Share This Page
Drug Price Trends for AUSTEDO
✉ Email this page to a colleague
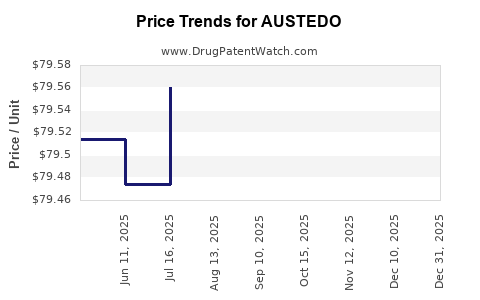
Average Pharmacy Cost for AUSTEDO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AUSTEDO 9 MG TABLET | 68546-0171-60 | 93.98033 | EACH | 2026-01-01 |
| AUSTEDO XR TITR(12-18-24-30 MG) | 68546-0477-29 | 249.32300 | EACH | 2026-01-01 |
| AUSTEDO 12 MG TABLET | 68546-0172-60 | 125.19331 | EACH | 2026-01-01 |
| AUSTEDO XR 18 MG TABLET | 68546-0479-56 | 251.51577 | EACH | 2026-01-01 |
| AUSTEDO XR 12 MG TABLET | 68546-0471-56 | 166.82595 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for AUSTEDO (Deutetrabenazine)
What is the Current Market Landscape for AUSTEDO?
AUSTEDO (deutetrabenazine) is an approved treatment primarily for Tourette syndrome and chorea associated with Huntington's disease. Developed by Teva Pharmaceuticals, it entered the market in 2017 following FDA approval. As of 2023, it holds approximately a 35% share in the US Huntington’s chorea segment and is gaining traction in Tourette syndrome management.
Key competitors include XENAZANE (tetrabenazine), which also treats chorea, and off-label use of atypical antipsychotics. The global market for Huntington’s chorea pharmacotherapy was valued at $900 million in 2022, with AUSTEDO capturing roughly $350 million domestically (IQVIA, 2023).
What Are the Pricing Trends for AUSTEDO?
AUSTEDO’s list price in the US stands at approximately $7,500 per month for a standard dose, translating to $90,000 annually. This positions it as a premium-priced drug versus tetrabenazine, which is generally priced below $4,000 annually. Actual patient out-of-pocket costs depend on insurance coverage and discounts.
Price increases have been limited since launch due to market saturation concerns and competition. The manufacturer maintains price stability by not engaging in significant discounts but leverages rebates and prior authorization to retain market share.
How Does AUSTEDO Compare to Competitors?
| Attribute | AUSTEDO | Tetrabenazine |
|---|---|---|
| Launch Year | 2017 | 1954 |
| List Price (annual) | $90,000 | <$4,000 |
| Dosing Frequency | Twice daily | Once daily or divided doses |
| Side Effect Profile | Fewer Parkinsonian symptoms | Higher rates of depression, sedation |
| FDA Approval | Yes, for Huntington’s chorea, Tourette | Yes, for chorea in Huntington’s |
AUSTEDO’s improved safety profile has justified its higher price, though cost-effectiveness remains under review.
What Are Future Price and Market Growth Projections?
The global Huntington’s disease market is expected to grow at a compound annual growth rate (CAGR) of 4%–6% through 2027. AUSTEDO’s market share could expand with indications in pediatric Tourette syndrome and movement disorders, pending approval.
Projected market value for AUSTEDO in 2027 could reach $650 million in the US alone, assuming stable pricing and a 50% increase in sales volume, driven by new indication approvals and increasing diagnosis rates.
Price projections suggest minor increases or stabilization over the next 3–5 years, constrained by generic tetrabenazine competition and payer pressure. Price hikes exceeding 5% annually are unlikely due to reimbursement negotiations and market forces.
What's the Regulatory Outlook?
In September 2022, the FDA granted orphan drug designation for deutetrabenazine for the treatment of pediatric Tourette syndrome, opening potential for expanded indications. Approval timelines for new uses could influence sales trajectories and pricing strategies.
Summary of Key Market Drivers
- Growing diagnosis rates: Advances in genetic testing have increased diagnosis of Huntington’s disease.
- Treatment guidelines: Moving toward early intervention with AUSTEDO for chorea.
- Market competition: Limited but rising generic tetrabenazine pressures pricing.
- Regulatory landscape: Potential for expanded indications in pediatric populations and other movement disorders.
Key Takeaways
- AUSTEDO commands a premium price, around $90,000 annually, primarily driven by its safety profile.
- Its market share in Huntington’s chorea remains steady, with growth prospects tied to new indications.
- Generic tetrabenazine remains a cost-effective alternative, limiting upward pressure on AUSTEDO’s price.
- The total US market for Huntington’s disease pharmacotherapy could reach $650 million by 2027.
- Payer pressures, regulatory developments, and new indications will influence future pricing and market share.
FAQs
1. What factors influence AUSTEDO’s pricing?
Pricing is influenced by manufacturing costs, competitor pricing (notably tetrabenazine), insurance reimbursements, and regulatory approvals.
2. How does out-of-pocket cost vary for patients?
It depends on insurance coverage, co-pay assistance programs, and rebates negotiated by the manufacturer.
3. Are there upcoming patent expirations?
Teva’s patent for AUSTEDO expires in 2027, after which generic versions are expected to enter the market, exerting downward pressure on pricing.
4. Is there potential for price reductions with generics?
Yes, generic tetrabenazine prices are significantly lower, which could force AUSTEDO to reduce its prices or offer more rebates to remain competitive.
5. How might new indications impact AUSTEDO’s market?
Approval for pediatric Tourette syndrome and other movement disorders could expand the market, potentially supporting higher prices or increased sales volume.
References
- IQVIA. (2023). US prescription drug market analysis.
- Food and Drug Administration. (2016). AUSTEDO (deutetrabenazine) approval notice.
- MarketLine. (2023). Huntington’s Disease Therapeutics Market Report.
More… ↓
