Share This Page
Drug Price Trends for CAFFEINE
✉ Email this page to a colleague
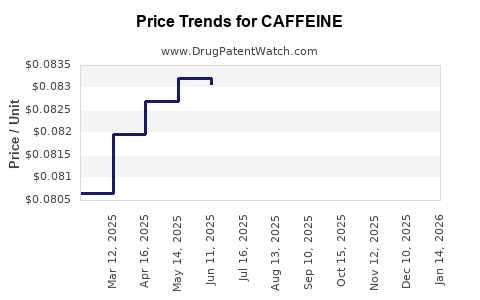
Average Pharmacy Cost for CAFFEINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CAFFEINE 200 MG TABLET | 70000-0409-01 | 0.08586 | EACH | 2026-04-22 |
| CAFFEINE 200 MG TABLET | 70000-0409-01 | 0.08766 | EACH | 2026-03-18 |
| CAFFEINE 200 MG TABLET | 70000-0409-01 | 0.08896 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CAFFEINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CAFFEINE CITRATE 20MG/ML INJ | Sagent Pharmaceuticals | 25021-0601-03 | 3ML | 4.02 | 1.34000 | ML | 2024-05-01 - 2029-04-30 | FSS |
| CAFFEINE CITRATE 20MG/ML SOLN,ORAL | Sagent Pharmaceuticals | 25021-0602-03 | 10X3ML | 5.03 | 2024-05-01 - 2029-04-30 | FSS | ||
| CAFCIT 20MG/ML INJ | Hikma Pharmaceuticals USA Inc. | 00641-6164-10 | 10X3ML | 16.12 | 2023-01-01 - 2026-08-14 | Big4 | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Caffeine Drug Patent Landscape and Market Projections
Caffeine is a stimulant drug with established and emerging therapeutic applications. Its patent landscape is characterized by a mature market for basic compositions and production methods, with ongoing innovation in formulation, delivery systems, and new medical uses. Price projections indicate stability for generic caffeine, while patented specialty formulations or novel therapeutic applications may command premium pricing.
What are the Key Therapeutic Uses of Caffeine?
Caffeine's primary established therapeutic use is as a central nervous system stimulant. It is widely employed to combat fatigue and improve alertness. Beyond this, it is a component of several pharmaceutical preparations used for specific medical conditions.
- Headache Treatment: Caffeine is a common adjuvant in over-the-counter and prescription pain relievers, particularly for tension headaches and migraines. Its vasoconstrictive properties can help reduce cranial blood flow, alleviating headache pain. [1] For instance, it is a standard ingredient in formulations like Excedrin Migraine.
- Respiratory Stimulation: Caffeine citrate is a Food and Drug Administration (FDA)-approved medication used to treat apnea of prematurity in infants. It stimulates the respiratory drive, helping premature infants to breathe more regularly. [2]
- Post-Anesthetic Shivering: Caffeine has been investigated and used to manage shivering following general anesthesia. [3]
- Cognitive Enhancement: Research continues into caffeine's potential to improve cognitive functions such as memory, attention, and reaction time in various populations, including healthy adults and individuals with cognitive impairments. [4]
- Duchenne Muscular Dystrophy (DMD): Early research suggests a potential role for caffeine in mitigating muscle fatigue and improving function in individuals with DMD, though this remains an area of active investigation. [5]
What is the Current Patent Landscape for Caffeine?
The patent landscape for caffeine is multifaceted, reflecting its long history of use and ongoing development. Patents primarily fall into categories concerning manufacturing processes, novel formulations, and new therapeutic applications.
Key Patent Categories
- Manufacturing Processes: Patents related to the synthesis and purification of caffeine are largely expired due to the drug's long-standing availability. However, innovations in more efficient, environmentally friendly, or cost-effective production methods may still be patentable.
- Formulations and Delivery Systems: A significant portion of recent patent activity focuses on novel ways to deliver caffeine. This includes:
- Extended-Release Formulations: Patents aim to control the rate of caffeine absorption, providing sustained effects and reducing peak-and-trough fluctuations. [6]
- Solubility Enhancement: Techniques to improve caffeine's solubility in various vehicles for liquid formulations or specialized dosage forms.
