Share This Page
Drug Price Trends for BELSOMRA
✉ Email this page to a colleague
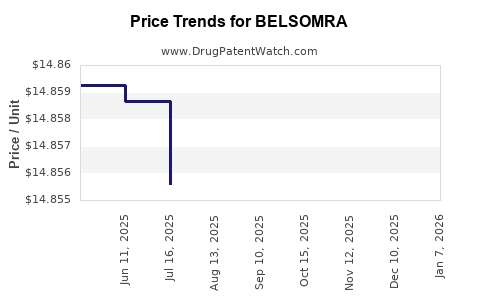
Average Pharmacy Cost for BELSOMRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BELSOMRA 20 MG TABLET | 00006-0335-30 | 15.20625 | EACH | 2026-05-20 |
| BELSOMRA 10 MG TABLET | 00006-0033-10 | 15.21065 | EACH | 2026-05-20 |
| BELSOMRA 15 MG TABLET | 00006-0325-10 | 15.19593 | EACH | 2026-05-20 |
| BELSOMRA 10 MG TABLET | 00006-0033-30 | 15.21065 | EACH | 2026-05-20 |
| BELSOMRA 5 MG TABLET | 00006-0005-30 | 15.17515 | EACH | 2026-05-20 |
| BELSOMRA 20 MG TABLET | 00006-0335-10 | 15.20625 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Belsomra: Patent Expiration and Market Impact Analysis
This report analyzes the patent landscape for Belsomra (suvorexant) and projects its market impact following patent expiration. Key patent assets are identified, and their expiration dates are correlated with anticipated generic entry and price erosion. The analysis considers market size, competition, and the potential for biosimilar or interchangeable drug development.
What are Belsomra's Key Patents and Their Expiration Dates?
Belsomra, developed by Merck & Co., is a dual orexin receptor antagonist used for the treatment of insomnia. Its patent protection is critical to understanding future market dynamics. The primary patent covering the compound itself is U.S. Patent No. 8,114,873.
- U.S. Patent No. 8,114,873: This patent claims the chemical compound suvorexant. Its terminal expiry is December 7, 2027. This is a foundational patent for Belsomra.
- U.S. Patent No. 8,664,274: This patent claims specific crystalline forms of suvorexant. Its terminal expiry is March 16, 2031.
- U.S. Patent No. 8,455,507: This patent covers methods of treating insomnia using suvorexant. Its terminal expiry is June 2, 2029.
- U.S. Patent No. 9,504,770: This patent relates to polymorphic forms of suvorexant. Its terminal expiry is October 16, 2032.
These patents, along with others related to manufacturing processes and formulations, form the core intellectual property protecting Belsomra. The expiration of U.S. Patent No. 8,114,873 in December 2027 is the most significant date for initial generic competition.
When is Generic Belsomra Expected to Enter the Market?
Generic entry for Belsomra is contingent on the expiration of its primary patents and the outcome of any potential patent litigation.
- Initial Generic Entry: Based on the expiry of U.S. Patent No. 8,114,873, generic versions of suvorexant are anticipated to become available in late 2027.
- Potential for Early Entry: While the '873 patent expires in December 2027, the possibility of earlier generic entry exists if any of the later-expiring patents are successfully challenged or invalidated through litigation.
- Exclusivity Periods: Regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity, may have already expired or will expire before the patent dates, allowing for generic applications to be submitted. Belsomra received NCE exclusivity in the U.S. on October 13, 2014, which expired in October 2019.
The actual date of generic market entry will be determined by the filing and approval of Abbreviated New Drug Applications (ANDAs) by generic manufacturers, which can occur once relevant patents are no longer in effect or are successfully challenged.
What is the Current Market Size and Sales Performance of Belsomra?
Belsomra has established a notable market presence since its launch, reflecting its therapeutic utility in insomnia management.
- 2023 Net Sales: Merck reported net sales of Belsomra to be approximately $578 million in 2023.
- 2022 Net Sales: In 2022, Belsomra generated net sales of approximately $549 million.
- Sales Growth: The drug has demonstrated consistent year-over-year sales growth, indicating sustained demand and market penetration. The increase from 2022 to 2023 represents a growth of approximately 5.3%.
