Share This Page
Drug Price Trends for BISOPROLOL FUMARATE
✉ Email this page to a colleague
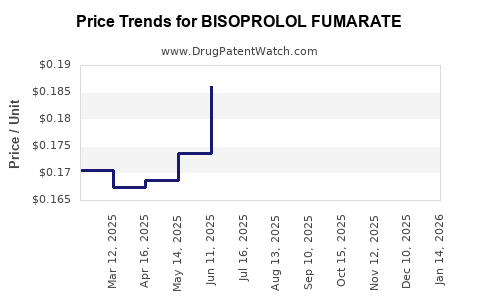
Average Pharmacy Cost for BISOPROLOL FUMARATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BISOPROLOL FUMARATE 10 MG TAB | 29300-0127-13 | 0.19902 | EACH | 2026-03-18 |
| BISOPROLOL FUMARATE 10 MG TAB | 43547-0617-03 | 0.19902 | EACH | 2026-03-18 |
| BISOPROLOL FUMARATE 10 MG TAB | 29300-0127-01 | 0.19902 | EACH | 2026-03-18 |
| BISOPROLOL FUMARATE 10 MG TAB | 43547-0617-10 | 0.19902 | EACH | 2026-03-18 |
| BISOPROLOL FUMARATE 10 MG TAB | 16714-0530-01 | 0.19902 | EACH | 2026-03-18 |
| BISOPROLOL FUMARATE 5 MG TAB | 72888-0092-30 | 0.15718 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BISOPROLOL FUMARATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BISOPROLOL FUMARATE 10MG TAB | Golden State Medical Supply, Inc. | 51407-0646-30 | 30 | 4.43 | 0.14767 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BISOPROLOL FUMARATE 10MG/HYDROCHLOROTHIAZIDE | Golden State Medical Supply, Inc. | 42799-0922-01 | 100 | 40.36 | 0.40360 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BISOPROLOL FUMARATE 2.5MG/HYDROCHLOROTHIAZIDE | Golden State Medical Supply, Inc. | 42799-0920-02 | 500 | 139.25 | 0.27850 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BISOPROLOL FUMARATE 10MG/HYDROCHLOROTHIAZIDE | Golden State Medical Supply, Inc. | 42799-0922-02 | 500 | 139.25 | 0.27850 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BISOPROLOL FUMARATE 2.5MG/HYDROCHLOROTHIAZIDE | Golden State Medical Supply, Inc. | 42799-0920-30 | 30 | 13.79 | 0.45967 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BISOPROLOL FUMARATE 10MG/HYDROCHLOROTHIAZIDE | Golden State Medical Supply, Inc. | 42799-0922-30 | 30 | 5.03 | 0.16767 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Bisoprolol Fumarate: Patent Landscape and Market Projections
Bisoprolol fumarate, a selective beta-1 adrenergic receptor blocker, faces a mature generics market. Key patents have expired, leading to intense price competition. Future market dynamics will be driven by biosimilar competition, evolving prescribing patterns, and potential niche indications.
What is the Patent Status of Bisoprolol Fumarate?
The patent landscape for bisoprolol fumarate is largely characterized by expired foundational patents. The original compound patents, which would have provided broad protection, expired decades ago. For instance, the initial patents for bisoprolol itself have long since lapsed in major markets like the United States and Europe.
- Compound Patents: Expired. Original patents covering the bisoprolol molecule were granted in the late 1970s and early 1980s.
- Formulation Patents: Some later-expiring patents may have existed for specific formulations (e.g., controlled-release versions), but these are also largely expired or nearing expiration in key regions. These patents typically offered a limited extension of market exclusivity.
- Method of Use Patents: Patents claiming new therapeutic uses for bisoprolol fumarate could have provided some exclusivity. However, the primary indications for bisoprolol fumarate (hypertension, angina, heart failure) are well-established and have been protected by earlier, now expired, patents.
- Manufacturing Process Patents: While novel manufacturing processes can be patented, they generally do not prevent competitors from marketing a generic version of the active pharmaceutical ingredient as long as the final product meets established quality and purity standards and does not infringe on other active patents.
- Pediatric Exclusivity: In the US, the FDA can grant an additional six months of market exclusivity for pediatric studies conducted per a written request. This is a time-limited exclusivity and does not represent a new patent. For bisoprolol fumarate, any such exclusivity related to its primary indications would have already been utilized and expired.
Given the age of the core compound and the expiration of most significant formulation and use patents, the market for bisoprolol fumarate is predominantly a generic one, with competition from multiple manufacturers.
What is the Current Market Size and Growth Trajectory for Bisoprolol Fumarate?
The global market for bisoprolol fumarate is substantial but characterized by a low-growth, mature trajectory. As a widely prescribed generic medication for cardiovascular conditions, its market size is significant in terms of volume but faces downward price pressure due to intense generic competition.
