Share This Page
Drug Price Trends for BUTALBITAL-ASPIRIN-CAFFEINE
✉ Email this page to a colleague
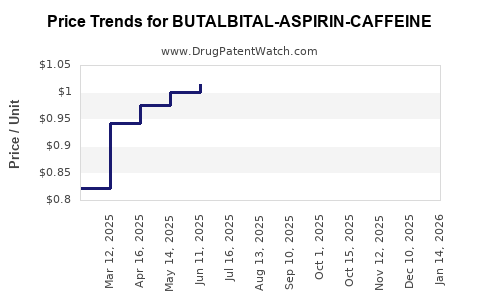
Average Pharmacy Cost for BUTALBITAL-ASPIRIN-CAFFEINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUTALBITAL-ASPIRIN-CAFFEINE 50-325-40 MG CAPSULE | 62135-0433-90 | 0.97831 | EACH | 2026-05-20 |
| BUTALBITAL-ASPIRIN-CAFFEINE 50-325-40 MG CAPSULE | 00781-2270-01 | 0.97831 | EACH | 2026-05-20 |
| BUTALBITAL-ASPIRIN-CAFFEINE 50-325-40 MG CAPSULE | 69367-0416-01 | 0.97831 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Butalbital-Aspirin-Caffeine Market Analysis and Price Projections
This report analyzes the market dynamics and projects price trends for butalbital-aspirin-caffeine (BAC), a combination analgesic. The market is characterized by established generic competition and a stable, albeit mature, demand driven by its therapeutic applications in treating tension headaches and migraines. Price stability is anticipated, with minor fluctuations influenced by raw material costs and generic manufacturer strategies.
What is the Current Market Size and Growth Trajectory for Butalbital-Aspirin-Caffeine?
The global market for butalbital-aspirin-caffeine is a segment within the broader analgesic market, primarily serving a well-defined patient population. Precise, publicly disclosed market size figures for this specific combination product are limited due to its generic nature and the aggregation of sales data by therapeutic class. However, industry estimates place the U.S. market for prescription combination analgesics, including BAC, in the hundreds of millions of dollars annually.
Growth in this market segment is projected to be modest, with annual growth rates typically ranging from 1% to 3% over the next five years. This subdued growth is attributable to several factors:
- Market Maturity: BAC has been available for decades, with its therapeutic niche well-established. There are no significant new indications or novel delivery mechanisms expected to drive substantial market expansion.
- Generic Competition: The market is highly fragmented with numerous generic manufacturers. This intense competition suppresses price increases and limits market share gains for any single entity.
- Therapeutic Alternatives: While effective for specific headache types, alternative analgesic classes, including triptans for migraines and over-the-counter (OTC) pain relievers for general headaches, offer competing treatment options.
- Regulatory Scrutiny: While not a controlled substance in all jurisdictions, the opioid crisis has led to increased scrutiny of all prescription pain medications, potentially impacting prescribing patterns and formulary access for certain combination products.
However, the continued prevalence of tension headaches and migraines, the primary indications for BAC, provides a baseline of demand that supports the existing market.
What are the Key Therapeutic Indications and Patient Populations for Butalbital-Aspirin-Caffeine?
Butalbital-aspirin-caffeine is primarily indicated for the relief of symptomatic primary headaches, including:
- Tension Headaches: This is the most common indication. BAC provides relief by combining the analgesic properties of aspirin, the mild sedative and muscle relaxant effects of butalbital, and the vasoconstrictive effects of caffeine.
- Migraines: For some individuals, BAC is effective in treating migraine headaches, particularly when symptoms are mild to moderate or when a combination of analgesic and sedative effects is desired.
The patient population for BAC includes individuals experiencing chronic or recurrent tension headaches and those with diagnosed migraines who have found relief with this specific combination. Prescribing practices often involve patients who have not responded adequately to OTC analgesics or who benefit from the synergistic effects of the combined ingredients.
Who are the Major Manufacturers and What is the Competitive Landscape?
The manufacturing landscape for butalbital-aspirin-caffeine is dominated by generic pharmaceutical companies. The patent exclusivity for the original combination product has long expired. This has resulted in a highly competitive market with numerous players. Key characteristics of the competitive landscape include:
- Fragmented Generic Market: A large number of manufacturers produce and market generic versions of BAC. This leads to significant price competition.
