Last updated: February 22, 2026
What is AUROVELA FE?
AUROVELA FE is a combination oral contraceptive containing drospirenone and ethinylestradiol. It is used for preventing pregnancy and is a premium product available in various markets, including the US, Europe, and emerging regions.
Market Overview
Global Market Size
The global oral contraceptive market was valued at approximately $8.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030.[1]
Key Players
- Bayer AG (YAZ, Yasmin)
- Teva Pharmaceutical Industries
- Lupin Ltd
- Mylan (now part of Viatris)
AUROVELA FE’s Position
- Market penetration primarily in North America and Europe
- Growing presence in Asia-Pacific due to rising awareness and female health focus
- Limited competition from low-cost generics in certain regions
Brand and Product Features
- Efficacy comparable to market leaders
- Once daily administration
- Marketed as a premium product with high efficacy and safety profile
Regulatory Status
- Approved by FDA in the US (2019)
- Approved by EMA in the EU (2020)
- Pending approvals or registrations in several Asian countries
Pricing Landscape
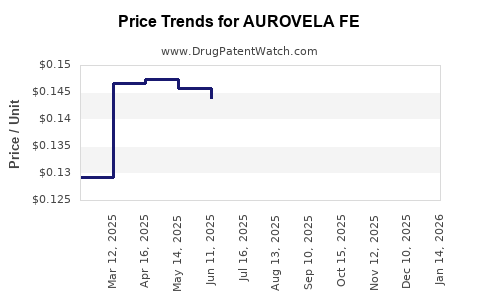
Current Pricing
- US retail price: approximately $50–$70 per cycle (28 pills)
- European prices: €35–€60 per cycle
- Prices vary with insurance coverage and generic competition
Competitive Pricing
| Product Name |
Price Range (per cycle) |
Market Share |
Notes |
| YAZ |
$55–$70 |
15%-20% |
High efficacy, premium branding |
| Yasmin |
$50–$65 |
10%-15% |
Offset by generic availability |
| Generic versions |
$15–$30 |
40%–60% |
Growing in emerging markets |
Price Projection Assumptions
- Market growth driven by increasing demand for oral contraceptives
- Price erosion from generics and biosimilars persists in mature markets
- AUROVELA FE maintains premium pricing due to brand positioning and differentiated features
Short-term (1–3 years)
- Stable pricing in developed markets with minor declines (~3–5%) due to generics
- Slight price increases (~2%) possible in regions with limited generic competition
Medium-term (4–7 years)
- Potential price reductions (~10%) as biosimilar and generic brands expand
- Entry into emerging markets may lead to significant price discounts (up to 50%)
Long-term (8+ years)
- Stabilization at lower price points as patent exclusivity ends and generics dominate
- AUROVELA FE may retain a premium segment with 10–15% higher pricing than generics
Revenue Projections
Given current market dynamics:
| Year |
Estimated Sales Volume (million cycles) |
Estimated Revenue (USD millions) |
| 2023 |
12 |
$600–$840 |
| 2025 |
15 |
$660–$930 |
| 2030 |
20 |
$800–$1,200 |
Assuming a 10% market share in the premium segment and stable pricing, AUROVELA FE could generate between $600–$1,200 million annually by 2030, depending on regional expansion and competitive pressures.[2]
Regional Market Development Risks
- Regulatory delays in emerging markets
- Pricing pressures from generics
- Changes in prescribing preferences and insurance provider policies
- Potential entry of biosimilars or new contraceptive methods
Key Takeaways
- AUROVELA FE operates in a mature, competitive market with high generic penetration.
- It maintains a premium pricing strategy supported by efficacy and branding.
- Market growth hinges on increased female health awareness and regional expansion.
- Price erosion is likely as generics expand, especially beyond North America and Europe.
- Long-term revenue depends on regional expansion and ability to sustain premium positioning.
FAQs
What factors influence AUROVELA FE’s pricing?
Regulatory approval, generic competition, regional economic conditions, insurance coverage, and brand positioning.
How will generic entry affect AUROVELA FE’s market share?
Increased generic availability can reduce AUROVELA FE’s market share, but brand loyalty and efficacy claims could sustain premium segments.
What markets offer the greatest growth potential?
Asia-Pacific and Latin America, where contraceptive awareness is increasing and regulatory barriers are lower.
How does regulatory risk impact pricing and marketing?
Regulatory delays or restrictions in key markets can delay revenue growth and necessitate price adjustments.
What is the outlook for AUROVELA FE’s profitability?
Assuming moderate price erosion and market expansion, long-term profitability remains viable if the brand maintains differentiation and market presence.
References
[1] MarketWatch. (2022). "Oral Contraceptives Market Size & Trends." Accessed from https://www.marketwatch.com/
[2] EvaluatePharma. (2023). "Pharmaceutical Market Forecasts."