- Global Market Value: While precise current figures fluctuate, the global market for bisoprolol fumarate generics is estimated to be in the hundreds of millions of U.S. dollars annually. For example, market research reports from the early 2020s have placed the bisoprolol market value between $500 million and $800 million USD [1]. This figure encompasses all generic formulations across major geographical regions.
- Growth Rate: The market exhibits a low compound annual growth rate (CAGR), typically in the low single digits (0-3%). This sluggish growth is attributed to:
- Market Saturation: Bisoprolol fumarate is a well-established first-line or second-line treatment for its approved indications, meaning most of the addressable patient population already has access to it.
- Price Erosion: The presence of numerous generic manufacturers leads to significant price competition, which limits overall market value growth even as unit sales remain stable or increase slightly.
- Competition from Other Beta-Blockers and Alternative Therapies: While bisoprolol is a preferred agent, it competes with other beta-blockers (e.g., metoprolol, atenolol, carvedilol) and alternative drug classes (e.g., ACE inhibitors, ARBs, calcium channel blockers, diuretics) for cardiovascular indications. Prescribing patterns can shift based on efficacy, side effect profiles, cost, and physician preference.
- Volume vs. Value: The market is driven more by volume than by value growth. More units are sold, but the revenue per unit is low and continues to decline.
- Regional Variations: Market size and growth can vary by region. Developed markets (North America, Europe) are generally larger in value due to higher healthcare spending but may show flatter growth. Emerging markets may exhibit slightly higher growth rates as access to essential medicines improves and healthcare infrastructure expands.
The market is stabilized by its established efficacy and broad therapeutic use, ensuring consistent demand, but is constrained from significant expansion by its generic status and intense competition.
What are the Key Therapeutic Areas and Prescribing Trends for Bisoprolol Fumarate?
Bisoprolol fumarate is primarily indicated for the management of cardiovascular diseases, with specific focus on heart failure, hypertension, and angina pectoris. Prescribing trends reflect its efficacy and safety profile within these established areas.
Primary Therapeutic Indications:
- Heart Failure (HF): Bisoprolol fumarate is a cornerstone therapy for stable chronic heart failure, particularly when used in conjunction with ACE inhibitors or ARBs, and diuretics. Its use in HF management is supported by landmark clinical trials, such as the CIBIS-III study, which demonstrated that initiating bisoprolol alongside conventional therapy, or even as the first-line agent in certain patient populations, led to comparable or improved outcomes compared to placebo [2].
- Hypertension (High Blood Pressure): It is widely prescribed as a monotherapy or in combination with other antihypertensive agents to lower blood pressure. Its selective beta-1 blockade provides antihypertensive effects with a potentially lower incidence of bronchoconstriction compared to non-selective beta-blockers.
- Angina Pectoris (Chest Pain): Bisoprolol fumarate helps reduce the frequency of anginal attacks by decreasing myocardial oxygen demand through reduced heart rate and contractility.
Prescribing Trends:
- Established First-Line/Second-Line Therapy: In developed markets, bisoprolol fumarate is often a preferred beta-blocker and a standard component of treatment guidelines for HF and hypertension. It frequently serves as a second-line agent when initial therapies are insufficient or as a component of triple or quadruple therapy for resistant hypertension.
- Shift Towards Beta-Blockers in Heart Failure: Guidelines from organizations like the European Society of Cardiology (ESC) and the American Heart Association (AHA) consistently recommend beta-blockers (including bisoprolol) as essential therapy for patients with symptomatic heart failure with reduced ejection fraction (HFrEF) [3]. The emphasis is on initiating and titrating to target doses to achieve maximal clinical benefit.
- Combination Therapies: Prescriptions often involve combination products or co-administration with other cardiovascular drugs. This is particularly common in hypertension and advanced heart failure management, where multiple drug classes are required to achieve therapeutic goals.
- Physician Preference and Guideline Adherence: Prescribing patterns are heavily influenced by clinical practice guidelines, physician experience, and local formularies. Generic availability also plays a significant role, with cost-effectiveness being a key consideration.
- Post-Myocardial Infarction (MI) Treatment: Bisoprolol is also indicated for the treatment of post-MI patients with diminished left ventricular function. This contributes to its sustained demand in cardiology settings.
- Limited Off-Label Use: While some beta-blockers are used off-label for conditions like anxiety or migraine prophylaxis, bisoprolol fumarate's primary use remains firmly within its approved cardiovascular indications due to its specific selectivity profile.
The trend is towards continued, steady use of bisoprolol fumarate within its established indications, driven by clinical guidelines and its favorable profile in managing serious cardiovascular conditions.
