Share This Page
Drug Price Trends for BASAGLAR
✉ Email this page to a colleague
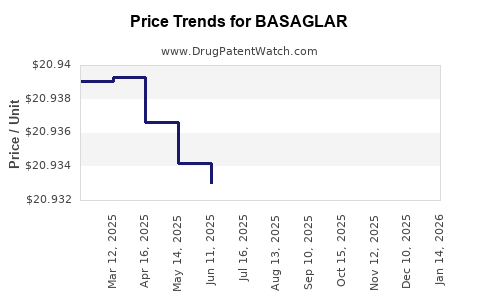
Average Pharmacy Cost for BASAGLAR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BASAGLAR 100 UNIT/ML KWIKPEN | 00002-7715-59 | 20.92997 | ML | 2026-04-22 |
| BASAGLAR 100 UNIT/ML KWIKPEN | 00002-7715-59 | 20.93087 | ML | 2026-03-18 |
| BASAGLAR 100 UNIT/ML KWIKPEN | 00002-7715-59 | 20.93041 | ML | 2026-02-18 |
| BASAGLAR 100 UNIT/ML KWIKPEN | 00002-7715-59 | 20.92930 | ML | 2026-01-21 |
| BASAGLAR 100 UNIT/ML KWIKPEN | 00002-7715-59 | 20.93104 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
BASAGLAR Market Analysis and Financial Projection
What is BASAGLAR?
BASAGLAR is a long-acting insulin glargine injection used to manage blood glucose levels in adults with diabetes mellitus type 1 and 2. Approved by the FDA in 2016, it is a biosimilar version of Sanofi’s Lantus (insulin glargine).
How does BASAGLAR fit into the insulin market?
The global insulin market was valued at approximately $24.2 billion in 2022. The rapid growth is driven by increasing diabetes prevalence, especially type 2 diabetes. BASAGLAR competes chiefly against originators like Lantus, and other biosimilars such as Lilly’s Basaglar.
What are the key market dynamics?
Competitive Landscape
- Lantus (Sanofi): Holds a significant share, with sales exceeding $4 billion in 2022.
- Basaglar (Eli Lilly): Approved as a biosimilar, capturing some market share from Lantus.
- Other biosimilars: E.g., Ganovo (BioChaperone), entered late 2020s, but with limited penetration.
Pricing and Reimbursement
- The average wholesale price (AWP) of Lantus in the U.S. was around $174 per pen as of 2022.
- Biosimilars like BASAGLAR have been priced 15–20% below their originators, with some discounts achieved through insurer negotiations.
- State Medicaid reimbursement rates and pharmacy benefit managers (PBMs) influence final patient costs.
Market Penetration
BASAGLAR’s market share has increased, driven by cost advantages and formulary placement. As of 2022, it accounts for approximately 10–15% of U.S. basal insulin prescriptions.
What are the price projections?
Short-Term Projections (2023–2025)
- Price stability: The average wholesale price for BASAGLAR likely remains within a 15% discount of Lantus, around $150–$165 per pen.
- Market share growth: Incremental increase driven by formulary placements and insurer contracts; projected to reach 20–25% in the U.S.
- Pricing pressure: May face bargaining from pharmacy benefit managers, affecting net prices.
Long-Term Projections (2026–2030)
- Potential price decrease: Biosimilar competition may reduce prices by an additional 10–15% over five years, driven by patent expirations (Lantus patent expired in 2015 globally).
- Market expansion: Growth in emerging markets could bring new revenue streams, although local pricing regulations may restrict pricing flexibility.
- Innovation inhibitors: No immediate biosimilar entrants for insulin glargine in development that threaten BASAGLAR’s position significantly.
Global Pricing Outlook
- In Europe, insulin prices are generally lower, with BASAGLAR priced at approximately €120–€140 per pen.
- In emerging markets, prices could be reduced by 30–50% due to pricing regulations and income levels.
What are the risks influencing price projections?
- Regulatory changes, such as import tariffs or price caps.
- Patent litigation outcomes could extend or shorten market exclusivity.
- Shifts toward alternative therapies, including ultra-long-acting insulins or oral agents.
- Potential biosimilar entrants with lower costs could press prices downward more aggressively.
How does government policy affect market and pricing?
- The U.S. Inflation Reduction Act of 2022 aims to cap insulin costs at $35 per month for Medicare recipients.
- European nations often employ negotiated pricing and reference pricing systems.
- Many countries have introduced or are considering price caps, impacting future revenue streams.
Key Takeaways
- BASAGLAR’s market position benefits from its lower price compared to Lantus.
- The overall insulin market is expected to grow, with biosimilars like BASAGLAR capturing increasing shares.
- Short-term prices are likely to remain stable with slight downward pressure, while long-term prices could decrease further due to competition.
- Market expansion in emerging regions provides potential growth but may also pressure pricing.
- Policy and regulatory changes constitute significant risks that can alter pricing strategies.
FAQs
Q1: Will BASAGLAR replace Lantus as the dominant basal insulin?
No. While BASAGLAR offers cost advantages, Lantus remains dominant due to brand recognition and healthcare provider familiarity. However, biosimilars are gradually increasing market share.
Q2: How will patent expirations impact BASAGLAR’s pricing?
Patent expirations encourage biosimilar competition, driving prices downward. BASAGLAR's grandfathered status may shield it temporarily, but biosimilars with lower costs could enter the market.
Q3: What factors could lead to a significant price reduction?
Market entry of new biosimilars, regulatory price controls, and negotiations from health authorities.
Q4: How does insurance coverage influence BASAGLAR pricing?
Insurance negotiations and formulary placements determine pharmacy benefit manager (PBM) discounts, directly affecting patient out-of-pocket costs.
Q5: Are there emerging therapies that threaten BASAGLAR’s market share?
Yes. Ultra-long-acting insulins like Tresiba (insulin degludec) and oral anti-diabetes drugs could lessen reliance on basal insulin injections.
Sources:
[1] IQVIA. "Global Insulin Market Report," 2022.
[2] U.S. Food and Drug Administration. "Biosimilar Insulins," 2016.
[3] Evaluate Pharma. "Insulin Market Forecast," 2023.
[4] Centers for Medicare & Medicaid Services. "Pricing and Reimbursement Policies," 2022.
[5] European Medicines Agency. "Pricing Regulations for Insulins," 2022.
More… ↓