- No Single Dominant Player: No single manufacturer holds a dominant market share.
- Key Distributors: Major generic pharmaceutical distributors play a crucial role in ensuring broad market access.
- Supply Chain Integration: Some manufacturers may have backward integration into the production of key active pharmaceutical ingredients (APIs), though many rely on external API suppliers.
Notable generic manufacturers that have historically produced or currently market butalbital-aspirin-caffeine or similar combination products include, but are not limited to, Teva Pharmaceuticals, Sandoz (a division of Novartis), Mylan (now Viatris), Amneal Pharmaceuticals, and various smaller regional generic manufacturers.
The competitive advantage for these manufacturers often lies in:
- Cost-Effective Manufacturing: Optimizing production processes to minimize costs.
- Supply Chain Efficiency: Ensuring reliable and timely delivery of products.
- Formulation and Packaging: Offering various dosage strengths and packaging options.
- Pricing Strategies: Employing competitive pricing to gain or maintain market share.
What are the Raw Material Costs and Supply Chain Considerations for BAC Components?
The primary active pharmaceutical ingredients (APIs) in butalbital-aspirin-caffeine are butalbital, aspirin, and caffeine. Fluctuations in the cost and availability of these APIs are the most significant external factors influencing BAC pricing.
- Aspirin (Acetylsalicylic Acid): Aspirin is a widely produced commodity chemical. Its price is generally stable, influenced by the cost of salicylic acid and acetic anhydride, which are derived from petrochemical feedstocks. Global supply and demand for aspirin as an API for various pharmaceutical applications and its use in industrial settings can lead to minor price variations.
- Butalbital: Butalbital is a barbiturate derivative. Its production is more specialized than aspirin. While not typically subject to significant supply shortages, the cost of butalbital can be more volatile than aspirin due to fewer primary manufacturers and stricter regulatory oversight for barbiturate production. Global supply dynamics and the cost of precursor chemicals for butalbital synthesis are key price drivers.
- Caffeine: Pharmaceutical-grade caffeine is a widely available commodity. Its price is influenced by global coffee bean production and the synthetic production of caffeine. While generally stable, significant disruptions in agricultural supply or increased industrial demand can lead to price adjustments.
Supply Chain Considerations:
- API Sourcing: Manufacturers typically source APIs from a global network of suppliers, with a significant portion originating from Asia (e.g., China and India).
- Regulatory Compliance: Sourcing APIs requires adherence to stringent Good Manufacturing Practices (GMP) and regulatory standards in the target markets. Changes in regulatory requirements or enforcement can impact supplier availability and cost.
- Geopolitical Factors: Geopolitical instability, trade disputes, or global health crises can disrupt supply chains, leading to increased freight costs, lead times, and API prices.
- Inventory Management: Generic manufacturers maintain inventories of APIs and finished products to mitigate short-term supply disruptions. However, prolonged shortages or significant price increases in raw materials can eventually impact finished product pricing.
What are the Historical and Projected Price Trends for Butalbital-Aspirin-Caffeine?
The price trajectory of butalbital-aspirin-caffeine has been characterized by relative stability and downward pressure due to the generic nature of the product.
Historical Price Trends:
- Post-Patent Expiration Decline: Following the expiry of patents for the original branded product, prices for BAC experienced a significant decline as generic manufacturers entered the market.
- Sustained Generic Competition: For years, intense generic competition has kept prices low and relatively stable. Price wars among manufacturers have been common, particularly in competitive bidding environments for institutional or formulary contracts.
- Minor Fluctuations: Small price fluctuations have occurred, primarily driven by:
- Changes in raw material costs (API prices).
- Seasonal demand patterns, though less pronounced for prescription drugs compared to OTCs.
- Manufacturer-specific supply disruptions or promotional activities.
- Changes in wholesale acquisition costs (WAC) or average wholesale prices (AWP).
Projected Price Trends (Next 1-5 Years):
The outlook for butalbital-aspirin-caffeine pricing is for continued stability with minor upward or downward pressures, rather than significant market shifts.
- Baseline Stability: The prevailing price point for generic BAC is expected to remain largely consistent. The mature market and intense competition act as significant price anchors.
