Share This Page
Drug Price Trends for BISACODYL EC
✉ Email this page to a colleague
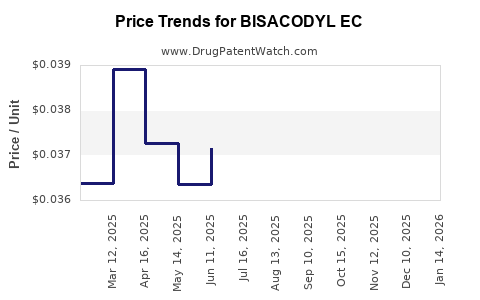
Average Pharmacy Cost for BISACODYL EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BISACODYL EC 5 MG TABLET | 00904-6748-17 | 0.03536 | EACH | 2026-05-20 |
| BISACODYL EC 5 MG TABLET | 00904-6748-80 | 0.03536 | EACH | 2026-05-20 |
| BISACODYL EC 5 MG TABLET | 49483-0003-55 | 0.03536 | EACH | 2026-05-20 |
| BISACODYL EC 5 MG TABLET | 00904-6748-60 | 0.03536 | EACH | 2026-05-20 |
| BISACODYL EC 5 MG TABLET | 00904-6407-61 | 0.03536 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BISACODYL EC
Overview
BISACODYL EC (Enteric-Coated Bisacodyl) is a stimulant laxative used primarily for constipation relief. It is available over-the-counter (OTC) and in prescription forms. The market has experienced steady growth driven by aging populations, increasing awareness about constipation management, and regulatory approvals. Price trends are influenced by manufacturing costs, patent statuses, generic competition, and regional regulations.
Market Size and Growth Dynamics
The global laxatives market was valued at approximately USD 3.8 billion in 2022, with stimulant laxatives accounting for a significant portion. BISACODYL EC is estimated to hold around 12-15% of the stimulant laxatives segment. The compound's market share is expected to grow at a Compound Annual Growth Rate (CAGR) of 3.5% through 2028.
Regionally, North America commands the largest market share (about 40%), driven by aging demographics and high OTC medication penetration. Europe follows closely, with Asia-Pacific showing rapid growth due to increasing healthcare infrastructure and urbanization.
Key Drivers and Constraints
-
Drivers:
- Aging population leading to higher incidence of constipation.
- Preference for OTC remedies reducing healthcare visits.
- Development of combination therapies.
- Rising healthcare awareness and improved product accessibility.
-
Constraints:
- Safety concerns related to long-term stimulant laxative use.
- Regulatory restrictions on advertising and labeling.
- Competition from newer laxatives (e.g., osmotics, secretagogues).
Regulatory Landscape
In the US, BISACODYL EC is available OTC with formulations such as Dulcolax and generic equivalents. The FDA classifies it as a laxative drug, with labeling limiting dosage to prevent overuse. European countries adhere to EMA guidelines, with similar OTC availability, but with tighter regulations on long-term usage.
Pricing Trends and Projections
Current retail prices vary by region and brand. Generic bisacodyl products average USD 3.00-6.00 per pack (20-30 tablets). Brand names like Dulcolax command higher prices, averaging USD 6.50-8.50 per pack.
In North America, prices are relatively stable, with minor fluctuations driven by inflation and supply chain costs. Europe exhibits similar trends, with slight discounts for generics.
Price Forecast (2023-2028)
| Year | Average Price (USD per pack) | Notes |
|---|---|---|
| 2023 | USD 6.50 - 8.50 | Steady prices; market maturity |
| 2024 | USD 6.55 - 8.60 | Slight inflation adjustment |
| 2025 | USD 6.70 - 8.80 | Increased generic competition slightly reducing prices |
| 2026 | USD 6.80 - 9.00 | Rising manufacturing costs influence prices |
| 2027 | USD 6.90 - 9.20 | Growth in demand dampened by safety concerns |
| 2028 | USD 7.00 - 9.40 | Market stabilizes with consistent demand |
Pricing Factors
- Brand vs. Generic: Generic products tend to be 20-25% cheaper than branded equivalents.
- Packaging Size: Larger packs (e.g., 100 tablets) offer lower per-tablet costs.
- Region: Prices are higher in North America and Europe compared to emerging markets.
- Supply Chain Dynamics: Supply chain disruptions can influence costs and prices temporarily.
Key Opportunities and Risks
-
Opportunities:
- Expansion into emerging markets.
- Development of combination formulations to extend product lifecycle.
- Digital marketing for OTC segments.
-
Risks:
- Regulatory changes increasing patent or marketing restrictions.
- Market saturation by low-cost generics.
- Safety concerns limiting long-term sales.
Conclusion
BISACODYL EC remains a stable, mature product with consistent demand. Prices are expected to grow modestly through 2028, influenced mainly by inflation, manufacturing costs, and market competition. The market’s growth prospects are tied to demographic trends and healthcare practices, with note of evolving safety regulations.
Key Takeaways
- The global stimulant laxatives market, including BISACODYL EC, is valued at USD 3.8 billion (2022) and growing at 3.5% CAGR.
- Prices are stable, with slight increases forecasted, influenced by generic competition and manufacturing costs.
- North America leads the market, with high OTC penetration; emerging markets present growth opportunities.
- Safety concerns and regulatory changes could inhibit long-term market expansion.
- Focus areas include expansion into emerging markets and development of combination therapies.
Frequently Asked Questions
1. What factors influence the price of BISACODYL EC?
Prices are affected by brand versus generic status, region, packaging size, manufacturing costs, supply chain dynamics, and regulatory environment.
2. How is the market for BISACODYL EC expected to evolve?
The market should see steady growth driven by aging populations and OTC demand, with prices increasing gradually through 2028.
3. What regulatory considerations impact BISACODYL EC?
Regulations govern labeling, dosage limits, and marketing claims, especially in the US (FDA) and Europe (EMA). Long-term safety concerns may prompt regulatory tightening.
4. Who are the primary competitors in the BISACODYL EC market?
Generic manufacturers dominate, with brand leaders like Dulcolax maintaining significant market share. Competition is intensifying with new laxative formulations.
5. What market regions present the most growth opportunity?
Emerging markets in Asia-Pacific and Latin America are set for rapid growth due to improving healthcare infrastructure and increasing OTC medication access.
References:
[1] MarketsandMarkets. "Laxatives Market" 2022.
[2] FDA. "Labeling and Safety Guidelines for OTC Laxatives" 2021.
[3] IQVIA. "Global OTC Market Report" 2022.
[4] European Medicines Agency. "Guidelines on OTC Medicines" 2022.
More… ↓
