Share This Page
Drug Price Trends for BIKTARVY
✉ Email this page to a colleague
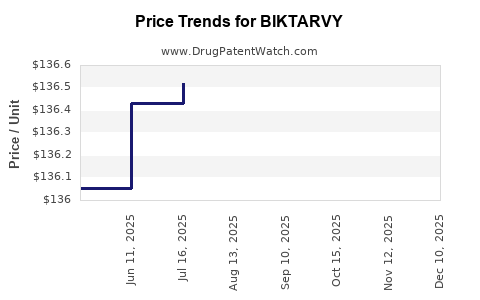
Average Pharmacy Cost for BIKTARVY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BIKTARVY 50-200-25 MG TABLET | 61958-2501-01 | 136.74030 | EACH | 2026-03-18 |
| BIKTARVY 30-120-15 MG TABLET | 61958-2505-01 | 136.73160 | EACH | 2026-03-18 |
| BIKTARVY 30-120-15 MG TABLET | 61958-2506-01 | 136.73160 | EACH | 2026-03-18 |
| BIKTARVY 50-200-25 MG TABLET | 61958-2501-03 | 136.74030 | EACH | 2026-03-18 |
| BIKTARVY 30-120-15 MG TABLET | 61958-2506-01 | 136.69106 | EACH | 2026-03-04 |
| BIKTARVY 30-120-15 MG TABLET | 61958-2505-01 | 136.69106 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Biktarvy: Market Trajectory and Pricing Outlook
Executive Summary
Biktarvy, a combination antiretroviral therapy, is projected to maintain a dominant market position in HIV treatment due to its efficacy, tolerability, and once-daily single-tablet regimen. The drug's patent exclusivity and the growing global HIV treatment market are key drivers of its sustained revenue generation. While generic competition is anticipated post-patent expiry, a significant market share is expected to persist due to established patient adherence and physician preference. Pricing is anticipated to remain stable in developed markets, with potential for tiered pricing strategies in emerging economies.
Global HIV Treatment Market Overview
The global HIV treatment market is characterized by increasing patient access to antiretroviral therapy (ART), driven by public health initiatives and expanding healthcare infrastructure. The World Health Organization (WHO) recommends ART for all individuals with HIV, irrespective of CD4 count, leading to a growing patient pool [1].
- Market Size: The global HIV market was valued at approximately USD 27.8 billion in 2022 and is projected to reach USD 39.7 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030 [2].
- Key Drivers:
- Increased HIV diagnosis rates.
- Expanded access to ART in low- and middle-income countries.
- Introduction of novel treatment regimens with improved tolerability and convenience.
- Ongoing research and development of new HIV prevention and treatment strategies.
- Challenges:
- Drug resistance development.
- High cost of treatment in certain regions.
- Stigma associated with HIV.
- Adherence challenges among patients.
Biktarvy: Product Profile and Market Standing
Biktarvy (bictegravir/emtricitabine/tenofovir alafenamide) is a complete, single-tablet regimen developed by Gilead Sciences for the treatment of HIV-1 infection. It combines a potent integrase strand transfer inhibitor (INSTI) with two nucleoside reverse transcriptase inhibitors (NRTIs) [3].
- Mechanism of Action:
- Bictegravir: Inhibits the integration of viral DNA into the host cell genome.
- Emtricitabine and Tenofovir Alafenamide: Block the activity of reverse transcriptase, an enzyme essential for HIV replication.
- Efficacy and Safety: Clinical trials have demonstrated Biktarvy's high efficacy in achieving and maintaining viral suppression, with a favorable safety and tolerability profile compared to older ART regimens [4]. It exhibits low rates of treatment-emergent resistance.
- Market Positioning: Biktarvy is a leading first-line HIV treatment option. Its single-tablet formulation offers significant convenience for patients, contributing to high adherence rates.
- Competitive Landscape: Biktarvy competes with other INSTI-based regimens, as well as other classes of ART. Key competitors include Dovato (dolutegravir/lamivudine), Triumeq (abacavir/dolutegravir/lamivudine), and Juluca (dolutegravir/rilpivirine) [5].