What are the Competitive Landscape and Pricing Dynamics for Bisoprolol Fumarate?
The competitive landscape for bisoprolol fumarate is defined by the presence of numerous generic manufacturers, leading to significant price erosion and commoditization of the product.
Competitive Landscape:
- Multiple Generic Manufacturers: The market is populated by a large number of pharmaceutical companies manufacturing and marketing generic bisoprolol fumarate. These include global players as well as regional and smaller specialized generic producers. Examples of companies involved in the generic bisoprolol market include Teva Pharmaceutical Industries, Sandoz (a Novartis division), Mylan (now Viatris), Aurobindo Pharma, and numerous others across North America, Europe, India, and China [4].
- Authorized Generic Products: Brand-name manufacturers sometimes introduce authorized generics (AGs) after patent expiry to compete with independent generic players, often with favorable supply agreements.
- Market Entry Barriers: The primary barriers to entry for new manufacturers are regulatory hurdles (obtaining ANDA approval in the US, MAA in Europe) and establishing cost-efficient manufacturing processes. However, for a well-established molecule like bisoprolol fumarate, these are often surmountable for companies with experience in generic drug production.
- Product Differentiation (Limited): Differentiation is minimal. It primarily revolves around:
- Price: The most significant differentiator.
- Supply Chain Reliability: Consistent availability is crucial.
- Dosage Forms/Packaging: While standard tablets are common, some variations in excipients or packaging might exist, but these do not offer clinical differentiation.
- Specific Formulations: Earlier, some extended-release (ER) or controlled-release (CR) formulations might have offered a period of differentiation, but these patents have largely expired, leading to generic versions of these as well.
Pricing Dynamics:
- Intense Price Competition: The core dynamic is fierce price competition. When the first generics enter the market, prices drop sharply. Subsequent generic entrants further accelerate this price erosion.
- Commoditization: Bisoprolol fumarate has become a commoditized pharmaceutical product. The active pharmaceutical ingredient (API) is widely available, and manufacturing processes are well-established and optimized for cost-efficiency.
- Wholesale Acquisition Cost (WAC) vs. Net Price: WAC prices are often listed, but actual transaction prices are significantly lower due to rebates, discounts, and contract pricing with wholesalers and pharmacy benefit managers (PBMs).
- Tender Systems and Group Purchasing Organizations (GPOs): In many healthcare systems (e.g., hospitals, national health services), tender systems and GPOs drive prices down through bulk purchasing and competitive bidding.
- Declining Average Selling Price (ASP): The ASP for generic bisoprolol fumarate has been on a continuous downward trend for years and is expected to remain so. The average price per tablet can be fractions of a cent in highly competitive tenders or large-volume contracts.
- Impact of New Entrants: The entry of new generic manufacturers, particularly those from lower-cost manufacturing regions like India and China, can further intensify price competition and drive down global pricing benchmarks.
- Price Stability (Relative): While prices continue to decline, the rate of decline may slow down once a certain minimum price point is reached, below which it becomes uneconomical for manufacturers to produce. However, this "floor" is very low for established generics.
The pricing of bisoprolol fumarate is primarily dictated by supply and demand in the generic market, where oversupply and a large number of competitors lead to sustained low prices and minimal profit margins for individual manufacturers.
What are the Future Market Projections and Potential Opportunities for Bisoprolol Fumarate?
The future market for bisoprolol fumarate is expected to remain stable with moderate volume growth offset by continued price erosion. Opportunities lie in operational efficiency, supply chain resilience, and potential minor niche developments.
Future Market Projections:
- Continued Generic Domination: The market will continue to be dominated by generic bisoprolol fumarate. No significant new branded products are anticipated that could disrupt this dynamic, given the drug's established efficacy and generic availability.
- Stable to Modest Volume Growth: Overall unit volume is projected to grow modestly, driven by an aging global population, increasing prevalence of cardiovascular diseases, and expanding access to healthcare in emerging markets. The CAGR for volume is likely to be in the 1-3% range globally.
- Persistent Price Erosion: Average selling prices will continue to decline, albeit at a potentially slower rate than in the initial post-patent expiry phase. This will result in low single-digit CAGR for market value, or even flat to slightly negative growth in some mature markets.
- Generic Competition Intensification: The ongoing emergence of new generic manufacturers, especially from cost-competitive regions, will sustain price pressure.
- Biosimilar Considerations (Not Applicable): Bisoprolol fumarate is a small molecule drug and is not subject to biosimilar competition, which is relevant only for biologic drugs.
Potential Opportunities:
- Operational Excellence and Cost Optimization: Manufacturers can achieve profitability through extreme efficiency in API sourcing, manufacturing, and distribution. Streamlining supply chains and achieving economies of scale are paramount.
