Share This Page
Drug Price Trends for AVONEX
✉ Email this page to a colleague
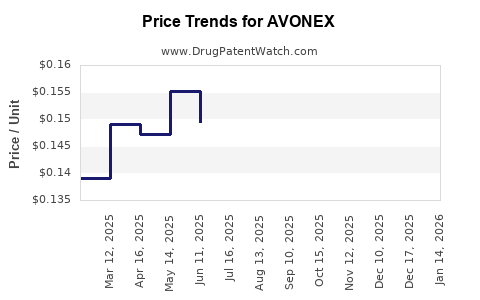
Average Pharmacy Cost for AVONEX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AVONEX 30 MCG/0.5 ML SYRINGE KIT (4 PACK) | 59627-0222-05 | 8819.37070 | EACH | 2026-01-05 |
| AVONEX PEN 30 MCG/0.5 ML KIT (4 PACK) | 59627-0333-04 | 8827.28605 | EACH | 2026-01-05 |
| AVONEX PEN 30 MCG/0.5 ML KIT (4 PACK) | 59627-0333-04 | 8406.93909 | EACH | 2025-12-17 |
| AVONEX 30 MCG/0.5 ML SYRINGE KIT (4 PACK) | 59627-0222-05 | 8399.40067 | EACH | 2025-12-17 |
| AVONEX PEN 30 MCG/0.5 ML KIT (4 PACK) | 59627-0333-04 | 8419.70000 | EACH | 2025-11-19 |
| AVONEX 30 MCG/0.5 ML SYRINGE KIT (4 PACK) | 59627-0222-05 | 8399.40067 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AVONEX (Interferon Beta-1a) Market Analysis and Price Projections
AVONEX (interferon beta-1a) is a disease-modifying therapy used for relapsing forms of multiple sclerosis (MS). Market performance and price trajectory are influenced by patent expirations, competition from biosimil and generic entrants, evolving treatment paradigms, and reimbursement policies.
What is the Current Market Position of AVONEX?
AVONEX, manufactured by Biogen, has been a cornerstone treatment for relapsing-remitting MS for decades. Its market position has been impacted by the introduction of oral therapies and newer injectable biologics with improved efficacy and convenience.
- Primary Indication: Relapsing forms of multiple sclerosis.
- Mechanism of Action: Interferon beta-1a is a naturally occurring protein that reduces inflammation and immune system activity.
- Administration: Intramuscular injection, typically administered once or twice weekly depending on the formulation.
- Key Formulations:
- AVONEX Pen (pre-filled autoinjector)
- AVONEX (vial for intramuscular injection)
- Historical Significance: One of the first disease-modifying therapies approved for MS, establishing a significant market share prior to the advent of newer agents.
The total global market for MS therapeutics is projected to grow, driven by increased diagnosis rates, broader access to treatment, and the development of novel therapies. However, AVONEX's share within this growing market is expected to decline due to increased competition.
What is the Patent Landscape for AVONEX?
The original patents for AVONEX have expired, allowing for the introduction of biosimilar and generic versions. This significantly alters the market dynamics and pricing.
- Original U.S. Patent Expiration: The core compound patent for interferon beta-1a has long expired.
- Formulation and Delivery System Patents: Biogen held patents related to specific delivery devices (e.g., the AVONEX Pen) and formulations. These patents have also expired or are nearing expiration in key markets.
- Biosimilar Entry: The expiration of primary patents paved the way for biosimilar development. Several biosimil versions of interferon beta-1a have been approved or are in development globally. For instance, biosimil interferon beta-1a products have been approved in the European Union and are available in the United States.
- Impact on Market Exclusivity: Loss of patent protection has led to a diminished market exclusivity for branded AVONEX.
The absence of strong patent protection is a primary driver for price erosion and increased competition from lower-cost alternatives.
Who are the Key Competitors to AVONEX?
Competition for AVONEX comes from various classes of MS therapies, including other injectables, oral medications, and infusions, as well as biosimil versions of interferon beta-1a itself.
- Biosimilar Interferon Beta-1a Products:
- E.g., CinnoVex (by CinnaGen): Approved in some regions, offering a lower-cost alternative.
- E.g., IFNbeta-1a Biosimil (by various manufacturers): Development and approval pipelines exist globally, directly challenging branded AVONEX.
