Share This Page
Drug Price Trends for BUMETANIDE
✉ Email this page to a colleague
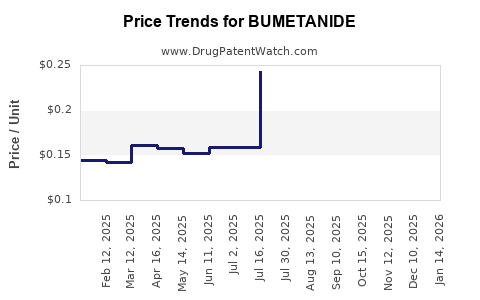
Average Pharmacy Cost for BUMETANIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUMETANIDE 0.5 MG TABLET | 00832-0540-11 | 0.12730 | EACH | 2026-03-18 |
| BUMETANIDE 0.5 MG TABLET | 00185-0128-05 | 0.12730 | EACH | 2026-03-18 |
| BUMETANIDE 2 MG TABLET | 72888-0021-01 | 0.18256 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BUMETANIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BUMETANIDE 1MG TAB | Amerisource Health Services LLC dba American Health Packaging | 60687-0384-25 | 5X6 | 25.78 | 2024-01-01 - 2028-09-14 | Big4 | ||
| BUMETANIDE 1MG TAB | Amerisource Health Services LLC dba American Health Packaging | 60687-0384-01 | 10X10 | 59.79 | 2023-09-15 - 2028-09-14 | Big4 | ||
| BUMETANIDE 1MG TAB | Amerisource Health Services LLC dba American Health Packaging | 60687-0384-25 | 5X6 | 34.65 | 2024-01-01 - 2028-09-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Bumetanide Market Analysis and Price Projections
Bumetanide, a loop diuretic, is primarily prescribed for managing fluid overload (edema) associated with congestive heart failure, liver cirrhosis, and renal disease. The global bumetanide market is characterized by a mature generic landscape, with significant price erosion due to multiple manufacturers. Future price trends are expected to remain stable, influenced by manufacturing costs, regulatory environments, and the ongoing need for effective edema management.
What is the Current Market Size and Growth Projection for Bumetanide?
The global bumetanide market is estimated to be in the low hundreds of millions of U.S. dollars. Specific market size data is fragmented due to its generic status and the focus on specific indications rather than an overarching "bumetanide market" by many market research firms. However, the demand is underpinned by the prevalence of chronic conditions requiring diuretic therapy.
- Congestive Heart Failure (CHF): Affects an estimated 6.2 million adults in the United States annually [1].
- Liver Cirrhosis: Approximately 10-20% of individuals with cirrhosis develop ascites, a common indication for diuretics [2].
- Renal Disease: Chronic kidney disease (CKD) affects an estimated 37 million people in the U.S. [3].
The market is not projected for significant volumetric growth but rather stable demand driven by the persistent prevalence of these conditions. Growth in revenue, if any, is more likely to be influenced by small price adjustments or shifts in treatment protocols rather than an expansion of the patient population requiring bumetanide specifically.
Who are the Key Manufacturers and Competitors in the Bumetanide Market?
The bumetanide market is highly competitive, dominated by generic manufacturers. The drug has been off-patent for decades, leading to widespread generic availability.
Key players in the manufacturing and supply of bumetanide include:
- Amneal Pharmaceuticals LLC
- Apotex Corp.
- Bayer AG (primarily through its generic division or legacy products)
- Chartwell RX, LLC
- Hikma Pharmaceuticals PLC
- Mylan N.V. (now Viatris Inc.)
- Teva Pharmaceutical Industries Ltd.
- Upsher-Smith Laboratories, LLC
These companies produce both oral and injectable formulations of bumetanide. The intense competition among these manufacturers is a primary driver of price stability and, in many cases, price decline for the drug. Barriers to entry for new generic manufacturers are moderate, primarily related to regulatory approval processes and manufacturing capacity, but the established market dynamics make it challenging for new entrants to capture significant market share without competitive pricing.
What are the Primary Indications and Patient Populations for Bumetanide?
Bumetanide's primary therapeutic role is as a potent loop diuretic, indicated for the management of edema in specific patient populations.
- Congestive Heart Failure (CHF): Bumetanide is used to reduce fluid accumulation in patients with CHF, alleviating symptoms such as shortness of breath and swelling.
