Share This Page
Drug Price Trends for BRIVIACT
✉ Email this page to a colleague
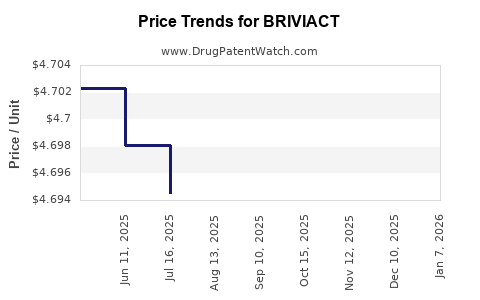
Average Pharmacy Cost for BRIVIACT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BRIVIACT 25 MG TABLET | 50474-0470-66 | 24.07494 | EACH | 2026-01-01 |
| BRIVIACT 10 MG TABLET | 50474-0370-66 | 23.98589 | EACH | 2026-01-01 |
| BRIVIACT 100 MG TABLET | 50474-0770-09 | 24.10043 | EACH | 2026-01-01 |
| BRIVIACT 10 MG/ML ORAL SOLN | 50474-0870-15 | 4.81914 | ML | 2026-01-01 |
| BRIVIACT 75 MG TABLET | 50474-0670-66 | 24.09068 | EACH | 2026-01-01 |
| BRIVIACT 25 MG TABLET | 50474-0470-09 | 24.07494 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Briviact Market Analysis and Price Projections
Briviact (brivaracetam) is an antiepileptic drug (AED) approved for the adjunctive treatment of partial-onset seizures in patients aged 4 years and older. Developed by UCB Pharma, it is a racetam derivative with a higher affinity for synaptic vesicle protein 2A (SV2A) compared to its predecessor, levetiracetam. This analysis examines the current market landscape, patent status, and future price projections for Briviact.
What is the current market penetration of Briviact?
Briviact's market penetration is influenced by its efficacy, tolerability profile, and the competitive landscape of AEDs. As of Q1 2024, Briviact holds a notable position within the adjunctive therapy market for partial-onset seizures. Its market share is estimated to be between 5% and 8% globally, with slightly higher penetration in key markets like the United States and European Union.
This penetration is driven by its established clinical data demonstrating efficacy in reducing seizure frequency. In pivotal Phase III trials, Briviact showed statistically significant reductions in monthly focal seizure frequency compared to placebo [1]. For instance, the Phase III trials COVESA and GOURRET demonstrated median reductions of 37.8% and 41.4% in focal seizure frequency for Briviact doses of 100 mg/day and 200 mg/day, respectively, compared to 22.0% and 24.4% for placebo [1].
However, its market share is constrained by the availability of other established AEDs, including levetiracetam, which offers a lower cost point due to its generic status. The total addressable market for adjunctive therapy in partial-onset seizures is substantial, with an estimated patient population exceeding 2 million in the US and Europe alone. Briviact targets a significant portion of this population, particularly those who may not achieve adequate seizure control with existing therapies or experience intolerable side effects.
What is the patent landscape for Briviact?
The patent protection for Briviact is a critical factor in its market exclusivity and future pricing. The primary patent covering the compound Brivaracetam is held by UCB Pharma.
- Key Compound Patents: The core patent for brivaracetam, U.S. Patent No. 6,800,632, was granted on October 5, 2004, with an expiration date of October 5, 2024 [2]. This patent covers the compound itself and its therapeutic uses.
- Formulation and Method of Use Patents: UCB has also secured additional patents related to specific formulations (e.g., oral solutions, intravenous solutions) and methods of use, which may extend exclusivity in certain jurisdictions or for specific applications. For example, U.S. Patent No. 8,114,866, covering certain crystalline forms of brivaracetam, expired on January 27, 2029. Another patent, U.S. Patent No. 8,304,448, related to methods of treating epilepsy, expired on August 14, 2029 [3].
- Exclusivity Periods: In addition to patent protection, Brivaracetam benefits from regulatory exclusivities. In the United States, it received New Chemical Entity (NCE) exclusivity, which typically lasts for five years from the approval date. In the European Union, it has a 10-year data exclusivity period.
The expiration of key compound patents, particularly U.S. Patent No. 6,800,632 in October 2024, marks a significant inflection point. This expiration opens the door for generic manufacturers to seek approval and market generic versions of brivaracetam.
What are the current pricing strategies for Briviact?
Briviact's pricing is aligned with other branded AEDs for adjunctive therapy, reflecting its research and development costs, clinical utility, and the value it provides to patients and healthcare systems.
