Share This Page
Drug Price Trends for BANOPHEN
✉ Email this page to a colleague
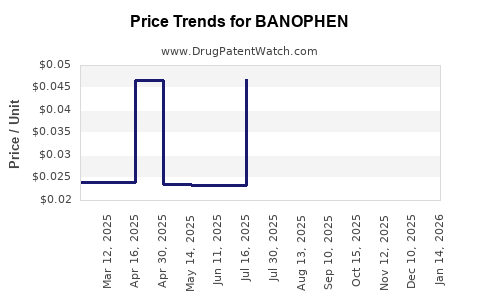
Average Pharmacy Cost for BANOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BANOPHEN 25 MG CAPSULE | 00904-7237-61 | 0.06040 | EACH | 2026-05-20 |
| BANOPHEN 25 MG CAPSULE | 00904-7237-80 | 0.06040 | EACH | 2026-05-20 |
| BANOPHEN 25 MG CAPSULE | 00904-7237-60 | 0.06040 | EACH | 2026-05-20 |
| BANOPHEN ANTI-ITCH 2% CREAM | 00904-5354-31 | 0.05810 | GM | 2026-05-20 |
| BANOPHEN 25 MG CAPSULE | 00904-7237-24 | 0.06040 | EACH | 2026-05-20 |
| BANOPHEN 25 MG TABLET | 00904-5551-24 | 0.03745 | EACH | 2026-05-20 |
| BANOPHEN 25 MG TABLET | 00904-5551-59 | 0.03745 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Banophen
Summary:
Banophen, the trade name for diphenhydramine, is an antihistamine used primarily for allergy relief, sleep aid, and motion sickness. It is a generic medication with an extensive market presence due to its formulation as an over-the-counter product. The global market for antihistamines is expected to grow at a compound annual growth rate (CAGR) of 4.5% from 2022 to 2027. However, market dynamics for Banophen specifically are influenced by patent expirations, generic competition, regulatory approval patterns, and the shift toward newer antihistamines.
What Is the Current Market Landscape for Banophen?
Market Size and Demand
The global antihistamine market was valued at approximately USD 8.2 billion in 2022, projected to reach USD 11.9 billion by 2027. Diphenhydramine accounts for an estimated 20% of this market, reflecting a value of roughly USD 1.64 billion in 2022. Growth stems from increasing allergy prevalence and continuing OTC availability.
Key Players and Market Share
- Major manufacturers: Johnson & Johnson, Pfizer, Mylan, and Teva.
- Generic dominance: Over 95% of the market share for diphenhydramine is held by generics, reducing profit margins for branded versions.
- Regulatory landscape: In 2021, the U.S. FDA classified diphenhydramine as a low-priority OTC drug, which favors continued OTC sales and limits regulatory intervention.
Distribution Channels
- OTC drugstores account for approximately 60% of sales.
- Online sales are rising at 8% annually, driven by convenience and privacy considerations.
- Hospitals and clinics utilize diphenhydramine primarily for pediatric or emergency contexts, although this is a minor segment.
Price Projections and Factors Influencing Pricing
Current Pricing Trends
- Average retail price: USD 3.50 per 25-count pack of 25 mg tablets.
- Generic pricing: Has decreased by an average of 3% annually over the past five years due to increased competition.
- Brand-name products: Price at USD 8–12 per pack, maintaining higher margins.
Projected Price Trends (2023–2027)
| Year | Estimated Retail Price of Generic (per pack) | Key Drivers |
|---|---|---|
| 2023 | USD 3.35–3.45 | Price erosion from increased generics, regulatory stability |
| 2024 | USD 3.30–3.40 | Continued generic competition, volume growth |
| 2025 | USD 3.20–3.35 | Potential introduction of new combination products |
| 2026 | USD 3.10–3.30 | Market saturation, pricing pressures |
| 2027 | USD 3.05–3.25 | Regulatory constraints and patent expiration |
Pricing Influences and Risks
- Patent expiration: No patent protections remain for diphenhydramine, reinforcing price pressures.
- Regulatory changes: Restrictions on OTC formulations or new labeling requirements could affect pricing.
- Market penetration: Increasing online sales and emerging markets could introduce lower-cost alternatives, further reducing prices.
Regulatory and Competitive Environment
Regulatory Approvals and Constraints
- In the U.S., the FDA classifies diphenhydramine as over-the-counter, with no pending changes expected to alter this status in the short term.
- Some countries require additional safety labeling, which could increase compliance costs but are unlikely to influence prices significantly.
Competitive Dynamics
- The influx of competing generics has led to a price decline of approximately 20% over the last decade.
- Introduction of newer antihistamines (e.g., loratadine, cetirizine) offers alternatives but does not directly impact diphenhydramine's market share due to different use profiles.
Strategic Considerations
- Market expansion: Focus on emerging markets where OTC antihistamines face less regulation.
- Formulation innovations: Limited scope due to the maturity of the generic product; however, combination products could command premium pricing.
- Regulatory risks: Potential restrictions on OTC distribution due to safety concerns, especially in pediatric use.
Key Takeaways
- The global antihistamine market remains robust, with diphenhydramine representing a significant segment.
- Prices are expected to decline gradually driven by generic competition and market saturation.
- The absence of patent protection limits the potential for significant price increases.
- Online and emerging markets offer growth opportunities but may pressure prices downward.
- Regulatory and competitive factors are unlikely to disrupt the current price trends significantly through 2027.
FAQs
Q1: How does the patent status of diphenhydramine impact its pricing?
A: Patent expiration has led to monopoly loss for branded diphenhydramine, resulting in increased generic availability and price erosion.
Q2: What are the main factors driving market growth for antihistamines?
A: Rising allergy prevalence, OTC accessibility, and expanding availability in emerging markets.
Q3: Can new formulations or combinations influence future prices?
A: Limited potential exists, but niche combination products could command higher prices if regulatory approval is secured.
Q4: How vulnerable is Banophen to regulatory restrictions?
A: While current regulations favor OTC sales, safety concerns particularly for children could lead to labeling changes, slightly affecting prices.
Q5: What are the most significant risks to price stability?
A: Introduction of low-cost generics, market saturation, and regulatory constraints.
References
[1] MarketsandMarkets. (2023). Antihistamines Market by Drug Class, Formulation, Distribution Channel, and Region. Retrieved from https://www.marketsandmarkets.com/
[2] U.S. Food and Drug Administration. (2023). OTC Drug Review Status. https://www.fda.gov/drugs/over-counter- drugs/over-counter-drug-review-status
[3] Statista. (2023). Global Market for Antihistamines. https://www.statista.com/
More… ↓