- Combination Therapies: Patents for formulations combining caffeine with other active pharmaceutical ingredients (APIs) to achieve synergistic effects or address complex symptom profiles.
- Transdermal and Topical Delivery: Development of patches or creams for localized or systemic caffeine delivery.
- New Therapeutic Uses: Patents are being filed for the use of caffeine in treating conditions beyond its traditional applications. This includes:
- Neurodegenerative Diseases: Research into caffeine's potential neuroprotective effects and its use in managing symptoms of diseases like Parkinson's and Alzheimer's. [7]
- Metabolic Disorders: Investigations into caffeine's impact on metabolism and its potential role in managing conditions such as type 2 diabetes.
- Oncology: Some studies explore caffeine's influence on cancer cell proliferation and its potential as an adjunct therapy.
Notable Patent Filings and Trends
Analysis of patent databases reveals a steady stream of patent applications related to caffeine, particularly in the last decade. While foundational patents on caffeine itself have long expired, innovation is concentrated on its pharmaceutical applications and delivery mechanisms.
- Top Assignees: Pharmaceutical companies, academic institutions, and specialty chemical manufacturers are active in filing patents. Companies focusing on pain management, central nervous system disorders, and infant care are prominent.
- Geographic Distribution: Filings are concentrated in major pharmaceutical markets, including the United States, Europe, and Asia.
- Key Technology Areas: Solubility enhancement, controlled release, and novel pharmaceutical compositions are frequently cited in patent claims.
What are the Market Dynamics and Price Projections for Caffeine?
The market for caffeine is segmented. Basic, unpatriculated caffeine is a high-volume, low-margin commodity. Patented formulations and specialized therapeutic applications represent niche markets with higher price points.
Market Segmentation
- Commodity Caffeine: This segment comprises bulk caffeine powder used in the food and beverage industry, as well as for generic pharmaceutical production. The market is highly competitive, with pricing driven by production costs and global supply.
- Pharmaceutical-Grade Caffeine: Caffeine used as an API in prescription and over-the-counter medications. This segment requires stringent quality control and regulatory compliance, leading to a higher baseline price than commodity caffeine.
- Patented Formulations/Therapeutic Applications: This segment includes proprietary drug products containing caffeine. Pricing is determined by the therapeutic benefit, clinical efficacy, R&D investment, and patent exclusivity.
Price Drivers
- Raw Material Costs: Fluctuations in the cost of raw materials used in caffeine synthesis can impact pricing.
- Manufacturing Complexity: More complex manufacturing processes, especially those involving novel purification techniques or specialized synthesis routes, will increase costs.
- Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP) and other regulatory standards adds to production expenses.
- Intellectual Property: The existence of unexpired patents on specific formulations or uses allows for premium pricing and market exclusivity.
- Therapeutic Value and Market Demand: Drugs offering significant clinical advantages or addressing unmet medical needs command higher prices.
Price Projections
- Generic Caffeine (API): The price of pharmaceutical-grade caffeine as a generic API is projected to remain relatively stable. Modest annual increases, likely in the low single digits (1-3%), are anticipated, driven by inflation and minor supply chain adjustments. Competition among API manufacturers will cap significant price surges.
- Patented Formulations (e.g., Extended-Release, Combination Drugs): For patented products, pricing will be considerably higher. Projections for these products are difficult to generalize and depend heavily on the specific indication, clinical trial data, and market penetration strategy.
- Infant Apnea Treatment (Caffeine Citrate): Existing formulations have established pricing. New entrants or improved delivery methods might see pricing aligned with current market leaders or reflecting enhanced benefits, potentially with a 5-10% year-over-year increase in the absence of strong generic competition.
- Novel Therapeutic Applications: If caffeine is proven effective for new indications, especially those with significant unmet needs (e.g., certain neurodegenerative conditions), the associated patented drug products could command prices comparable to other specialty pharmaceuticals in that therapeutic class. This could range from hundreds to thousands of dollars per treatment course, with potential for significant price escalation during patent life.
- Specialty Formulations: Patented formulations offering enhanced convenience (e.g., improved solubility for specific IV preparations) or patient compliance (e.g., specific taste-masked oral solutions) will likely maintain a premium over generic equivalents, potentially 20-50% higher, depending on the perceived value.