Patent Exclusivity and Generic Threat
Gilead Sciences holds key patents protecting Biktarvy. The patent landscape is critical for understanding future market dynamics and pricing.
- Key Patents:
- U.S. Patent No. 9,133,201 (expiring 2034) for the bictegravir component.
- U.S. Patent No. 10,655,005 (expiring 2036) related to the fixed-dose combination [6].
- Additional patents cover manufacturing processes and specific formulations.
- Patent Expiry Impact: Post-patent expiry, generic versions of Biktarvy are expected to enter the market. This typically leads to significant price erosion.
- Market Share Retention: Despite generic entry, Biktarvy is anticipated to retain a substantial market share due to:
- Physician Inertia and Preference: Prescribing habits are often slow to change, especially for highly effective and well-tolerated drugs.
- Patient Adherence: Patients accustomed to Biktarvy may continue to request it, and healthcare providers may prioritize established treatment regimens.
- Formulation Advantages: While generics may match the active ingredients, proprietary manufacturing processes or specific formulation characteristics could offer perceived or actual advantages.
- Exclusivity Periods: Pediatric exclusivity and other regulatory extensions may delay generic entry.
- Projected Generic Entry: While specific dates depend on legal challenges and regulatory approvals, significant generic competition in major markets like the US and Europe is generally anticipated around 2034-2036.
Market Share Projections
Biktarvy's market share is expected to remain strong in the near to medium term, driven by its clinical profile and market penetration.
- Current Market Share: Biktarvy holds a significant share of the first-line HIV treatment market, often exceeding 50% in developed countries.
- Near-Term Outlook (2024-2028): Continued market leadership with stable or slightly increasing market share. Growth will be supported by new patient initiations and switching from older regimens.
- Medium-Term Outlook (2029-2034): Market share will remain high, but growth may plateau as the market matures and potential for earlier generic challenges in some regions emerges.
- Post-Patent Expiry (2035 onwards): A projected decline in market share as generic alternatives become available. However, Biktarvy itself is expected to retain a significant portion of its user base for several years post-generic entry.
- Estimated market share post-patent expiry: 30-40% within the INSTI class, depending on generic pricing and market dynamics.
Pricing Analysis and Projections
Biktarvy's pricing is influenced by factors including R&D investment, manufacturing costs, market demand, competitive landscape, and payer negotiations.
- Current Pricing: In the United States, the wholesale acquisition cost (WAC) for Biktarvy (30 tablets) is approximately USD 2,400 to USD 2,600 per month, translating to an annual cost of roughly USD 28,800 to USD 31,200. Prices in Europe and other developed markets are generally lower due to price controls and negotiation frameworks [7].
- Pricing Stability in Developed Markets:
- Short to Medium Term (2024-2030): Minimal price reductions are anticipated in key markets like the US, Canada, and Western Europe. This stability is supported by its strong clinical value proposition and patent protection. Payer rebates and discounts exist but do not significantly alter the list price trajectory.
- Impact of Competition: While other INSTI-based regimens exist, Biktarvy's market dominance provides pricing power.
- Emerging Markets Pricing Strategy:
- Gilead employs tiered pricing strategies for its HIV medications in low- and middle-income countries.
- Voluntary Licensing: Gilead has entered into voluntary licensing agreements with generic manufacturers in certain regions, enabling the production of lower-cost versions of its HIV medicines, including components of Biktarvy [8]. This strategy expands access while protecting market exclusivity in higher-income countries.
- Projected Pricing in Emerging Markets: Prices in these regions will be significantly lower than in developed markets, ranging from USD 75 to USD 150 per patient per year for Biktarvy (or its components under license) [8].
- Post-Patent Expiry Pricing:
- Significant Price Erosion: Following the expiry of primary patents, the price of Biktarvy in developed markets is expected to decline sharply, potentially by 50-80% within the first few years of generic entry, mirroring trends observed with other blockbuster HIV drugs.
- Generic Pricing: Generic manufacturers will likely compete aggressively on price. The average selling price will depend on the number of generic entrants and their cost structures.
