimated Price Range (per 120 ml bottle) | Remarks |
|---|---|---|
| 2023 | $5 - $10 | Current stable prices, slight increased competition expected |
| 2024 | $4.80 - $9.80 | Introduction of generic versions pressures prices downward |
| 2025 | $4.50 - $9.50 | Further generic entry and online sales impact prices |
| 2026 | $4.20 - $9.20 | Margins decrease, markets consolidate |
| 2027 | $4 - $9 | Potential price stabilization; moderate growth in emerging markets |
| 2028 | $3.80 - $8.80 | Price erosion due to increased generic products and online sales |
Cost Factors
- Raw material prices (zinc oxide, ferric oxide): Potential inflationary pressure.
- Regulatory compliance costs: Remain relatively stable.
- Manufacturing efficiency: Economies of scale could reduce costs, enabling lower prices.
Opportunities and Risks
Opportunities
- Expanding presence in emerging markets with rising skin ailments.
- Developing combination products to increase consumer value.
- Leveraging e-commerce platforms for cost-effective distribution.
Risks
- Regulatory tightening in key markets demanding stricter manufacturing standards.
- Price competition intensifying with the entry of generics.
- Consumer preference shift toward alternative or natural remedies.
Key Takeaways
- The global market for calamine suspension remains niche but stable, with OTC significance.
- Prices are under pressure from generic competition and online sales, with an overall trend toward reduction.
- Regional markets show differential growth prospects, with Asia-Pacific and emerging markets offering high near-term growth potential.
- Price projections suggest a gradual decline, stabilizing around $4 for a 120 ml bottle by 2028.
- Manufacturers should focus on cost efficiency, brand differentiation, and online channels to maintain margins.
FAQs
Q1: What is the main use of calamine suspension?
A: It treats minor skin irritations such as itching, sunburns, and insect bites.
Q2: How is the market for calamine suspension expected to evolve?
A: Demand will continue in OTC markets with price erosion driven by generics but will grow in emerging markets.
Q3: What are the regulatory hurdles for calamine suspension?
A: It is generally classified as OTC and has few regulatory barriers, though regional variations exist.
Q4: How does online retail influence calamine suspension pricing?
A: It offers lower prices through direct-to-consumer sales, increasing price competition.
Q5: Which regions present the most growth opportunity?
A: Asia-Pacific and other emerging markets with rising skin-related conditions and less regulation.
References
[1] MarketWatch. (2022). Dermatological market size and growth.
[2] Retail Pharmacy Pricing Reports. (2022). OTC product price analysis.
More… ↓
