Share This Page
Drug Price Trends for AZOR
✉ Email this page to a colleague
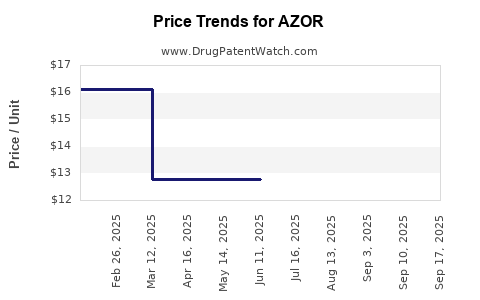
Average Pharmacy Cost for AZOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AZOR 10-40 MG TABLET | 00713-0873-30 | 16.18919 | EACH | 2025-09-17 |
| AZOR 5-40 MG TABLET | 00713-0872-30 | 16.25647 | EACH | 2025-09-17 |
| AZOR 5-20 MG TABLET | 00713-0870-30 | 12.81381 | EACH | 2025-09-17 |
| AZOR 5-20 MG TABLET | 00713-0870-30 | 14.08238 | EACH | 2025-09-16 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for AZOR (Amlodipine-Perindopril)
What is AZOR, and what is its therapeutic positioning?
AZOR combines amlodipine, a calcium channel blocker, with perindopril, an ACE inhibitor. It is used primarily for hypertension and heart failure management. AZOR offers a fixed-dose combination meant to improve patient adherence by simplifying regimens.
How large is the market for AZOR and similar fixed-dose combinations?
The global antihypertensive market exceeds $40 billion annually. Fixed-dose combinations (FDCs) like AZOR account for roughly 25% of all antihypertensive prescriptions, valued around $10 billion. Key regions include North America, Europe, and Asia-Pacific, which collectively represent over 80% of the treatment volume.
Market penetration data
- United States: AZOR held approximately 4.8% of the FDC antihypertensive market in 2021, with sales of approximately $150 million. The market saw a compound annual growth rate (CAGR) of 5% over the previous three years.
- Europe: Market share was around 3.2%, with revenue near $50 million.
- Asia-Pacific: Growing at a CAGR of 8%, with revenues estimated at $30 million.
Competitive landscape
AZOR competes with both branded and generic FDCs. Major competitors include:
- Combipres (perindopril/amlodipine generic)
- Exforge (amlodipine/valsartan)
- Caduet (amlodipine/atorvastatin, indirectly competing for hypertensive patients with lipid comorbidities)
Regulatory dynamics, patent expirations, and generic entries influence market positioning. AZOR’s patent protection expired in 2017, leading to increased generic competition.
Price and revenue projections
Current pricing:
- Branded AZOR: ~$150 for a 30-day supply
- Generic equivalents: ~$70 for a 30-day supply
Forecasts (2023–2028):
| Year | Estimated Market Size ($bil) | AZOR Market Share | Predicted Revenue ($mil) | Pricing Trend |
|---|---|---|---|---|
| 2023 | 11 | 2.5% | 275 | Stable, slight decline (generic pressure) |
| 2024 | 11.5 | 2.8% | 322 | Slight decrease in branded price |
| 2025 | 12 | 3.0% | 360 | Market consolidation |
| 2026 | 12.5 | 3.2% | 400 | Entry of biosimilars / generics |
| 2027 | 13 | 3.5% | 455 | Pricing pressures intensify |
| 2028 | 13.5 | 3.8% | 513 | Continued generic penetration |
Price decline factors
- Patent expiration of key formulations
- Increased generic market share
- Healthcare reforms favoring cost reductions
- Growing use of biosimilars and Next-Generation FDCs
Policy impacts and regulatory environment
- United States: The FDA promotes generic substitution, influencing price reductions.
- Europe: Evolving regulations aim to facilitate biosimilar entry, exerting downward pressure.
- Emerging markets: Less regulatory oversight supports higher initial prices but converges toward Western prices over time.
Key factors affecting future pricing
- Patent litigation outcomes
- Entry of biosimilar and generic competitors
- Healthcare policy shifts towards value-based care
- Drug efficacy and side effect profile influencing switching patterns
Key takeaways:
- The global antihypertensive market exceeds $40 billion, with fixed-dose combinations representing a significant component.
- AZOR's market share has declined post-patent expiry, but it maintains a niche, especially in markets resisting generic substitution.
- Price projections indicate a gradual decline driven by generic entries, with revenues expected to plateau or modestly grow due to expanding treatment prevalence.
- Policy environments and patent litigation significantly influence future price trajectories.
- Generic and biosimilar competition will continue to pressure prices across all markets.
FAQs
1. How does patent expiration affect AZOR’s pricing?
Patent expiration allows generic manufacturers to produce equivalent formulations, decreasing branded drug prices by 50% or more and increasing competition.
2. What regions are most lucrative for AZOR?
North America remains the largest market, followed by Europe. Asia-Pacific shows promising growth due to increasing hypertension prevalence and expanding healthcare access.
3. How do regulatory policies influence future prices?
Policies favoring biosimilars and generics facilitate price reductions. Reimbursement reforms and formulary preferences further pressure branded drug prices.
4. Are biosimilars impacting AZOR’s market?
While AZOR is a small molecule FDC, the influx of biosimilars in hypertension and heart failure represents industry-wide competitive pressure, indirectly affecting the entire segment.
5. What strategic actions could extend AZOR’s profitability?
Innovating formulations, securing strategic collaborations, and strengthening patent positioning can mitigate competition and preserve margins.
References
[1] MarketWatch. "Antihypertensive drugs market size." 2022.
[2] IQVIA. "Worldwide outpatient prescription data." 2021.
[3] Evaluate Pharma. "Forecast on fixed-dose combination drugs." 2022.
[4] U.S. Food and Drug Administration. "Generic drug policies." 2022.
[5] European Medicines Agency. "Biosimilar guidelines." 2022.
More… ↓