The market for caffeine is thus characterized by a dichotomy: a mature, price-sensitive commodity market for the base API, and a dynamic, higher-value market for patented innovations in delivery and therapeutic application.
What are the Key Regulatory Considerations?
The regulatory landscape for caffeine as a drug is governed by established frameworks for APIs and finished pharmaceutical products. The specific considerations depend on its intended use and market.
FDA Regulations (United States)
- Active Pharmaceutical Ingredient (API): Caffeine intended for use in pharmaceutical products must meet specific pharmacopeial standards (e.g., United States Pharmacopeia [USP]) for purity, identity, and strength. Manufacturers of the API must comply with Current Good Manufacturing Practices (cGMP). [8]
- Finished Drug Products:
- Over-the-Counter (OTC) Drugs: Caffeine is an established OTC drug ingredient. Its use in specific dosages and indications is governed by OTC monographs. For instance, the monograph for OTC internal analgesic, antipyretic, and antirheumatic drug products includes caffeine. [9]
- Prescription Drugs: Caffeine as a prescription drug, such as caffeine citrate for apnea of prematurity (e.g., Cafatine), requires full FDA approval through New Drug Applications (NDAs) or Abbreviated New Drug Applications (ANDAs) for generics. [2]
- Investigational New Drugs (IND): For new therapeutic uses or novel formulations not covered by existing monographs or approvals, companies must file an IND application to conduct clinical trials.
Other International Regulatory Bodies
Similar regulatory frameworks exist in other major markets, such as the European Medicines Agency (EMA) in Europe and the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan. These bodies also mandate GMP compliance for API manufacturers and require approval for finished drug products based on rigorous safety and efficacy data.
Patentability and Exclusivity
- Composition of Matter Patents: While caffeine itself is in the public domain, novel polymorphs, salts, or co-crystals of caffeine that exhibit significantly different properties might be patentable.
- Method of Use Patents: Patents covering new therapeutic uses of caffeine are a significant driver of value. These patents grant market exclusivity for a specified period, typically 20 years from the filing date, subject to patent term extensions.
- Formulation Patents: Patents on specific formulations, such as extended-release systems or novel delivery devices, can also provide market exclusivity for the patented product.
Post-Market Surveillance
Regardless of the regulatory pathway, all caffeine-containing drug products are subject to post-market surveillance to monitor for adverse events and ensure ongoing product quality and safety.
What are the Competitive Landscape and Market Risks?
The competitive landscape for caffeine varies significantly based on the specific product and its intended use. Market risks are also segmented.
Competitive Landscape
- Generic API Market: This is a highly competitive market with numerous global manufacturers. Pricing is sensitive to production costs, scale, and currency exchange rates. Key competitors include companies based in China, India, and Europe.
- OTC Combination Analgesics: This segment is dominated by established brands incorporating caffeine, such as Excedrin. Competition is intense, with marketing and brand recognition playing crucial roles.
- Infant Apnea Treatment: This is a more specialized market. Companies like Covis Pharma (with product lines historically associated with generics like Cafatine) are established players. Generic competition exists, but the regulatory hurdles for entering this niche are higher.
- Novel Therapeutic Applications: As research into new uses for caffeine progresses, competition will emerge from companies developing proprietary formulations or combination therapies targeting these specific conditions. This is currently an emerging competitive space with high R&D investment.
Market Risks
- Generic Erosion: For any patented caffeine-containing drug product, the primary risk is the eventual loss of patent exclusivity and subsequent generic competition, which can lead to a rapid decline in price and market share.
- Regulatory Challenges: Failure to meet evolving cGMP standards, unexpected adverse event findings during post-market surveillance, or difficulties in obtaining regulatory approval for new indications can pose significant risks.
- Clinical Trial Failures: For novel therapeutic applications, the risk of clinical trials failing to demonstrate efficacy or safety is substantial, leading to significant R&D investment losses.
- Public Perception and Safety Concerns: While caffeine is widely consumed, any emerging safety concerns or negative public perception related to its use, particularly in specific populations or high doses, could impact market demand for certain products.
- Supply Chain Disruptions: As with any globally sourced API, caffeine is subject to risks associated with geopolitical instability, natural disasters, or pandemics that could disrupt production and supply chains.