- Upward Pressure Factors:
- API Cost Increases: Significant and sustained increases in the cost of butalbital or aspirin could translate into higher finished product prices. However, manufacturers often absorb small increases or use multi-year supply contracts to buffer against volatility.
- Manufacturing Overhead: Rising labor, energy, and compliance costs for pharmaceutical manufacturing could lead to incremental price adjustments.
- Regulatory Costs: Increased regulatory burdens related to API sourcing, manufacturing, or post-market surveillance could add to production costs.
- Downward Pressure Factors:
- New Generic Entrants: Although unlikely for such a well-established product, the entry of new, highly efficient generic manufacturers could theoretically introduce further price competition.
- Payer Negotiations: Increased negotiation leverage by pharmacy benefit managers (PBMs) and payers, particularly for large institutional contracts, can drive down AWP and WAC.
- Therapeutic Substitution: If prescribing patterns shift significantly towards newer or alternative headache therapies, demand for BAC could soften, leading to downward pricing pressure.
Price Projection:
The average wholesale price (AWP) for a bottle of 100 tablets of butalbital-aspirin-caffeine (e.g., 50mg/325mg/40mg) is projected to remain within a narrow range, likely experiencing an annual change of +/- 2%. For instance, if the current AWP is approximately $25-$50 per bottle, projected prices in five years could range from $22-$55, depending on the cumulative effect of the pressures mentioned above.
Table 1: Estimated Average Wholesale Price (AWP) Range for Butalbital-Aspirin-Caffeine (100 ct Bottle)
| Year | Estimated AWP Range (USD) |
|---|---|
| 2024 | $25 - $50 |
| 2025 | $25 - $51 |
| 2026 | $25 - $52 |
| 2027 | $24 - $53 |
| 2028 | $24 - $54 |
Note: AWP is a reference price and actual pharmacy dispensing prices will vary based on payer contracts, dispensing fees, and patient co-pays.
What are the Regulatory Considerations and Their Impact on Pricing?
Regulatory policies significantly influence the market for butalbital-aspirin-caffeine, impacting both manufacturing and pricing.
- FDA Oversight: As a prescription drug, BAC is subject to the U.S. Food and Drug Administration's (FDA) oversight for manufacturing quality, labeling, and safety.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP regulations, which are periodically updated. Compliance requires ongoing investment in facilities, processes, and quality control, contributing to manufacturing costs.
- API Sourcing Regulations: The FDA scrutinizes the origin and quality of APIs. Changes in import policies or increased enforcement of regulations related to foreign API manufacturers can affect supply chain costs and reliability.
- DEA Scheduling (Limited): While not typically classified as a Schedule II or III controlled substance in the U.S. like pure opioids or barbiturates, butalbital itself is a barbiturate. Its scheduling can vary by jurisdiction. If butalbital were to face stricter scheduling or increased monitoring requirements by the Drug Enforcement Administration (DEA) or equivalent international bodies, this could increase compliance costs and potentially impact supply availability and price, although this is considered a low-probability event for the existing BAC combination.
- Drug Efficacy and Safety Program (DESI) Review: While BAC products have been on the market for a long time, ongoing safety reviews or post-market surveillance by the FDA can lead to new labeling requirements or, in rare cases, necessitate product withdrawals or reformulation if significant safety concerns arise. Such events could disrupt supply and impact pricing.
- Generic Drug Approval Process: The FDA's Abbreviated New Drug Application (ANDA) process for generic drugs aims to ensure equivalence to brand-name drugs. The efficiency of this process influences how quickly new generic competitors can enter the market, impacting price competition.
- Payer Policies: While not direct regulatory bodies, the policies of Medicare, Medicaid, and private insurance companies heavily influence prescribing and reimbursement, effectively acting as market shapers. Formularies, prior authorization requirements, and preferred drug lists can steer physicians and patients towards or away from BAC, impacting demand and indirectly pricing power.
The overall impact of regulatory considerations is to maintain a baseline of manufacturing costs and ensure product quality and safety. However, significant changes in regulations, particularly concerning API sourcing or the classification of butalbital, could introduce greater price volatility or upward cost pressures.
What are the Key Drivers of Demand for Butalbital-Aspirin-Caffeine?
The demand for butalbital-aspirin-caffeine is driven by a combination of medical need and established clinical practice.