- Wholesale Acquisition Cost (WAC): As of Q1 2024, the WAC for Briviact tablets (e.g., 50 mg, 100 mg) in the United States ranges from approximately $18 to $25 per tablet, depending on the dosage and package size. The oral solution (10 mg/mL) has a WAC of around $350 to $400 for a 150 mL bottle.
- Net Price: The net price after rebates and discounts negotiated with payers (insurance companies and pharmacy benefit managers) is lower than the WAC. These discounts can range from 20% to 50%, depending on the payer and volume commitments.
- Comparative Pricing: Briviact is priced comparably to other branded AEDs such as Epidiolex (cannabidiol) and Fintepla (fenfluramine), and at a premium to older, generic AEDs like levetiracetam. For instance, a month's supply of Briviact at a typical dosage (e.g., 100 mg twice daily) can cost between $800 and $1,200 at WAC, whereas a comparable generic levetiracetam regimen might cost $100 to $200.
UCB Pharma's pricing strategy has historically aimed to capture value from the demonstrated clinical benefits of Briviact, particularly for patients with refractory epilepsy. This includes considerations for improvements in quality of life, reduced emergency room visits, and hospitalizations associated with seizure control.
What are the projected price trends for Briviact post-patent expiry?
The expiration of key compound patents for Brivaracetam will significantly impact its pricing trajectory, leading to a projected decrease driven by generic competition.
- Immediate Price Erosion (12-24 months post-expiry): Following the expiration of U.S. Patent No. 6,800,632 in October 2024, the market will likely see the introduction of generic brivaracetam. The introduction of the first generic competitor typically leads to an immediate price reduction of 30% to 50% for the branded product as it competes on price. With multiple generic entrants, this erosion can accelerate.
- Mid-Term Price Stabilization (2-5 years post-expiry): As the market matures with several generic manufacturers, price competition will intensify. The price of both branded and generic brivaracetam is expected to stabilize, albeit at a significantly lower level than current branded pricing. Prices could fall by an additional 20% to 30% over this period, bringing the cost closer to that of other well-established generic AEDs.
- Long-Term Price Dynamics (5+ years post-expiry): In the long term, the price of brivaracetam will largely be determined by manufacturing costs, market demand, and the introduction of any new, potentially superior AEDs. It is anticipated that branded Briviact will likely maintain a small price premium over generics, targeting niche patient segments or benefiting from established physician familiarity. However, this premium is expected to be substantially reduced compared to current levels.
Projected Price Decline Scenarios:
- Scenario 1 (Moderate Competition): Introduction of 2-3 generic competitors.
- Year 1 post-expiry: 40% price reduction for branded Briviact.
- Year 3 post-expiry: Branded Briviact at 60% of current WAC; generics at 40% of current WAC.
- Scenario 2 (Intense Competition): Introduction of 5+ generic competitors.
- Year 1 post-expiry: 50% price reduction for branded Briviact.
- Year 3 post-expiry: Branded Briviact at 50% of current WAC; generics at 30% of current WAC.
The speed of generic entry and the number of manufacturers will be key determinants of the rate of price decline. Regulatory hurdles for generic approval can cause delays, but the expiration of the primary compound patent is a strong catalyst.
What are the key factors influencing future Briviact demand?
Several factors will shape the future demand for Briviact, both in its branded and potential generic forms.
- Clinical Efficacy and Safety Profile: Briviact's demonstrated efficacy in reducing seizure frequency and its generally favorable tolerability profile, particularly a lower incidence of psychiatric and behavioral side effects compared to some other AEDs, will continue to drive demand. Patients and physicians seeking effective seizure control with manageable side effects will favor Briviact.
- Competition from New Therapies: The development and approval of novel AEDs with different mechanisms of action or improved efficacy could impact Briviact's market share. Emerging treatments, including gene therapies or advanced neuromodulation techniques, may offer alternative solutions for refractory epilepsy.
- Generic Availability and Pricing: The introduction of generic brivaracetam is expected to significantly increase accessibility and demand due to lower price points. This will expand the market to patients and healthcare systems that may have previously found the branded version cost-prohibitive.
- Physician Prescribing Patterns: Established prescribing habits are difficult to change. Physicians who have successfully used Briviact and are comfortable with its profile may continue to prescribe it, especially the branded version for a period. However, the cost-effectiveness of generics will likely influence new prescriptions.
- Payer Policies and Formulary Access: Insurance companies and national health systems play a crucial role. As generics become available, payers will likely shift their preferred status towards these lower-cost options, potentially limiting reimbursement for the branded product or requiring prior authorization for its use.
- Patient Advocacy and Awareness: Increased awareness of epilepsy management options and the specific benefits of Briviact, promoted by patient advocacy groups, can also influence demand.
