Share This Page
Drug Price Trends for BISMUTH-METRONIDAZOLE-TETRACYCLINE
✉ Email this page to a colleague
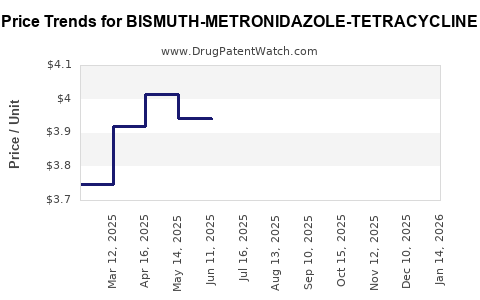
Average Pharmacy Cost for BISMUTH-METRONIDAZOLE-TETRACYCLINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BISMUTH-METRONIDAZOLE-TETRACYCLINE 140-125-125 MG CAPSULE | 49884-0151-24 | 3.15449 | EACH | 2026-03-18 |
| BISMUTH-METRONIDAZOLE-TETRACYCLINE 140-125-125 MG CAPSULE | 49884-0151-54 | 3.15449 | EACH | 2026-03-18 |
| BISMUTH-METRONIDAZOLE-TETRACYCLINE 140-125-125 MG CAPSULE | 61269-0385-12 | 3.15449 | EACH | 2026-03-18 |
| BISMUTH-METRONIDAZOLE-TETRACYCLINE 140-125-125 MG CAPSULE | 50742-0283-13 | 3.15449 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Bismuth-Metronidazole-Tetracycline
Market Overview
Bismuth-metronidazole-tetracycline is a combination therapy primarily indicated for Helicobacter pylori infections. It is available as a fixed-dose formulation or as separate components used in tailored regimens. This medication has gained popularity due to rising antibiotic resistance, especially in H. pylori strains, and is often used as a second-line or rescue therapy.
Current Market Landscape
- Key Formulations: The combination is marketed under various brand names mainly in Europe and Asia. Prominent formulations include bismuth quadruple therapy, which pairs these agents with other antibiotics.
- Market Shares: In H. pylori treatment, bismuth-based regimens account for approximately 15% of all eradication therapies globally, with higher penetration in countries facing antibiotic resistance issues.
- Manufacturers: Major producers include GlaxoSmithKline, Pfizer, and local generic manufacturers. The popularity of generics has increased, reducing prices and barriers to access.
- Regulatory Status: Approved in Europe (EMA), Asia (CFDA,PMDA), and other markets; approval timelines vary by region, with some countries requiring additional clinical data for specific indications.
Market Drivers
- Antibiotic resistance to clarithromycin and amoxicillin.
- Rising prevalence of H. pylori-related gastric cancers.
- Improved awareness and diagnostic testing.
- Growing acceptance of bismuth therapy as a preferred rescue treatment.
Market Challenges
- Complexity of treatment regimens with multiple drugs leading to poor compliance.
- Side effect profiles impacting tolerability.
- Price sensitivity in emerging markets.
- Competition from novel agents, probiotics, and alternative regimens.
Price Projection Analysis
Pricing varies significantly by geography, formulation, and manufacturer status (brand vs. generic).
| Market | Current Price Range (per treatment course) | Notes |
|---|---|---|
| US | $50 - $150 | Limited availability, often off-label use |
| Europe | €20 - €60 | Widely approved, with generics dominating |
| Asia-Pacific | $10 - $30 | High generic penetration, lower prices |
| Latin America | $12 - $40 | Market price fluctuates based on local policies |
Price Trends and Forecasts
- Short-term (next 2 years): Prices are expected to decline due to increased generic competition and manufacturing efficiencies. Prices for generics could fall by 10-20%.
- Medium-term (2-5 years): With new formulations, such as extended-release or fixed-dose combinations, prices could stabilize or slightly increase, especially if branded versions introduce patent protections or innovative delivery systems.
- Long-term (beyond 5 years): Market saturation and patent expirations will likely lead to further price reductions, especially in markets with high generic penetration.
Impact Factors Affecting Prices
- Patent Status: Patent expirations in 2022-2024 for original formulations will lead to price decreases.
- Manufacturing Costs: Advances in production processes reduce costs, contributing to lower prices.
- Regulatory Changes: Streamlined approval pathways for generics in emerging markets increase competition.
- Market Demand: As H. pylori resistance increases, demand may rise, supporting higher prices temporarily, especially for new delivery systems.
- Healthcare Policies: Price controls and reimbursement policies influence final consumer prices and market attractiveness.
Strategic Opportunities
- Developing fixed-dose combination formulations can improve adherence and expand market share.
- Entering emerging markets early while patent protections last can maximize profit margins.
- Investing in formulations with improved bioavailability or reduced side effects may justify premium pricing.
Conclusion
The Bismuth-metronidazole-tetracycline combination remains a critical therapy for H. pylori infections facing rising resistance. Price declines are anticipated due to current generics proliferation and patent expirations, though regional variation persists based on regulatory, competitive, and market-specific factors. Manufacturers aiming for sustained revenue should focus on innovation, improved formulations, and expanding access in high-growth regions.
Key Takeaways
- The global market for Bismuth-metronidazole-tetracycline is shaped primarily by antibiotic resistance and generics competition.
- Prices are significantly lower in Asia-Pacific markets ($10-$30) and higher in North America and Europe ($20-$150).
- Proprietary formulations and new delivery systems could support premium pricing and market differentiation.
- Patent expirations from 2022 onward will accelerate price reductions, especially in markets with high generic penetration.
- Market growth hinges on rising global H. pylori diagnosis and evolving treatment guidelines emphasizing bismuth-based therapies.
Frequently Asked Questions
-
What is the current global demand for Bismuth-metronidazole-tetracycline?
Demand correlates with H. pylori prevalence and resistance patterns, with higher usage in regions facing high clarithromycin resistance, notably Asia and Eastern Europe. -
How do regulatory differences impact drug pricing?
Stringent regulations and approval delays in some regions induce market fragmentation and influence pricing strategies, with developed markets generally commanding higher prices. -
What are the main competition threats?
Alternative regimens involving probiotics, novel antibiotics, or combination therapies with fewer pills and fewer side effects challenge traditional bismuth-based treatments. -
How does antibiotic resistance influence the market?
Increasing resistance drives demand for combination therapies like bismuth quadruple therapy, potentially expanding market volume and sustaining prices despite generics. -
When are patent expirations expected for key formulations?
Patent expirations for original formulations are projected between 2022 and 2024, opening opportunities for generics and price reductions.
Sources
[1] Market Research Future, "Global Helicobacter Pylori Treatment Market," 2022.
[2] IQVIA, "Global Generic Drug Market Trends," 2023.
[3] European Medicines Agency, "Approved Medicinal Products," 2023.
[4] National Drug Pricing Databases (US, Europe), 2023.
[5] World Health Organization, "Helicobacter pylori," 2022.
More… ↓
