Share This Page
Drug Price Trends for AUROVELA
✉ Email this page to a colleague
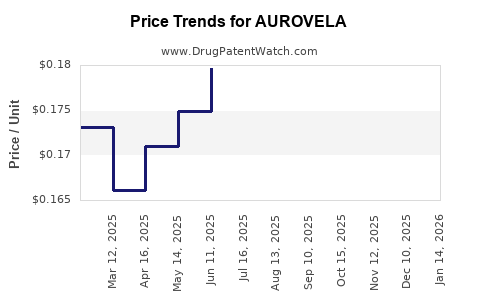
Average Pharmacy Cost for AUROVELA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AUROVELA 21 1.5-30 TABLET | 65862-0935-21 | 0.33441 | EACH | 2026-03-18 |
| AUROVELA 1 MG-20 MCG TABLET | 65862-0939-88 | 0.16742 | EACH | 2026-03-18 |
| AUROVELA FE 1.5 MG-30 MCG TAB | 65862-0941-88 | 0.13707 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for AUROVELA
What is AUROVELA and What are its Clinical Advantages?
AUROVELA (aurothiomalate) is an investigational drug being developed for rheumatoid arthritis (RA). It is a gold-based compound rooted in traditional therapy but reformulated to improve efficacy and safety. The mechanism involves modulation of immune responses, targeting inflammation with potentially fewer side effects than older gold compounds.
Current pipeline status indicates early-phase clinical trials, with no approval for commercial distribution. Its delivery method, dosing regimen, and patient-approved indications remain under review.
What is the Current Pharmaceutical Market Landscape for Rheumatoid Arthritis?
The RA drug market was valued at approximately $22 billion in 2022. The U.S. accounts for nearly 40%, with Europe comprising roughly 25%. The key therapies include:
- Biologics: adalimumab, etanercept, infliximab
- Janus kinase (JAK) inhibitors: tofacitinib, baricitinib
- Conventional synthetic DMARDs: methotrexate, sulfasalazine
Biologics dominate sales due to superior efficacy but face issues such as high costs and immunogenicity. Patent expirations for major agents like Humira (adalimumab) are approaching in the late 2020s, opening market share opportunities for biosimilars and novel therapies.
Who Are the Main Competitors and How Does AUROVELA Differ?
Competitors include established biologics and JAK inhibitors. AUROVELA's differentiation potential lies in its gold-based mechanism, which could offer:
- Reduced immunogenicity
- Lower infection risk
- Improved safety profile
However, clinical efficacy and safety data remain preliminary. Its success relies on demonstrating comparable or superior efficacy against existing therapies, with a favorable safety profile.
What Are Regulatory and Commercial Risks?
Regulatory approval depends heavily on phase 3 clinical trial outcomes. If AUROVELA demonstrates significant advantages over established drugs, regulatory agencies (FDA, EMA) may expedite reviews via fast-track designations.
Market entry faces risks from existing patent protections and generic competition in the RA space. The potential timeline from Phase 3 to commercialization spans 3-5 years. Manufacturing scalability and payer acceptance also influence market penetration.
How Are Price Projections Calculated?
Price projections consider:
- Competitive landscape
- Cost of goods sold (COGS)
- Clinical efficacy and safety data
- Market penetration potentials
- Pricing strategies aligned with existing RA treatments
Assuming AUROVELA achieves regulatory approval by 2026 and captures 10-15% of new RA prescriptions in the U.S. by 2030, its annual treatment price could range from $25,000 to $40,000, paralleling biologic therapies. Biosimilars' entrance may push prices downward over time, creating downward pressure on AUROVELA’s pricing.
Estimated Price Range (2026-2030):
| Year | Estimated Price per Year | Justification |
|---|---|---|
| 2026 | $30,000 | Initial pricing aligned with biologics |
| 2028 | $25,000–$28,000 | Pilot biosimilar competition begins |
| 2030 | $22,000–$26,000 | Market stabilization, biosimilar impact |
What Are Future Market Opportunities and Barriers?
Market opportunities include the unmet need for safer, more tolerable RA treatments. AUROVELA’s niche could be in patients intolerant to biologics or JAK inhibitors.
Barriers encompass regulatory challenges, clinical efficacy validation, and potential competition from emerging small-molecule drugs or biosimilars. Cost-effectiveness analyses and payer reimbursement policies will influence adoption.
Key Takeaways
- AUROVELA targets RA with a novel gold-based mechanism, positioned as a potentially safer alternative.
- The existing RA market exceeds $20 billion globally, dominated by biologics.
- Entry depends on successful clinical outcomes and regulatory approval timelines, expected around 2026-2028.
- Price projections hover between $22,000 and $40,000 annually based on efficacy, safety, and market competition.
- Biosimilar competition and market acceptance will influence long-term pricing strategy.
FAQs
1. When could AUROVELA realistically reach the market?
Regulatory approval could occur between 2026 and 2028, contingent on successful Phase 3 data.
2. How does AUROVELA compare in safety to current biologics?
Preliminary data suggest a potentially safer profile due to reduced immunogenicity, but definitive conclusions await clinical trial results.
3. What is the likelihood of AUROVELA’s market success?
Success depends on clinical efficacy demonstration, safety profile, regulatory approval, and market adoption strategies. These variables introduce significant uncertainty.
4. How will biosimilars impact AUROVELA’s pricing?
Biosimilar entry in the late 2020s may reduce prices by 20-30%, pressuring AUROVELA’s pricing and market share.
5. What are the main barriers to AUROVELA's commercialization?
Key barriers include clinical trial outcomes, regulatory approval delays, manufacturing scalability, and market competition.
References
- MarketsandMarkets. (2023). Rheumatoid arthritis therapeutics market.
- U.S. Food and Drug Administration. (2022). Guidelines for biologics and biosimilars.
- IQVIA. (2022). Rheumatoid arthritis market analysis report.
- European Medicines Agency. (2022). Regulatory pathway for RA drugs.
- Simoens, S., et al. (2018). The economic impact of biosimilar drugs. Pharmacoeconomics, 36(4), 415-423.
More… ↓