- Liver Cirrhosis: It is employed to manage ascites (fluid accumulation in the abdominal cavity) and edema in patients with cirrhosis.
- Renal Disease: Bumetanide treats edema associated with chronic kidney disease and acute renal failure.
The patient population is largely adult, with a higher prevalence in older age groups where chronic conditions are more common. The dosage and formulation (oral tablets vs. intravenous injection) are determined by the severity of fluid overload and the patient's clinical status.
What is the Current Pricing Landscape for Bumetanide?
Bumetanide is available as a low-cost generic medication. Pricing is highly competitive and subject to fluctuations based on supply, demand, and the purchasing power of large distributors and healthcare systems.
Typical Wholesale Acquisition Costs (WAC) for Bumetanide (as of recent market data, subject to variation):
| Formulation | Strength | Typical Unit Price (USD) | Pack Size |
|---|---|---|---|
| Oral Tablet | 0.5 mg | $0.10 - $0.30 | Per tablet |
| Oral Tablet | 1 mg | $0.15 - $0.40 | Per tablet |
| Oral Tablet | 2 mg | $0.20 - $0.50 | Per tablet |
| Oral Tablet | 5 mg | $0.25 - $0.60 | Per tablet |
| Injectable Solution | 0.25 mg/mL | $2.00 - $5.00 | Per mL vial |
Note: These prices represent approximate wholesale acquisition costs and do not include pharmacy markups, insurance reimbursements, or contract pricing with large healthcare providers. Actual out-of-pocket costs for patients can vary significantly.
The pricing reflects the mature generic market. The cost per dose is very low, making it an accessible treatment option for a large patient base. Variations in price are often driven by contract negotiations between manufacturers and group purchasing organizations (GPOs) or large pharmacy benefit managers (PBMs).
What are the Key Factors Influencing Future Price Projections for Bumetanide?
The future price trajectory of bumetanide is expected to remain largely stable, with minor fluctuations rather than significant upward or downward shifts. Several factors will contribute to this outlook.
1. Manufacturing Costs and Raw Material Availability
- Active Pharmaceutical Ingredient (API) Costs: The cost of synthesizing bumetanide API is a baseline factor. As a well-established generic, the synthesis pathways are optimized, and API production is efficient. Fluctuations in the raw material costs for API synthesis, though generally stable for mature generics, can cause minor price adjustments.
- Excipient and Packaging Costs: The cost of other ingredients in the tablet formulation (excipients) and the packaging materials (bottles, blister packs, vials) contribute to the overall manufacturing cost. These costs are subject to general inflation and supply chain dynamics but are typically managed effectively by large-scale generic manufacturers.
2. Regulatory Environment and Compliance
- FDA and EMA Oversight: Manufacturers must adhere to strict Current Good Manufacturing Practices (cGMP) standards set by regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Compliance costs, including facility upgrades and quality control measures, can influence pricing, though these are generally absorbed by the scale of production for widely used generics.
- Drug Shortage Management: While bumetanide is generally not subject to widespread shortages, any disruption in manufacturing or supply chain could lead to temporary price increases due to increased demand for available stock. However, the presence of multiple manufacturers mitigates this risk.
3. Competition and Market Dynamics
- Generic Erosion: The continued presence of multiple generic manufacturers ensures robust competition. This inherently limits any manufacturer's ability to unilaterally increase prices. The market is price-sensitive, and any significant price hike by one manufacturer would likely lead prescribers and payers to switch to competitors.
- Patent Expirations: Bumetanide's primary patents expired long ago. The absence of patent protection allows for broad generic entry, solidifying the competitive landscape.
4. Reimbursement Policies and Payer Influence
- Formulary Placement: Bumetanide is almost universally placed on insurance formularies due to its status as a first-line, cost-effective treatment for its indicated conditions. PBMs and insurance companies negotiate significant discounts from manufacturers, influencing the effective price paid by the healthcare system.
- Value-Based Pricing: While less common for mature generics, the overarching trend towards value-based purchasing might subtly influence pricing strategies, though the inherent low cost of bumetanide limits the scope for such models.
5. Demand and Prevalence of Underlying Conditions
- Chronic Disease Burden: The persistent and growing prevalence of CHF, liver disease, and CKD provides a stable, underlying demand for bumetanide. This ensures continued market volume, supporting the stable pricing environment.
