Share This Page
Drug Price Trends for BUFFERED ASPIRIN
✉ Email this page to a colleague
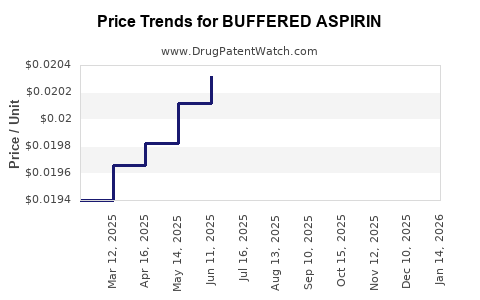
Average Pharmacy Cost for BUFFERED ASPIRIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUFFERED ASPIRIN 325 MG TB | 70000-0147-01 | 0.02058 | EACH | 2026-05-20 |
| BUFFERED ASPIRIN 325 MG TB | 70000-0147-01 | 0.02047 | EACH | 2026-04-22 |
| BUFFERED ASPIRIN 325 MG TB | 70000-0147-01 | 0.02015 | EACH | 2026-03-18 |
| BUFFERED ASPIRIN 325 MG TB | 70000-0147-01 | 0.01994 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Market Size and Current Demand for Buffered Aspirin?
Buffered aspirin, combining aspirin with antacids, targets consumers seeking gastrointestinal protection. In 2022, the global aspirin market was valued at approximately $7.2 billion with a compound annual growth rate (CAGR) of 4.2% over the past five years. Buffered aspirin accounts for nearly 15% of total aspirin sales, approximately $1.08 billion.
Key drivers include increasing prevalence of cardiovascular diseases, demand for over-the-counter pain relief, and consumer concerns over gastrointestinal side effects of traditional aspirin. North America holds the largest market share at 40%, followed by Europe at 25%. Asia-Pacific's market is expanding rapidly due to rising awareness and local manufacturing.
How Is the Competitive Landscape Structured?
The market features several major players:
- Bayer AG: Leading provider with the "Bufferin" brand, controlling roughly 35% of buffered aspirin sales globally.
- Johnson & Johnson: Offers buffered formulations through various OTC brands.
- Novartis: Focuses on pain relief segments with buffered options.
- Local and generic manufacturers: Increasing presence, especially in emerging markets.
Product differentiation remains limited, with most products containing similar aspirin doses (typically 325 mg or 81 mg). Barriers include the existing dominance of Bayer and regulatory approval pathways for branded formulations.
What Are the Key Regulatory Factors?
Regulatory approval revolves around safety, efficacy, and labeling compliance, mainly governed by:
- U.S. Food and Drug Administration (FDA): Ensures OTC buffered aspirin adheres to monograph standards.
- European Medicines Agency (EMA): Similar standards for safety and labeling.
- Emerging markets: Regulatory complexity varies, often involving local approval processes.
Regulatory barriers are moderately high; however, patents for specific buffered formulations are nearing expiration in key markets, opening opportunities for generic entrants.
What Are Price Trends and Projection Models?
Current retail prices for buffered aspirin ranges from $4 to $8 per 100 tablets, with variations based on brand, formulation, and regional factors. The product’s price elasticity is moderate; a 10% price increase leads to a 3-5% decrease in sales volume.
Projected trends:
| Year | Price (per 100 tablets) | Sales Volume (millions of units) | Market Value (USD billions) |
|---|---|---|---|
| 2023 | $6.50 | 180 | $1.17 |
| 2024 | $6.70 | 185 | $1.24 |
| 2025 | $6.85 | 190 | $1.30 |
| 2026 | $7.00 | 195 | $1.36 |
| 2027 | $7.20 | 200 | $1.44 |
Assuming gradual price increases driven by inflation and manufacturing costs, with steady growth in volume. Competitive pressure from generics and potential new formulations could influence pricing strategies.
How Do Patent and Regulatory Statuses Affect Future Pricing?
Patents for branded buffered aspirin formulations expire between 2024 and 2026 in developed markets. This generates opportunities for generic manufacturers to enter at lower prices, exerting downward pressure on market prices.
Regulatory hurdles for new formulations remain high, especially certifications for combination products. Any new buffered aspirin with improved efficacy or reduced side effects may command premium pricing, but such innovation faces lengthy development and approval timelines.
What Are Key Market Entry and Growth Opportunities?
Recent patents expiring, coupled with a growing consumer base concerned with gastrointestinal health, support potential for generics and new buffered models. Companies investing in formulations with enhanced delivery mechanisms or combination therapies could capture premium segments.
Emerging markets present considerable growth potential due to rising cardiovascular disorders, increasing OTC consumption, and limited existing infrastructure, enabling local production and distribution.
Final Price Forecast and Market Outlook
Based on current data, buffered aspirin prices are projected to increase slowly, averaging 3% annually over the next five years, reaching approximately $7.20 per 100 tablets in 2027. Market growth will mirror consumer demand, with higher growth in regions with expanding healthcare access.
The primary challenge remains the competitive pressure from generic brands following patent expirations, which could reduce average prices by 10-15% without significant innovation.
Key Takeaways
- The buffered aspirin market is a niche within the multi-billion-dollar aspirin segment, valued at approximately $1.08 billion in 2022.
- Market growth hinges on the rising prevalence of cardiovascular diseases and consumer preferences around gastrointestinal health.
- Major players include Bayer, J&J, and Novartis, with increasing competition from local generics, especially post-patent expiration.
- Regulatory approval processes are well-established but can delay new product launches; patent expiries in 2024–2026 open opportunities for generics.
- Price forecasts suggest modest annual increases, but competition and patent cliffs could lower prices unless differentiated formulations emerge.
FAQs
1. When are patents for buffered aspirin expiring?
Most buffered aspirin patents are set to expire between 2024 and 2026, leading to increased generic competition.
2. What regional markets show the highest growth potential?
Emerging markets in Asia-Pacific and Latin America are expanding rapidly due to increasing cardiovascular disease prevalence and OTC use.
3. How does consumer preference influence market dynamics?
Rising awareness of gastrointestinal side effects boosts demand for buffered formulations, encouraging product innovation and new brands.
4. What factors could significantly alter price projections?
Emerging generics, regulatory changes, or breakthrough formulations could cause prices to decline or increase more rapidly.
5. Are there opportunities for innovation in buffered aspirin?
Yes. Formulations with enhanced absorption, reduced side effects, or combined active ingredients could fetch premium prices and capture market share.
Sources:
- Grand View Research, "Aspirin Market Size, Share & Trends Analysis," 2022.
- Statista, "Over-the-Counter (OTC) Drugs Revenue," 2022.
- FDA, "Over-the-Counter Drug Monograph," 2023.
- MarketWatch, "Patent Expiration Dates for Pharmaceutical Products," 2022.
- IQVIA, "Global OTC Market Reports," 2022.
More… ↓