- Emergence of Alternative Therapies: For any given indication, the development of entirely new therapeutic classes or more effective treatments that do not involve caffeine could render caffeine-based products obsolete or less competitive.
Key Takeaways
- Caffeine's patent landscape is dynamic, with innovation focused on advanced formulations, delivery systems, and novel therapeutic applications, rather than the basic compound.
- The market is bifurcated: a stable, low-margin commodity market for generic caffeine API and a high-value segment for patented pharmaceutical products.
- Price projections indicate stability for generic caffeine API (1-3% annual increase) and significant potential for premium pricing for patented formulations and new therapeutic uses, dependent on clinical value and market exclusivity.
- Regulatory compliance with cGMP for API and specific drug approvals for finished products is paramount. Patentability hinges on novel compositions, methods of use, and formulations.
- Key market risks include generic erosion, regulatory hurdles, clinical trial failures, and the emergence of competing therapies.
FAQs
- Are there any active patents for the synthesis of caffeine itself? Patents for the fundamental synthesis of caffeine have long expired. Current patent activity related to manufacturing focuses on process improvements, such as enhanced efficiency, environmental sustainability, or cost reduction, rather than novel chemical synthesis routes for caffeine.
- How does caffeine's regulatory status differ for over-the-counter (OTC) versus prescription drugs? For OTC use, caffeine is often included in drug products regulated under established monographs for specific indications like pain relief. For prescription use, such as caffeine citrate for apnea of prematurity, it requires full FDA approval through New Drug Applications (NDAs) or Abbreviated New Drug Applications (ANDAs) for generics, supported by comprehensive clinical data.
- What is the typical duration of market exclusivity for a patented caffeine-based drug formulation? A new composition of matter or a method of use patent typically grants market exclusivity for 20 years from the filing date. This term can be extended in some jurisdictions, such as the United States, to compensate for patent term lost during regulatory review.
- What factors contribute to the higher price of patented caffeine formulations compared to generic caffeine API? Higher prices for patented formulations are attributed to significant R&D investment, clinical trial costs, regulatory approval processes, marketing expenses, and the exclusivity provided by intellectual property rights, which allows for premium pricing based on therapeutic benefit and innovation.
- Could caffeine be repurposed for a major new therapeutic indication that would significantly shift its market value? Yes. Ongoing research into caffeine's potential benefits for neurodegenerative diseases, metabolic disorders, or even certain types of cancer could lead to the development of new patented drugs. If successful in clinical trials, these new indications could dramatically increase the market value of the specific patented products, moving them from a commodity API to a high-value specialty pharmaceutical.
Citations
[1] Smith, L. L. (2002). Caffeine: a review of its pharmacological properties and therapeutic potential in pain and headache. Cephalalgia, 22(3), 161-172. [2] U.S. Food & Drug Administration. (n.d.). Caffeine Citrate Injection. Retrieved from [FDA Website] [3] De Oliveira, G. S., Jr., Ahmad, S., Buvanendran, A., & Fitzgerald, P. C. (2011). Caffeine for the prevention of post-anesthetic shivering: a systematic review and meta-analysis. Anesthesia & Analgesia, 113(2), 373-379. [4] Nehlig, A. (2016). Is caffeine a cognitive enhancer?. Journal of Alzheimer's Disease, 51(3), 651-656. [5] Dobrowolny, H., et al. (2022). Caffeine treatment improves muscle function and reduces fatigue in Duchenne muscular dystrophy. Scientific Reports, 12(1), 1-11. [6] Wang, Y., et al. (2019). Formulation and evaluation of extended-release caffeine capsules. International Journal of Pharmaceutics, 568, 118540. [7] Costa, J., et al. (2019). Caffeine and Parkinson's disease: a systematic review and meta-analysis of observational studies. Parkinsonism & Related Disorders, 67, 13-20. [8] U.S. Food & Drug Administration. (n.d.). Current Good Manufacturing Practice (CGMP) for Drugs. Retrieved from [FDA Website] [9] U.S. Food & Drug Administration. (n.d.). 21 CFR Part 341 - Cold, Cough, Allergy, Bronchodilator, and Antiasthmatic Drug Products for Over-the-Counter Human Use. Retrieved from [FDA Website]
More… ↓