- Prevalence of Headache Disorders:
- Tension Headaches: These are extremely common, affecting a significant portion of the adult population. The World Health Organization (WHO) estimates that tension-type headaches are the most common type of headache, affecting up to 70% of adults [1]. BAC remains a go-to prescription for persistent or severe tension headaches.
- Migraines: While not as common as tension headaches, migraines represent a substantial patient population. Approximately 12% of the U.S. population suffers from migraines [2]. BAC is an established option for managing migraine symptoms.
- Established Prescribing Patterns: Physicians have been prescribing BAC for decades. For many clinicians, it is a familiar and trusted treatment for the conditions it targets. This inertia in prescribing habits supports sustained demand.
- Patient Familiarity and Efficacy: Patients who have found relief with BAC are likely to seek it again. The combination of ingredients provides a multi-faceted approach to pain relief that can be more effective for some than single-ingredient analgesics.
- Cost-Effectiveness (Generic): As a generic medication, BAC is generally more affordable than many branded migraine-specific treatments, making it an attractive option for patients with limited insurance coverage or high co-pays.
- Formulary Access: BAC is widely covered by insurance formularies, ensuring broad accessibility for patients.
Conversely, factors that could temper demand include:
- Availability of Newer Therapies: The development of novel migraine treatments (e.g., CGRP inhibitors) and improved OTC options provides alternatives that may be preferred by some patients and physicians due to efficacy profiles or reduced side effects.
- Concerns over Barbiturates: While BAC is generally considered safe when used as directed, butalbital is a barbiturate. Healthcare providers and patients may be increasingly cautious about prescribing or using medications containing barbiturates, especially in the context of broader concerns about prescription drug misuse.
- Side Effect Profiles: The side effect profiles of each component (e.g., gastrointestinal issues with aspirin, sedation with butalbital, potential for caffeine dependence) can influence patient preference and physician prescribing.
Key Takeaways
Butalbital-aspirin-caffeine (BAC) operates within a mature, highly competitive generic drug market. Its demand is anchored by the persistent prevalence of tension headaches and migraines, supported by established clinical practice and patient familiarity. The market is characterized by a large number of generic manufacturers, leading to significant price competition and historical price stability.
Future price projections indicate continued stability with modest fluctuations. Upward price pressures may arise from increasing raw material (API) costs, manufacturing overhead, and regulatory compliance burdens. Downward pressures will likely stem from ongoing generic competition and payer negotiations. No significant market expansion is anticipated, with growth projected in the low single digits (1-3% annually). Regulatory oversight by agencies like the FDA primarily ensures quality and safety, indirectly influencing manufacturing costs.
Frequently Asked Questions
-
What is the typical dosage strength of butalbital-aspirin-caffeine? Butalbital-aspirin-caffeine is commonly prescribed in a dosage strength of 50 mg butalbital, 325 mg aspirin, and 40 mg caffeine per capsule or tablet. Variations may exist.
-
Are there any significant side effects associated with butalbital-aspirin-caffeine? Common side effects can include dizziness, drowsiness, lightheadedness, nausea, and upset stomach. More serious side effects, though rare, can occur. The use of butalbital, a barbiturate, carries a risk of dependence.
-
Can butalbital-aspirin-caffeine be purchased over-the-counter? No, butalbital-aspirin-caffeine is a prescription-only medication in the United States and most other countries due to the presence of butalbital.
-
How does butalbital-aspirin-caffeine compare to other migraine treatments like triptans? BAC is generally considered a treatment for mild to moderate migraines and tension headaches. Triptans are specifically designed for moderate to severe migraines and target serotonin receptors to relieve migraine symptoms. The choice between BAC and triptans depends on headache severity, individual patient response, and physician recommendation.
-
What factors could lead to a significant price increase for butalbital-aspirin-caffeine in the near future? A significant price increase would likely be triggered by a widespread and sustained shortage of a key active pharmaceutical ingredient (API), particularly butalbital, due to manufacturing issues or regulatory actions affecting API suppliers. Increased global demand for APIs coupled with supply chain disruptions could also contribute.
Citations
[1] World Health Organization. (n.d.). Headache disorders. Retrieved from https://www.who.int/news-room/fact-sheets/detail/headache-disorders
[2] American Migraine Foundation. (n.d.). About Migraine. Retrieved from https://americanmigrainefoundation.org/understanding-migraine/about-migraine/
More… ↓