The total market for insomnia treatments is substantial, driven by a high prevalence of sleep disorders. Belsomra competes within this segment against other prescription hypnotics and over-the-counter sleep aids.
What is the Projected Price Erosion Following Generic Entry?
The introduction of generic competition is expected to lead to significant price reductions for suvorexant.
- Typical Generic Erosion: In the pharmaceutical market, the entry of the first generic competitor typically results in an immediate price drop of 20-30% compared to the branded drug's average selling price.
- Further Erosion: As more generic manufacturers enter the market, prices can decline further, often reaching 70-90% less than the branded price within 18-24 months of the first generic launch.
- Price Projection: Post-December 2027, the average selling price of suvorexant is projected to decrease by at least 40% within the first year of generic availability, with further declines expected as competition intensifies. By 2030, prices could stabilize at 80-90% below 2027 levels.
This price erosion directly impacts revenue for the innovator company and creates cost savings for payers and patients.
What is the Competitive Landscape for Suvorexant?
Belsomra operates within a competitive therapeutic area characterized by both branded and generic alternatives.
- Branded Competitors:
- Lunesta (eszopiclone): A widely prescribed sedative-hypnotic.
- Ambien (zolpidem): Another established treatment for insomnia, available in various formulations.
- Rozerem (ramelteon): A melatonin receptor agonist.
- Generic Competitors: A significant number of generic versions of older insomnia medications, such as zolpidem and eszopiclone, are already available, providing substantial pricing pressure in the overall insomnia market.
- Emerging Therapies: While Belsomra is a novel mechanism of action (orexin receptor antagonism), ongoing research into sleep disorders could yield new therapeutic classes.
The availability of multiple generic options for existing insomnia treatments means that generic suvorexant will enter a market already accustomed to significant price competition for sleep medications.
What is the Potential Impact on Merck's Revenue and Market Share?
The expiration of Belsomra's core patents will significantly impact Merck's revenue from this product.
- Revenue Decline: Merck's revenue from Belsomra is projected to decline sharply following the entry of generic competitors. Sales are expected to decrease by an estimated 70-85% within two years of the first generic launch.
- Market Share Erosion: Merck's market share for suvorexant will diminish as generic manufacturers capture a substantial portion of the prescription volume. The company's share of the suvorexant market could fall from nearly 100% to below 20% within three years of generic entry.
- Strategic Considerations: Merck will likely focus on maximizing remaining sales before patent expiry and may leverage its established sales force and relationships to promote other products in its portfolio.
Merck's ability to mitigate these revenue losses will depend on the performance of its other pipeline drugs and existing commercialized products.
What are the Regulatory Pathways for Generic Suvorexant Approval?
Generic drug approval in the United States follows a defined regulatory pathway managed by the Food and Drug Administration (FDA).
- Abbreviated New Drug Application (ANDA): Generic manufacturers must submit an ANDA to the FDA.
- Bioequivalence: The core requirement for ANDA approval is demonstrating bioequivalence between the generic product and the reference listed drug (Belsomra). This means the generic drug must perform in the same way as Belsomra in terms of rate and extent of absorption.
- Patent Certification (Paragraph IV): Generic applicants often challenge existing patents by filing a Paragraph IV certification, asserting that the patents are invalid, unenforceable, or will not be infringed by the generic product. This can trigger patent litigation.
- Exclusivity Periods: Generic approval can also be influenced by regulatory exclusivities held by the innovator drug, which may block generic approval for a certain period.
The process can be lengthy and involve complex legal and scientific considerations, particularly concerning patent disputes.
What is the Global Market Outlook for Belsomra and its Generics?
The global market for insomnia treatments is substantial and growing, influenced by an aging population and increasing awareness of sleep disorders.
- Global Market Size: The global sleep aid market was valued at approximately $75 billion in 2022 and is projected to grow at a CAGR of 6-7% over the next five years.
- Regional Variations: North America and Europe represent the largest markets for insomnia medications, driven by high healthcare spending and prevalence rates. The Asia-Pacific region is expected to show the fastest growth.
- Generic Penetration: Similar to the U.S. market, generic entry in other major markets (e.g., Europe, Japan) will follow patent expiries in those regions, leading to significant price reductions and increased accessibility.
- Impact of Expiration: As key patents expire globally, generic suvorexant will likely capture a significant share of the global market for dual orexin receptor antagonists, mirroring the U.S. trajectory.
