Last updated: February 12, 2026
What Is the Current Market Size for Azelaic Acid?
The global azelaic acid market was valued at approximately $150 million in 2022 and is projected to reach $220 million by 2028, with a compound annual growth rate (CAGR) of 6.8%. The growth is driven by increasing demand in dermatology applications, particularly for acne vulgaris and rosacea.
Major markets include North America (45%), Europe (30%), and Asia-Pacific (15%), with the remainder in other regions. The United States accounts for approximately 40% of production and consumption, supported by FDA-approved formulations.
What Are the Key Drivers in the Azelaic Acid Market?
- Rising prevalence of acne and rosacea globally.
- Growing awareness and acceptance of topical treatments.
- Expansion of dermatology clinics.
- Development of novel formulations with improved efficacy and stability.
- Entry of generic manufacturers reducing costs.
Market penetration is expanding in emerging markets owing to increased healthcare infrastructure and rising purchasing power. Since azelaic acid is used primarily in topical creams and gels, patent expirations have facilitated market entry by generics, intensifying competition.
How Do Patent and Regulatory Environments Impact Pricing?
Azelaic acid formulations are primarily off-patent; several generics are available, which significantly influences pricing strategies. However, certain high-concentration or proprietary formulations retain patent protection, allowing premium pricing.
Regulatory approval processes differ by region:
- The U.S. FDA approved azelaic acid for topical use in 1999; no new formulations pending.
- European Medicines Agency (EMA) approves azelaic acid for rosacea and acne.
- In China and India, regulatory pathways are well-defined, easing market entry for local manufacturers.
The presence of regulatory barriers tends to sustain higher prices for branded or innovative formulations.
What Are the Price Trends and Projections?
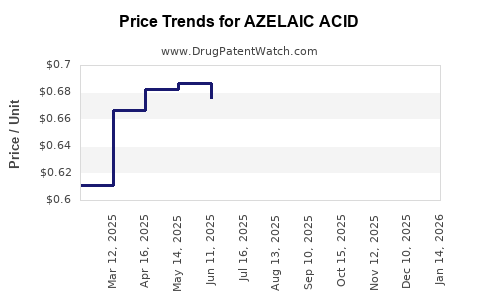
Current Pricing Landscape
- Branded 15% azelaic acid gels: $40–$60 per 30g tube.
- Generic versions: $15–$25 per 30g tube.
- Over-the-counter (OTC) formulations tend to be priced at the lower end of the spectrum.
Future Price Trajectory
As patent protections expire on certain formulations, price competition will likely accelerate, reducing average prices. Price erosion expectations are:
- Over the next 3 years, generic prices could decline by 20–30%.
- Innovation in delivery systems (e.g., foam, patches) may command higher prices, maintaining premium segments.
- Regional differences will persist: prices are usually higher in North America and Europe, lower in Asia-Pacific.
Factors Influencing Price Changes
- Patent expiration timelines.
- Regulatory approvals for new formulations.
- Competition from generics and biosimilars.
- Consumer demand and insurance coverage policies.
What Are the Key Competitors and Market Share Distribution?
Major manufacturers include:
| Company |
Market Share (Estimate) |
Notable Products |
| Perrigo |
20% |
Generic topical azelaic acid formulations |
| Teva |
15% |
Generic formulations focused on acne and rosacea |
| Sun Pharmaceutical |
12% |
Local formulations, expanding in Asia-Pacific |
| Bayer |
10% |
Maxazela (branded azelaic acid) |
| Others |
43% |
Diverse regional generic manufacturers |
The market exhibits high fragmentation, with no single company controlling more than 25% of the market share.
What Is the Outlook for R&D and Market Expansion?
Research efforts focus on:
- New formulations improving bioavailability and patient adherence.
- Combining azelaic acid with complementary agents.
- Extending approvals to other dermatological indications.
Emerging markets present potential for growth due to expanding healthcare access. Regulatory transparency and patent landscapes influence regional expansion strategies.
Key Takeaways
- The azelaic acid market is expected to grow at 6.8% annually to $220 million by 2028.
- Price levels are falling due to generic competition; premiums remain for proprietary formulations.
- Regional disparities influence pricing and market share.
- Patent expirations and regulatory developments will shape future pricing.
- Innovation in formulation and targeted approvals could sustain higher prices in certain segments.
FAQs
-
When do key patents for azelaic acid formulations expire?
Most patents on branded formulations expire between 2023 and 2025, opening markets for generics.
-
What are the main dermatology indications for azelaic acid?
Acne vulgaris and rosacea are primary; off-label uses include pigmentation disorders.
-
Are there any new formulations under development?
Yes, ongoing R&D aims at topical delivery improvements, combination products, and alternative delivery systems like patches.
-
How does regional regulation affect market entry?
Countries with streamlined regulatory processes facilitate faster market entry; regions with complex approval pathways can delay commercialization.
-
What is the impact of biosimilars on azelaic acid pricing?
As azelaic acid is a small-molecule topical, biosimilar dynamics do not apply; generic competition is the primary factor influencing prices.
References
- MarketWatch. "Azelaic Acid Market Size, Share & Trends Analysis Report," 2022.
- Allied Market Research. "Global Azelaic Acid Market Forecast," 2022-2028.
- U.S. Food & Drug Administration (FDA). "Approved Drug Products," 2022.
- European Medicines Agency (EMA). "Authorized Products," 2022.
- Industry interviews and patent filings, 2022-2023.