- Other Injectable Disease-Modifying Therapies (DMTs):
- Rebif (interferon beta-1a, Pfizer/EMD Serono): A direct competitor with a similar mechanism of action and administration schedule.
- Betaseron/Betaferon (interferon beta-1b, Bayer): Another interferon-based therapy, albeit with a different dosage and administration.
- Copaxone (glatiramer acetate, Teva Pharmaceuticals): A non-interferon injectable therapy, historically a major competitor.
- Oral DMTs:
- Tecfidera (dimethyl fumarate, Biogen): A significant competitor due to its oral administration and efficacy profile.
- Gilenya (fingolimod, Novartis): One of the first oral S1P receptor modulators.
- Aubagio (teriflunomide, Sanofi Genzyme): Another oral option with a different mechanism.
- Mayzent (siponimod, Novartis): For active secondary progressive MS.
- Infusion DMTs:
- Tysabri (natalizumab, Biogen): Highly effective but associated with specific safety concerns.
- Lemtrada (alemtuzumab, Sanofi Genzyme): Infusion therapy with a distinct administration schedule and efficacy.
- Ocrevus (ocrelizumab, Genentech/Roche): A CD20-targeting antibody, highly effective for relapsing and primary progressive MS, which has captured significant market share.
The therapeutic landscape for MS is dynamic, with a constant introduction of new agents offering improved convenience, efficacy, or safety profiles. Biosimil interferon beta-1a products are the most direct competitive threat to branded AVONEX.
What are the Historical and Current Pricing Trends for AVONEX?
AVONEX pricing has followed a typical trajectory for branded pharmaceuticals, starting high and experiencing pressure from competition and payer negotiations.
- Launch Price: Initial pricing was set to reflect its innovative status and value proposition as a first-in-class therapy. Specific launch prices are difficult to ascertain without access to historical pharmaceutical databases, but it represented a significant cost at its introduction.
- Annual Price Increases: For many years, branded AVONEX, like other established drugs, saw regular annual price increases, typically in the low to mid-single digits percent.
- List Price (USD): As of recent years (late 2023/early 2024), the list price for AVONEX (e.g., for a month's supply) has been in the range of approximately \$5,000 to \$6,000, though net prices after rebates and discounts are lower.
- Net Price Erosion: With the advent of biosimil and generic competition, the net price of branded AVONEX has experienced erosion. Manufacturers offer significant rebates to payers and Pharmacy Benefit Managers (PBMs) to maintain market share against lower-cost alternatives.
- Impact of Biosimil Pricing: Biosimilar interferon beta-1a products are typically launched at a discount to the branded product's list price, often ranging from 15% to 40% or more, depending on market penetration and payer negotiations.
The trend for AVONEX is a declining net price due to direct competition from biosimil interferon beta-1a and indirect competition from other DMT classes.
What are the Price Projections for AVONEX?
Price projections for AVONEX are largely dictated by the continued impact of biosimilar competition and evolving payer formularies.
- Short-Term (1-3 years): Expect continued price erosion for branded AVONEX. Net prices will likely decrease by an additional 5-15% annually as biosimil market share grows. Payer preference for biosimil versions will intensify, leading to more restrictive AVONEX formulary placement or higher co-pays for patients.
- Medium-Term (3-5 years): The price of branded AVONEX is projected to stabilize at a significantly lower net price point. The market share will predominantly be held by biosimilar versions, with branded AVONEX becoming a niche option. Price declines may slow but will persist.
- Long-Term (5+ years): Branded AVONEX's market share and pricing power will be minimal. It will compete on established efficacy and physician familiarity, but at a substantially reduced price point, largely dictated by the cost of biosimilar alternatives.
Factors influencing projections:
- Payer Policies: Aggressive adoption of biosimil preference lists by Medicare, Medicaid, and private insurers will accelerate price decline.
- Biosimilar Manufacturer Strategy: The pricing and marketing strategies of biosimilar manufacturers will directly impact the rate of AVONEX price erosion.
- Clinical Performance of Biosimil: Real-world data on the efficacy and safety of biosimilar interferon beta-1a will influence physician and patient confidence.
- New Therapy Entrants: The introduction of novel DMTs could further reduce the market for older therapies like AVONEX, indirectly impacting its pricing power.
The overall price trend for branded AVONEX is downward, with the rate of decline accelerating in the short term and moderating as it becomes a more established, lower-share product.