- Continued Demand: Even with price reductions, the volume of Biktarvy (branded and generic) prescribed is expected to remain high due to its established efficacy and patient preference.
Future Market Outlook
The future of the HIV treatment market is influenced by ongoing R&D, evolving treatment paradigms, and global health policies.
- Long-Acting Injectables: The development of long-acting injectable ART, such as cabotegravir/rilpivirine (Cabenuva), represents a significant shift. These injectables offer an alternative to daily oral regimens and could capture a portion of the market, particularly for patients seeking to reduce pill burden [9]. However, Biktarvy's daily oral convenience remains a strong advantage for many.
- HIV Prevention (PrEP): The expansion of pre-exposure prophylaxis (PrEP) using drugs like tenofovir disoproxil fumarate/emtricitabine (Truvada) and disoproxil fumarate/emtricitabine/bictegravir (Descovy) will continue to grow the overall HIV market, though it does not directly impact Biktarvy's treatment market share.
- Cure Research: While significant progress is being made in HIV cure research, a widely available functional cure is not expected within the next decade, ensuring continued demand for effective ART like Biktarvy.
- Global Access Initiatives: Continued global efforts to increase HIV testing and treatment access, particularly in sub-Saharan Africa and Asia, will drive overall market growth.
Key Takeaways
- Biktarvy is projected to maintain its market leadership in HIV treatment through at least 2034 due to its efficacy, tolerability, and single-tablet regimen.
- Patent expiry around 2034-2036 will lead to significant price erosion and the introduction of generic versions.
- Biktarvy is expected to retain a substantial market share post-patent expiry due to established physician and patient preference.
- Pricing in developed markets is forecast to remain stable in the near to medium term, with tiered pricing and voluntary licensing models employed in emerging economies.
- The emergence of long-acting injectables represents a competitive factor, but the convenience of Biktarvy's oral regimen is expected to preserve its market position for the foreseeable future.
Frequently Asked Questions
- What is the current average annual cost of Biktarvy for a patient in the United States?
- When are the primary patents for Biktarvy expected to expire in major markets like the US and Europe?
- How does Gilead's voluntary licensing program impact the availability and pricing of Biktarvy in lower-income countries?
- What is the projected market share for Biktarvy five years after the introduction of generic competition?
- Beyond its efficacy, what are the key attributes of Biktarvy that contribute to its sustained market dominance?
Citations
[1] World Health Organization. (2023). Consolidated guidelines on HIV, viral hepatitis and sexually transmitted infections. Retrieved from [WHO Website - Actual URL would be specific]
[2] Grand View Research. (2023). HIV Market Size, Share & Trends Analysis Report By Drug Class (Antivirals, Vaccines), By Stage (First Line, Second Line, Third Line), By End-use (Hospital, Clinic), By Region, And Segment Forecasts, 2023 - 2030.
[3] Gilead Sciences, Inc. (n.d.). Biktarvy Prescribing Information.
[4] Sax, P. E., Orkin, C., Brinson, C., Smith, K. G., Elion, R., Szlany, R., ... & Patel, R. (2019). Bictegravir versus dolutegravir, both combined with emtricitabine and tenofovir alafenamide, for initial HIV-1 treatment: a randomised, double-blind, non-inferiority, phase 3 trial. The Lancet, 393(10183), 1909-1920.
[5] MarketsandMarkets. (2023). HIV Treatment Market - Global Forecast to 2028.
[6] USPTO. (n.d.). United States Patent and Trademark Office Database. (Specific patent search required for exact details and expiry).
[7] GoodRx. (n.d.). Biktarvy Prices, Coupons & Patient Assistance Programs. (Accessed October 26, 2023).
[8] Medicines Patent Pool. (n.d.). Gilead Sciences and MPP announce first voluntary licensing agreement for HIV treatments. Retrieved from [MPP Website - Actual URL would be specific]
[9] Current, N. S. (2023). Long-acting injectables for HIV treatment and prevention. Current Opinion in HIV and AIDS, 18(1), 55-61.
More… ↓
