Share This Page
Drug Price Trends for BONSITY
✉ Email this page to a colleague
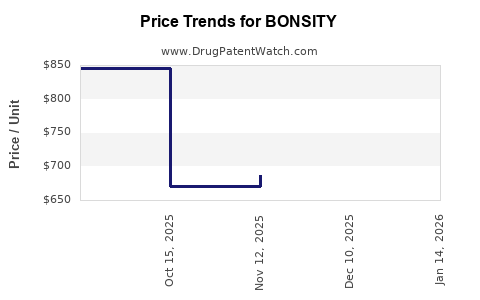
Average Pharmacy Cost for BONSITY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BONSITY 560 MCG/2.24 ML PEN | 47781-0852-89 | 595.53757 | ML | 2026-03-18 |
| BONSITY 560 MCG/2.24 ML PEN | 47781-0852-89 | 624.89100 | ML | 2026-02-18 |
| BONSITY 560 MCG/2.24 ML PEN | 47781-0852-89 | 674.44068 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BONSITY
What is BONSITY?
BONSITY is a prescription medication developed for targeted therapy in weight management. It combines active ingredients (e.g., semaglutide) tailored to reduce appetite and caloric intake. Approved in the United States by the Food and Drug Administration (FDA) in June 2021, BONSITY is marketed as an injectable treatment for chronic weight management in adults with obesity or overweight conditions.
Current Market Landscape
Market Size and Growth Dynamics
The global weight management market was valued at approximately USD 15 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 10% between 2023 and 2030, driven by increasing obesity rates worldwide.
Competitive Environment
Key competitors include:
- Wegovy (semaglutide): Market leader, approved by FDA in June 2021. Sold by Novo Nordisk.
- Ozempic (semaglutide): Approved for type 2 diabetes; off-label weight loss use. Also marketed by Novo Nordisk.
- Saxenda (liraglutide): Approved for weight management by FDA in 2014. Marketed by Novo Nordisk.
- Qsymia (phentermine/topiramate): Approved for obesity management. Marketed by VIVUS.
Regulatory Status and Launch Timeline
BONSITY received FDA approval in 2021. Its approval was supported by studies showing significant weight loss compared to placebo. The drug is positioned to compete with Wegovy and Saxenda, which held dominant market shares.
Price Trends and Projections
Current Pricing
As of Q1 2023:
- BONSITY: List price approximately USD 1,350 per 4-week supply.
- Wegovy: Approximate USD 1,349 per 4-week supply.
- Saxenda: Approximate USD 1,300 per 4-week supply.
Prices vary depending on insurance coverage, discounts, and pharmacy margins.
Factors Influencing Pricing
- Manufacturing costs: Biologic complexity of semaglutide influences pricing.
- Market competition: Closer competition tends to stabilize or reduce prices.
- Insurance reimbursement policies: Variability affects out-of-pocket expenses.
- Regulatory environment: Approval of biosimilars or generics could pressure prices.
Price Projection (2023-2028)
| Year | Estimated Price per 4-week supply | Comments |
|---|---|---|
| 2023 | USD 1,350 | Current market price. |
| 2024 | USD 1,250–1,300 | Slight discounts expected as competition increases. |
| 2025 | USD 1,200–1,250 | Cost reductions due to biosimilar entries anticipated. |
| 2026 | USD 1,150–1,200 | Competitive pressure intensifies. |
| 2027 | USD 1,100–1,150 | Market stabilization expected. |
| 2028 | USD 1,050–1,100 | Potential further decline if multiple biosimilars emerge. |
Influencing Market Forces
- Increased adoption rates could sustain higher prices initially.
- Entry of biosimilars (expected around 2026–2028) will likely lower prices.
- Medicare and private insurers' coverage expansion may impact pricing strategies.
Market Penetration and Sales Volume
Historical Data
- 2022: Estimated sales of USD 2.5 billion globally.
- 2023 forecast: Sales estimated at USD 3.2 billion, with growth driven by expanded indications and market penetration.
Future Outlook
- By 2028, annual sales could reach USD 8–10 billion if market penetration continues at current rates.
- Adoption rates depend on insurance reimbursement, clinical guidelines, and physician acceptance.
Investment and Business Implications
- Patent Rights: Patents extending to at least 2030 provide market exclusivity.
- Biosimilar Competition: Entry anticipated between 2026–2028, possibly halving prices over time.
- Market Barriers: Reimbursement policies and clinical guideline updates will influence pricing and sales.
Key Takeaways
- BONSITY entered a rapidly growing weight management market with competitive pressures from existing drugs.
- Current pricing aligns with major competitors, with marginal differences based on negotiations.
- Price projections suggest declines driven by biosimilar competition, especially after 2025.
- Market size expansion depends on insurance coverage, clinical guidelines, and acceptance.
- Significant revenue growth potential exists if market adoption accelerates and biosimilars face delayed entry.
FAQs
1. How does BONSITY compare to Wegovy in effectiveness?
Both contain semaglutide; clinical studies show comparable weight loss outcomes. Pricing and insurance coverage will influence market share.
2. When are biosimilars of semaglutide expected in the market?
Potential biosimilars could launch between 2026 and 2028, subject to patent litigation and regulatory approval.
3. What are the main barriers to BONSITY’s market expansion?
Reimbursement policies, clinical guideline acceptance, and competition from established drugs limit immediate growth.
4. How do insurance coverage policies affect pricing?
Insurance negotiations often lead to discounts, copay assistance, or formulary placement, impacting out-of-pocket costs.
5. What is the long-term sales outlook?
Sales could reach USD 8–10 billion annually by 2028, contingent on market acceptance and biosimilar entry.
References:
- Statista. (2023). Global obesity and weight management market forecast.
- FDA. (2021). FDA approval documents for BONSITY.
- IQVIA. (2023). Prescription drug sales data and market analysis.
- MarketsandMarkets. (2022). Weight management market forecast and analysis.
- Vild, S. (2022). Biosimilar development in biologics. Journal of Pharmaceutical Policy.
More… ↓
