Share This Page
Drug Price Trends for BUTALBITAL-ACETAMINOPHEN
✉ Email this page to a colleague
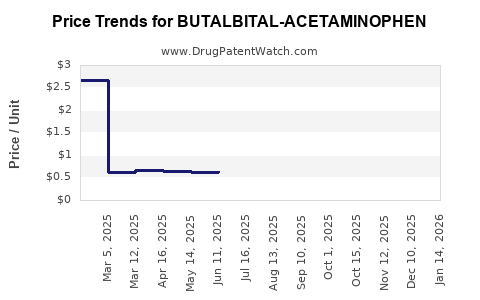
Average Pharmacy Cost for BUTALBITAL-ACETAMINOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUTALBITAL-ACETAMINOPHEN-CAFFEINE-CODEINE 50-325-40-30 MG CP | 51991-0073-01 | 0.82452 | EACH | 2026-05-20 |
| BUTALBITAL-ACETAMINOPHEN 50-325 MG TAB | 00603-2540-21 | 0.60430 | EACH | 2026-05-20 |
| BUTALBITAL-ACETAMINOPHEN 50-325 MG TAB | 35573-0456-02 | 0.60430 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is BUTALBITAL-ACETAMINOPHEN and its current market status?
Butalbitol-acetaminophen combines butalbital, a barbiturate, with acetaminophen for headache and migraine relief. It is classified as a controlled substance in the United States and other jurisdictions due to its barbiturate component. The drug has seen a decline in use owing to safety concerns, regulatory restrictions, and the emergence of newer therapies.
Market presence has diminished since the early 2000s due to increasing regulation and abuse potential concerns, leading many formulary shifts toward triptans and non-pharmacological approaches.
What are the regulatory considerations impacting market access and pricing?
The drug falls under Schedule III in the US (per DEA classification), with restrictions on prescribing and dispensing. Marketing approval continues in select markets, particularly where regulatory barriers are less stringent.
- In the US, the FDA has not approved new formulations of butalbital-acetaminophen since 2014.
- Several countries have moved to restrict or ban barbiturate-containing combination drugs altogether.
Implication: Regulatory restrictions limit market size and affect pricing potential.
How is the current market structured for butalbitol-acetaminophen?
The market is small and primarily generic, with limited branded equivalents. Key players include manufacturers like Teva, Mylan, and generic distributors.
- Market size: Estimated at under US$50 million annually in the US as of 2022.
- Pricing: Retail prices have declined, with a typical 30-tablet pack costing between US$10 and US$20 for generics.
Prescription volumes have fallen sharply, driven by safety concerns and competition from triptans, CGRP antagonists, and non-drug options.
What are the price projection trends for butalbitol-acetaminophen?
Given current regulatory and market dynamics:
| Year | Projected Market Size (USD millions) | Price Trends | Key Factors |
|---|---|---|---|
| 2023 | 40-50 | Stable but declining | Regulatory restrictions, declining prescriptions |
| 2024-2025 | 35-45 | Slight decline, possible stabilization | Continued shift to new therapies |
| 2026-2030 | 30-40 | Minor fluctuations; possible further decline | Growing preference for non-barbiturate migraine treatments |
The decline reflects a shrinking niche, with no substantial growth expected unless regulatory or formulary changes occur.
What are alternative therapies impacting the market?
- Triptans: Sumatriptan, rizatriptan; dominant migraine-specific drugs.
- CGRP inhibitors: Erenumab, fremanezumab; increased adoption.
- NSAIDs: Over-the-counter and prescription options.
- Non-drug therapies: Behavioral modifications, neuromodulation.
These alternatives are reducing demand for butlbitol-acetaminophen as a primary therapy.
What pricing strategies could influence future values?
Limited opportunities exist for premium pricing due to generic status and regulatory constraints. Manufacturers may seek to:
- Reduce prices further to maintain volume.
- Formulate combination products with newer agents, though regulatory hurdles are high.
- Focus marketing on niche markets with fewer alternatives.
What are the key risks and opportunities?
Risks:
- Regulatory bans in major markets.
- Physician preference shifting toward more effective therapies.
- Potential litigation related to safety concerns.
Opportunities:
- Development of reformulated, safer barbiturate combinations.
- Entry into emerging markets with less stringent regulations.
- Niche marketing for patients with specific needs unserved by newer drugs.
Summary
The market for butalbital-acetaminophen is in decline due to safety and regulatory issues, with global sales under US$50 million. Price projections indicate continued shrinkage, averaging around US$30-40 million in global revenues over the next five years. Regulatory trends and the availability of more effective, safer alternatives undermine market stability. Opportunities involve niche applications and emerging market penetration, but substantial growth prospects remain limited.
Key Takeaways
- The drug’s market is declining due to safety concerns and regulatory restrictions.
- US sales are under US$50 million annually, with prices under US$20 per pack.
- Growth prospects are limited; current trends suggest further market contraction.
- Alternative migraine therapies reduce demand for butalbitol-acetaminophen.
- Future strategies should focus on niche markets and regulatory navigation.
FAQs
1. Why has the use of butalbitol-acetaminophen decreased?
Safety concerns, regulatory restrictions, and the availability of better, more targeted therapies have led to decreased prescriptions.
2. Which regions have the most lenient regulations on barbiturate-containing drugs?
Some emerging markets and countries with less restrictive drug schedules permit prescriptions, but overall restrictions are tightening globally.
3. Are there any potential markets for reintroducing butalbitol-acetaminophen?
Limited, primarily in regions with lax regulation or for specific patient subsets with contraindications to newer therapies.
4. What are the main safety concerns associated with butalbitol-acetaminophen?
Risk of dependence, respiratory depression, and liver toxicity due to acetaminophen.
5. How might future regulatory changes impact pricing?
Easing restrictions could support minor price stabilization or increases, but current trends favor price reductions due to declining demand.
Sources:
- FDA Drug Database. (2022). https://www.accessdata.fda.gov
- IQVIA. (2022). Global Analysis of Migraine Drugs.
- DEA Scheduling for Barbiturates. (2022). https://www.deadiversion.usdoj.gov
- Pharmaceutical Market Reports. (2022). U.S. Prescription Drug Trends.
- Prescriber Trends in Headache Management. (2021). Journal of Pain Management.
More… ↓