- Supply Chain Resilience and Reliability: Companies that can guarantee consistent supply and robust quality control will maintain market share, especially in large institutional contracts or government tenders where supply disruption is unacceptable.
- Emerging Markets: While pricing is lower, expanding healthcare access and infrastructure in emerging economies in Asia, Africa, and Latin America present opportunities for volume growth. Establishing local manufacturing or strategic partnerships in these regions can be advantageous.
- Combination Products (Limited): While many combination products exist, there might be opportunities for fixed-dose combinations (FDCs) with newer antihypertensive or cardiovascular agents if they meet specific unmet clinical needs or simplify patient regimens and adhere to evolving guidelines. However, developing and gaining approval for novel FDCs is costly and time-consuming.
- Formulation Improvements (Niche): While major formulation patents have expired, there might be very niche opportunities for improved drug delivery systems (e.g., enhanced solubility, alternative release profiles for specific patient groups) if they offer a demonstrable clinical advantage and can secure regulatory approval. These would likely represent very small market segments compared to standard immediate-release tablets.
- Pediatric Formulations (Niche): Development of palatable or easier-to-administer pediatric formulations could create a small, specialized market segment, although the primary indications for bisoprolol are less common in very young children compared to adults.
The future of bisoprolol fumarate lies in efficient, high-volume production of a well-established generic. Significant market expansion is unlikely, making operational efficiency and supply chain reliability the primary drivers of success for manufacturers.
Key Takeaways
- Bisoprolol fumarate's patent protection has expired, resulting in a highly competitive generic market.
- The global market is valued in the hundreds of millions of U.S. dollars, with low single-digit growth.
- Primary indications include heart failure, hypertension, and angina pectoris, with stable prescribing patterns guided by clinical practice.
- Intense price competition among numerous generic manufacturers has led to significant price erosion and commoditization.
- Future market growth will be modest, driven by volume in emerging markets, while price declines will continue.
- Opportunities lie in operational efficiency, supply chain reliability, and navigating emerging markets, rather than novel product development.
Frequently Asked Questions
-
Are there any remaining patents that could extend market exclusivity for bisoprolol fumarate? No significant patents covering the basic compound, primary formulations, or core therapeutic uses remain that would grant market exclusivity in major markets. Any minor patents that might still exist typically cover very specific, less common formulations or manufacturing processes that do not prevent generic competition.
-
What is the typical price range for generic bisoprolol fumarate tablets? The price can vary significantly based on volume, contract terms, and region, but generic bisoprolol fumarate tablets are among the lowest-cost prescription medications. In large institutional tenders or contracts, the price per tablet can be as low as a few U.S. cents or even fractions of a cent.
-
Are there any new therapeutic indications for bisoprolol fumarate being investigated? While bisoprolol fumarate is a well-researched drug, significant new therapeutic indications are unlikely to emerge. Its role is firmly established in cardiovascular diseases, and exploration for novel uses is rare for such mature generic compounds.
-
What is the impact of competition from other beta-blockers on the bisoprolol fumarate market? Bisoprolol fumarate competes with a range of other beta-blockers (e.g., metoprolol, atenolol, carvedilol). Prescribing decisions are influenced by guidelines, physician preference, patient response, and cost. While it holds a strong position, particularly in heart failure, competition within the beta-blocker class contributes to the overall pricing pressure.
-
How does the supply chain for bisoprolol fumarate typically operate? The supply chain generally involves API manufacturers (often in India and China), generic drug formulators, distributors, wholesalers, and finally, pharmacies and healthcare institutions. The generic nature of the drug means that multiple suppliers for each component of the supply chain exist, contributing to competitive pricing.
Citations
[1] Grand View Research. (2023). Bisoprolol Market Size, Share & Trends Analysis Report By Drug Type (Generic, Branded), By Indication (Hypertension, Heart Failure, Angina Pectoris, Others), By Dosage Form (Tablets, Others), By Region, And Segment Forecasts, 2023 - 2030. [2] CIBIS-III Investigators. (2003). The Cardiac Insufficiency Bisoprolol Study III (CIBIS-III): a randomized trial to compare the effects of starting bisoprolol immediately or after four weeks on morbidity and mortality in patients with chronic heart failure. European Heart Journal, 24(20), 1880-1889. [3] Heidenreich, P. A., Bozkurt, B., Aguilar, D., Brar, S., Cleveland, J. C., Jr, Douglas, P. S., ... & Committee on Guidelines for Cardiovascular ..., & American Heart Association Clinical ... (2022). 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation, 145(18), e895-e1032. [4] Various pharmaceutical industry reports and company product listings (e.g., FDA Orange Book, EMA product databases, company annual reports and product catalogs) from 2020-2023 indicate the widespread presence of generic bisoprolol fumarate from numerous manufacturers.
More… ↓