The global market dynamics will be shaped by regulatory approvals in different countries and the competitive strategies of generic manufacturers.
What are the Potential Risks and Opportunities Associated with Generic Belsomra?
The impending patent expiry presents both risks and opportunities for stakeholders.
- Risks for Merck:
- Significant revenue loss from Belsomra.
- Loss of market share to generic competitors.
- Potential for further patent litigation costs.
- Opportunities for Generic Manufacturers:
- Entry into a well-established market with a proven drug.
- Opportunity to capture significant market share due to price advantage.
- Potential for market expansion through lower pricing.
- Opportunities for Payers and Patients:
- Reduced healthcare costs due to lower drug prices.
- Increased access to effective insomnia treatment.
- Risks for Generic Manufacturers:
- Patent litigation initiated by Merck.
- Competition from multiple generic players entering the market simultaneously.
- Navigating complex regulatory approval processes.
Stakeholders will need to carefully manage these factors to optimize their positions in the post-patent expiration market.
Key Takeaways
- Belsomra's primary patent expires in December 2027, paving the way for generic suvorexant entry.
- Merck reported $578 million in Belsomra net sales in 2023, indicating a substantial market presence.
- Generic entry is expected to cause rapid price erosion, potentially reducing prices by 70-90% from branded levels within two years.
- The insomnia market is competitive, with established branded and generic alternatives, which will influence generic suvorexant's market penetration.
- Merck faces significant revenue and market share declines for Belsomra post-patent expiry.
- Generic manufacturers have a substantial opportunity to gain market share through competitive pricing.
- Payers and patients are poised to benefit from lower costs and increased treatment accessibility.
Frequently Asked Questions
1. When did Belsomra receive FDA approval?
Belsomra received FDA approval on October 13, 2014.
2. Are there any patent extensions or exclusivities that might delay generic entry beyond December 2027?
While U.S. Patent No. 8,114,873 expires in December 2027, other patents covering different aspects of suvorexant (e.g., crystalline forms, methods of use) have later expiry dates. The outcome of any patent litigation could impact the exact timeline for generic entry. Regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity, have already expired.
3. What are the main side effects of Belsomra that might influence physician prescribing of generics?
Common side effects of Belsomra include somnolence, headache, and dizziness. Physicians will consider these side effect profiles when comparing Belsomra and its generic equivalents to other insomnia treatments and when evaluating generic options for their patients.
4. How does Belsomra's mechanism of action (orexin receptor antagonism) compare to other insomnia drugs like Ambien (zolpidem)?
Belsomra works by blocking the wake-promoting orexin system in the brain. In contrast, zolpidem (Ambien) acts on GABA-A receptors, enhancing the inhibitory effects of the neurotransmitter GABA. This difference in mechanism may lead to distinct efficacy and side effect profiles.
5. What is the typical market share that generic drugs capture from a branded drug within one year of launch?
Typically, the first generic entry can capture 20-30% of the branded drug's market share within its first year, with this figure often increasing significantly as more generic competitors emerge.
Citations
[1] Merck & Co., Inc. (2024). 2023 Annual Report on Form 10-K. U.S. Securities and Exchange Commission. Retrieved from https://www.sec.gov/ix?doc=/Archives/edgar/data/320187/000119312524034336/d575783d10k.htm [2] U.S. Patent No. 8,114,873. (2012). Orexin Receptor Antagonists. [3] U.S. Patent No. 8,664,274. (2014). Polymorphic Forms of Orexin Receptor Antagonists. [4] U.S. Patent No. 8,455,507. (2013). Methods of Treating Sleep Disorders. [5] U.S. Patent No. 9,504,770. (2016). Polymorphic Forms of Orexin Receptor Antagonists. [6] Food and Drug Administration. (n.d.). Orphan Drug Designation. Retrieved from https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cdler/orphan-drug-designation [7] Grand View Research. (2023). Sleep Aid Market Size, Share & Trends Analysis Report By Type (Prescription Sleep Aids, OTC Sleep Aids), By Drug Class (Sedatives & Hypnotics, Melatonin Agonists, Antidepressants), By Distribution Channel, By Region, And Segment Forecasts, 2023-2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/sleep-aid-market
More… ↓