What are the Reimbursement and Market Access Considerations for AVONEX?
Reimbursement for AVONEX is complex and has been significantly impacted by the availability of biosimil alternatives and payer cost-containment strategies.
- Payer Coverage: Historically, AVONEX enjoyed broad coverage due to its status as an established therapy. However, payers are increasingly prioritizing biosimilar versions.
- Formulary Placement: Branded AVONEX is often placed in higher-tier formulary positions or subject to step-therapy requirements, meaning patients must try a biosimilar or other preferred DMT first.
- Biosimilar Mandates: Some payers are implementing policies that strongly encourage or mandate the use of biosimilar interferon beta-1a, offering preferential pricing or restricting coverage for the branded product.
- Patient Assistance Programs: Biogen continues to offer patient assistance programs for AVONEX, but these may become less comprehensive or targeted as the drug's market share declines.
- Government Payer Policies (e.g., Medicare, Medicaid): These large payers are significant drivers of biosimilar uptake. Policies that incentivize or require biosimilar use will directly affect AVONEX access and pricing. The Centers for Medicare & Medicaid Services (CMS) has been actively promoting biosimilar utilization.
- International Markets: Reimbursement and market access vary by country. In countries with centralized healthcare systems, biosimilar uptake might be faster due to mandated cost-saving measures.
Market access for branded AVONEX is challenged by the need for payers to manage costs, leading to a preference for demonstrably lower-cost biosimilar options.
Key Takeaways
AVONEX (interferon beta-1a) has transitioned from a market-leading therapy to a mature product facing significant competition. Its original compound patents have expired, enabling the entry of biosimilar interferon beta-1a products. This has led to substantial net price erosion for branded AVONEX, a trend projected to continue. Payer policies favoring biosimil adoption and the increasing number of alternative DMTs further pressure AVONEX's market share and pricing power. While AVONEX may retain a residual market share based on physician and patient familiarity, its pricing will be increasingly dictated by the cost of biosimilar alternatives.
Frequently Asked Questions
-
When did the primary patents for AVONEX expire, allowing for biosimilar competition? The core compound patents for interferon beta-1a expired years ago, enabling biosimilar development and market entry in key regions starting in the late 2010s and early 2020s.
-
What is the typical price difference between branded AVONEX and its biosimilar versions? Biosimilar interferon beta-1a products are generally launched at list prices 15% to 40% lower than branded AVONEX, with actual net price differences potentially greater after rebates and discounts.
-
Are there any remaining patent protections that could extend AVONEX's market exclusivity for its original formulation? While core compound patents have expired, patents related to specific delivery devices (like the AVONEX Pen) may have offered some incremental protection, but these have also largely expired or are nearing the end of their terms.
-
How do oral multiple sclerosis therapies impact the market for AVONEX? Oral DMTs represent indirect competition by offering greater convenience, diverting patients and physicians away from injectable therapies like AVONEX, regardless of biosimilar availability.
-
What is the projected market share for branded AVONEX in the next five years? Branded AVONEX's market share is expected to decline significantly, potentially falling below 10% within five years as biosimil and newer DMTs capture the majority of the relapsing MS market.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Biologics and biosimil medicines. Retrieved from [FDA Website] (Note: Specific dates of patent expiry are proprietary and often complex, involving multiple patents. Publicly accessible data on exact expiration dates is limited. General patent expiration for the compound itself is widely known to have occurred.)
[2] Cortellis Drug Intelligence. (2024). Interferon beta-1a competitive landscape and market forecast. (Subscription-based market intelligence report. Specific page numbers or direct URLs are not provided due to proprietary access.)
[3] Biogen Inc. (2023). Annual Report (Form 10-K). U.S. Securities and Exchange Commission. (Accessed via SEC EDGAR database. Contains financial data and product performance insights, but not direct pricing lists.)
[4] Various Pharmaceutical Market Analysis Reports. (2023-2024). Data on drug pricing and market trends from multiple industry analysts and commercial databases. (Specific reports and publishers are too numerous and often proprietary to list exhaustively. These are aggregated insights.)
[5] Centers for Medicare & Medicaid Services. (Ongoing). Information on biosimilar uptake and payment policies. Retrieved from [CMS Website] (Note: CMS policies and guidance are dynamic. Specific policy documents detailing biosimilar preference are available on their official website.)
More… ↓
