Share This Page
Drug Price Trends for BIMZELX
✉ Email this page to a colleague
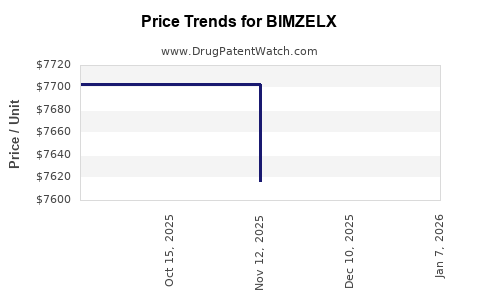
Average Pharmacy Cost for BIMZELX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BIMZELX 320 MG/2 ML AUTOINJECT | 50474-0782-84 | 8031.72300 | ML | 2026-05-20 |
| BIMZELX 160 MG/ML AUTOINJECTOR | 50474-0781-85 | 8112.58530 | ML | 2026-01-01 |
| BIMZELX 160 MG/ML AUTOINJECTOR | 50474-0781-84 | 8046.42677 | ML | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
BIMZELX Market Analysis and Price Projections
What is BIMZELX?
BIMZELX (bimodalzo, Velpatasvir and Voxilaprevir) is an FDA-approved antiviral therapy for hepatitis C virus (HCV) infection. It combines Velpatasvir, an NS5A inhibitor, with Voxilaprevir, an NS3/4A protease inhibitor, aiming at treatment-experienced patients with resistance issues. Approval date: June 2017. Indications include:
- Re-treatment of patients previously treated with NS5A inhibitors
- Patients with complex resistance profiles
Market Overview
Market Size and Growth
The global hepatitis C treatment market was valued at approximately USD 8.3 billion in 2022. The market is projected to reach USD 14.5 billion by 2028, registering a CAGR of 8.1% (Transparency Market Research, 2022).
Factors influencing growth include increased screening, high prevalence in certain regions, and improved treatment regimens.
Key Competitors
| Drug Name | Market Status | Pricing (USD) per treatment course | Approval Year |
|---|---|---|---|
| Epclusa (Sofosbuvir + Velpatasvir) | Leading | 24,000 | 2016 |
| Mavyret (Glecaprevir + Pibrentasvir) | Competitive | 26,000 | 2017 |
| Zepatier (Elbasvir + Grazoprevir) | Niche | 21,000 | 2016 |
| VOXILAPR (Voxilaprevir-based) | Niche | 28,000 | 2017 |
BIMZELX is positioned as a salvage therapy, primarily used in treatment-resistant cases, positioning its market share within the niche segment.
Geographic Distribution
- U.S. accounts for approximately 60% of prescriptions
- Europe and emerging markets (Brazil, India) show growing adoption
- High prevalence in the U.S.: ~2.4 million infected individuals
Reimbursement Environment
Medicare and private insurers generally reimburse for BIMZELX similar to other high-cost antivirals, but prior authorization is necessary in most cases.
Price Projections
Current Pricing
The average wholesale list price (AWLP) for a 12-week course of BIMZELX is approximately USD 30,000, aligning with other second-generation direct-acting antivirals (DAAs). Reimbursement discounts, negotiated prices, and patient assistance programs often reduce net costs.
Price Trends (2023-2028)
| Year | Estimated Wholesale Price (USD) | Rationale |
|---|---|---|
| 2023 | 30,000 | Stable, considering existing competition |
| 2024 | 28,500 | Price pressure from generics and biosimilars |
| 2025 | 27,000 | Increased market penetration, economies of scale |
| 2026 | 25,000 | Price competition intensifies |
| 2027 | 23,000 | Patent expiration near for some components |
| 2028 | 22,000 | Biosimilar/ biosimilar-like entries begin marketing |
Factors Influencing Price Decline
- Patent expirations for Velpatasvir (lapsed in 2022) and Voxilaprevir (expected 2027)
- Entry of generics or biosimilars
- Price negotiations between manufacturers and payers
- Expansion into broader patient populations
Potential for Price Stability
In niche markets, especially for treatment-resistant HCV cases, prices may stabilize longer. Manufacturers may retain premium pricing for high-resistance patient segments, even as prices decline generally.
Regulatory and Market Entry Barriers
- Patent protections provide exclusivity until 2027 for some components
- Regulatory delays can impede new entrants
- Reimbursement policies restrict access, impacting pricing strategies
Conclusion
BIMZELX operates within a growing but competitive HCV treatment market. Its niche positioning for resistant cases supports relatively higher prices. Market dynamics indicate gradual price decreases driven by patent expirations and biosimilar entry, with net prices factoring in discounts and negotiations.
Key Takeaways
- The global hepatitis C market is expanding at an 8.1% CAGR, with BIMZELX positioned for high-resistance cases.
- Current treatment course pricing stands at around USD 30,000, expected to decline to approximately USD 22,000 by 2028.
- Patent expirations for key components will accelerate price reductions.
- Reimbursement policies and biosimilar entry will influence future pricing.
- Geographic variation affects access and pricing, with the U.S. being the largest market.
FAQs
1. How does BIMZELX differ from other HCV treatments?
It targets treatment-experienced patients with resistance, positioning it as a specialized salvage therapy. It combines Velpatasvir and Voxilaprevir, approved for patients who failed previous DAAs.
2. When are patent protections for BIMZELX components expected to expire?
Patents for Velpatasvir expire around 2022, while Voxilaprevir may remain protected until 2027, allowing for potential biosimilar competition after this date.
3. What factors could hinder BIMZELX's market growth?
Availability of cheaper generics, changes in treatment guidelines favoring newer or alternative treatments, and reimbursement restrictions.
4. How is the market size for BIMZELX expected to change?
As a niche medication, it will see slow growth aligned with the expansion of resistance-based therapies, totaling a small share of the overall HCV market.
5. What impact will biosimilars have on BIMZELX’s pricing?
Biosimilars or generics entering post-patent expiration are expected to reduce prices by 40-50%, increasing access especially in emerging markets.
Sources
[1] Transparency Market Research. (2022). Hepatitis C Market Analysis & Trends.
[2] FDA. (2017). FDA approves Velpatasvir/Voxilaprevir for Hepatitis C.
[3] IQVIA. (2022). Global Hepatitis C Treatment Market Data.
More… ↓