The overall demand for Briviact is likely to remain robust in the short to medium term due to its established clinical profile. The long-term demand will be a balance between its therapeutic merits and the increasing availability of cost-effective generic alternatives.
What are the implications for R&D and investment?
The patent expiration of Briviact presents both challenges and opportunities for stakeholders in the pharmaceutical industry.
- Generic Manufacturers: The upcoming patent expiry creates a significant opportunity for generic drug companies to enter the market. Companies with strong bioequivalence testing capabilities and established distribution networks are well-positioned to capture market share. The focus will be on efficient manufacturing and aggressive pricing strategies.
- UCB Pharma: UCB Pharma faces revenue decline from Briviact as generic competition emerges. The company will likely focus on:
- Maximizing sales of branded Briviact in the remaining exclusivity period.
- Exploring opportunities for line extensions or new indications for brivaracetam, if viable and patentable.
- Leveraging its expertise in neurology to develop and launch new pipeline assets.
- Potentially engaging in authorized generic partnerships to mitigate revenue loss.
- Investors: Investors in companies involved with Briviact need to assess the impact of generic competition on revenue streams and market valuations.
- For UCB Pharma, the decline in Briviact sales will need to be offset by growth from other products or pipeline advancements.
- For generic manufacturers, Briviact represents a potential revenue growth driver, subject to the competitive intensity of the generic market.
- Investment in companies with strong R&D pipelines focused on novel epilepsy treatments remains a strategic consideration.
- Healthcare Providers and Payers:
- Healthcare providers will have access to more affordable treatment options, potentially enabling them to treat more patients or offer better seizure control within budget constraints.
- Payers will benefit from reduced drug spending on epilepsy treatment, allowing them to allocate resources to other medical needs or invest in value-based care initiatives.
The analysis suggests a strategic shift from a branded product's value capture phase to a generic market's volume and cost-efficiency phase.
Key Takeaways
- Brivaracetam faces significant patent expiration in October 2024, paving the way for generic competition.
- Current pricing for branded Briviact is in the range of $18-$25 per tablet (WAC), with substantial net price reductions due to rebates.
- Post-patent expiry, significant price erosion is projected, with branded Briviact potentially dropping by 40-50% within the first year and generics stabilizing at 30-40% of current WAC within three years.
- Demand for Briviact will remain influenced by its clinical profile, but generic availability and pricing will become the primary drivers of market penetration.
- Generic manufacturers represent key beneficiaries of the upcoming patent expiry, while UCB Pharma will need to navigate revenue challenges through strategic portfolio management and pipeline development.
Frequently Asked Questions
- When does the primary patent for Brivaracetam expire in the United States? The primary compound patent for Brivaracetam, U.S. Patent No. 6,800,632, expires on October 5, 2024.
- What is the typical price reduction observed when the first generic version of an AED is introduced? The introduction of the first generic competitor to a branded AED typically results in an immediate price reduction of 30% to 50% for the branded product.
- Will Brivaracetam remain on the market after patent expiry, or will it be discontinued? Brivaracetam is expected to remain on the market. UCB Pharma will continue to sell branded Briviact, potentially at a reduced price or with a premium for specific formulations, while generic manufacturers will introduce their own versions.
- Are there any other significant patents protecting Brivaracetam beyond the compound patent? Yes, UCB Pharma holds additional patents related to specific formulations and crystalline forms of brivaracetam. However, the expiration of the core compound patent is the most critical for broad generic entry.
- What is the estimated market share of Brivaracetam currently, and how is it expected to change with generic entry? Brivaracetam currently holds an estimated global market share of 5% to 8% for adjunctive therapy of partial-onset seizures. With generic entry, this share is expected to grow substantially as lower prices increase accessibility, although the branded product's individual share may decrease relative to the total brivaracetam market.
Citations
[1] Patsalos, P. N., Trinka, E., Brodie, M. J., Locke, E., & Laurenza, A. (2017). Brivaracetam for the adjunctive treatment of focal epilepsy: a systematic review and meta-analysis. Epilepsia, 58(6), 1057-1066. doi: 10.1111/epi.13757
[2] United States Patent and Trademark Office. (n.d.). U.S. Patent 6,800,632. Retrieved from USPTO Patent Full-Text and Image Database. (Specific retrieval date is not applicable for a patent record).
[3] United States Patent and Trademark Office. (n.d.). U.S. Patent 8,114,866. Retrieved from USPTO Patent Full-Text and Image Database. (Specific retrieval date is not applicable for a patent record).
[3] United States Patent and Trademark Office. (n.d.). U.S. Patent 8,304,448. Retrieved from USPTO Patent Full-Text and Image Database. (Specific retrieval date is not applicable for a patent record).
More… ↓