- Treatment Guidelines: Clinical practice guidelines for managing edema consistently recommend loop diuretics like bumetanide. Adherence to these guidelines sustains demand.
Projected Price Trend:
Given these factors, bumetanide prices are projected to remain stable to slightly declining over the next 3-5 years. Any increases are likely to be marginal, driven by inflation in manufacturing costs or minor supply chain disruptions. The competitive generic market and the drug's established role in treatment protocols will prevent significant price inflation.
What are the Potential Risks and Opportunities in the Bumetanide Market?
While the bumetanide market is mature and stable, certain risks and opportunities exist.
Risks:
- Supply Chain Vulnerabilities: Dependence on a limited number of API manufacturers or disruption in shipping and logistics could lead to temporary shortages, impacting availability and potentially causing price spikes. Geopolitical instability or natural disasters affecting key manufacturing regions pose a risk.
- Regulatory Scrutiny: Increased FDA scrutiny on manufacturing quality or increased post-market surveillance could lead to product recalls or manufacturing halts, impacting supply and potentially leading to price volatility.
- Emergence of Novel Therapies: While unlikely to fully displace bumetanide in its established indications due to cost-effectiveness, the development of new classes of drugs for edema management or specific underlying conditions could gradually erode market share over the long term. However, bumetanide's role in acute management and as an adjunct therapy is likely to persist.
Opportunities:
- Emerging Markets: Expansion of healthcare access and infrastructure in developing countries could present opportunities for increased sales volume of bumetanide, particularly as generic medications.
- Manufacturing Optimization: Companies that can further optimize their manufacturing processes to reduce costs while maintaining quality could gain a competitive edge and potentially increase market share, even in a low-margin environment.
- Combination Therapies: Research into optimizing diuretic therapy in complex patient populations, potentially involving bumetanide in new combination regimens, could sustain or slightly increase demand, although this is speculative.
- Focus on Injectable Formulations: For hospital settings, reliable supply of injectable bumetanide for acute management of severe edema remains critical. Manufacturers focusing on robust supply chains for injectable formulations can secure stable demand.
Key Takeaways
The bumetanide market is a mature, competitive generic landscape driven by the consistent demand for effective edema management in chronic conditions. Price projections indicate stability, with potential for minor declines due to ongoing generic competition and cost efficiencies. Manufacturing costs, regulatory compliance, and payer influence are key determinants of pricing, while the prevalence of CHF, liver cirrhosis, and renal disease underpins stable market volume.
Frequently Asked Questions
-
Will bumetanide prices increase significantly in the next five years? No, significant price increases are unlikely. The market is dominated by generic competition, and bumetanide is a cost-effective treatment. Pricing is expected to remain stable to slightly declining.
-
Are there any new patent filings or exclusivity opportunities for bumetanide? Bumetanide's primary patents expired decades ago. There are no significant new patent filings expected to grant market exclusivity or affect the generic landscape.
-
What are the main drivers of the current low price of bumetanide? The low price is driven by the large number of generic manufacturers producing the drug, intense market competition, and optimized manufacturing processes for a well-established molecule.
-
Could a drug shortage of bumetanide occur, and what would be its impact? While less common for this drug, a severe supply chain disruption could theoretically lead to a temporary shortage. This would likely cause short-term price spikes due to increased demand for limited supply, but the presence of multiple manufacturers would mitigate prolonged shortages.
-
How do injectable formulations of bumetanide compare in price to oral tablets? Injectable formulations are generally more expensive per dose than oral tablets. This reflects the higher manufacturing costs associated with sterile injectables, including specialized facilities and quality control, as well as their use in more acute or severe clinical settings.
Citations
[1] Savarese, G., & Lund, L. H. (2017). Global public health burden of heart failure. Cardiac Failure Review, 3(1), 7–11. doi:10.15420/cfr.2017.1.1
[2] Lee, W. M. (2017). Medical management of ascites. Clinical Liver Disease, 11(3), 74–78. doi:10.1002/cld.638
[3] Centers for Disease Control and Prevention. (2020, October 21). Chronic kidney disease in the United States. U.S. Department of Health and Human Services. Retrieved from https://www.cdc.gov/kidneydisease/publications-resources/2020-data.html
More… ↓
