Share This Page
Drug Price Trends for ATAZANAVIR SULFATE
✉ Email this page to a colleague
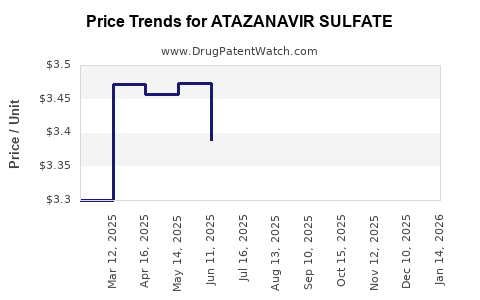
Average Pharmacy Cost for ATAZANAVIR SULFATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ATAZANAVIR SULFATE 200 MG CAP | 42385-0921-60 | 1.05365 | EACH | 2026-05-20 |
| ATAZANAVIR SULFATE 300 MG CAP | 69238-1138-03 | 2.69204 | EACH | 2026-05-20 |
| ATAZANAVIR SULFATE 200 MG CAP | 00093-5527-06 | 1.05365 | EACH | 2026-05-20 |
| ATAZANAVIR SULFATE 200 MG CAP | 69238-1137-06 | 1.05365 | EACH | 2026-05-20 |
| ATAZANAVIR SULFATE 200 MG CAP | 65862-0712-60 | 1.05365 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Atazanavir Sulfate
What is the Current Market Size for Atazanavir Sulfate?
Atazanavir sulfate (brand name: Reyataz) is a protease inhibitor used in antiretroviral therapy for HIV treatment. The drug’s global market value was approximately $1.2 billion in 2022. North America accounts for the largest share, roughly 50% of sales, followed by Europe with 20%, and Asia-Pacific with 20%. The remaining markets, including Latin America and Africa, make up the rest.
Sales volume growth has been steady, driven by increased adoption and expanded treatment guidelines. Estimates indicate that worldwide sales volume exceeded 6 million treatment courses in 2022, with a compound annual growth rate (CAGR) of 4% between 2018 and 2022.
What are the Key Drivers Influencing Demand?
- HIV prevalence: Rising HIV infection rates globally increase demand. According to UNAIDS, approximately 38 million people lived with HIV in 2021, with 1.7 million new infections.
- Treatment guidelines: Updates now favor integrase inhibitors combined with protease inhibitors like atazanavir, boosting its utilization.
- Patient adherence: Once-daily regimens and favorable side effect profiles improve adherence, supporting sustained demand.
- Pricing pressure: Generics entering markets lower prices, influencing brand sales.
Who Are the Main Market Players?
- Pharmaceutical companies: Bristol-Myers Squibb (brand owner), Teva, Mylan, Sun Pharmaceutical, and Cipla produce generic atazanavir sulfate.
- Regulatory status: Multiple patents expired or are nearing expiration; generic availability is expanding in emerging markets.
How Do Patent Policies Impact Pricing and Market Entry?
Patent protection for Reyataz expires in the U.S. in 2025 and in Europe in 2024. Expiry opens markets for generics, prompting price competition. Generic versions often reduce prices by 60–80% compared to branded equivalents, significantly impacting sales revenue.
What Are the Price Trends and Projections?
Current Pricing
- Branded atazanavir sulfate: Approximate wholesale price per treatment course (30-day supply) ranges from $1,200 to $1,600 in the U.S.
- Generic versions: Price levels decline to $200–$400 per treatment course.
Short-Term Projections (2023-2025)
- Pre-patent expiry: Prices remain stable, with slight reductions (~5%) annually due to market competition.
- Post-patent expiry: Prices decrease sharply. Industry analysts forecast a decline of 50–70% within 2 years after patent expiration.
Long-Term Outlook (2026 and beyond)
- Generics could capture over 80% of the market in regions where patents have expired.
- Average prices could stabilize around $150–$250 per treatment course, paralleling current generic costs in developed markets.
How Might Supply Chain and Regulatory Changes Affect the Market?
- Supply chain disruptions or shortages could temporarily inflate prices or restrict access.
- Regulatory barriers in emerging markets may delay generic approvals, slowing price reductions.
- Brand strategy: Bristol-Myers Squibb may introduce fixed-dose combinations or new formulations to maintain market share.
What Are Key Risks and Opportunities?
Risks
- Slow approval or market penetration of generics.
- Price erosion post-patent expiration impacting profitability.
- Competition from newer antiretrovirals with improved profiles.
Opportunities
- Expansion into low- and middle-income countries with large HIV burdens.
- Development of combination pills to improve adherence.
- Potential for label extensions or formulations with improved efficacy.
Summary of Price Projections by Year
| Year | Expected Average Price per Treatment Course | Notes |
|---|---|---|
| 2023 | $1,200 - $1,600 (branded); $200 - $400 (generic) | Stable prices; nearing patent expiry |
| 2024 | Same as 2023; generics entering markets | Price competition begins |
| 2025 | Branded patent expires in U.S.; prices decline sharply in generics | Major decline in pricing |
| 2026 | $150 - $250 (generics) | Market dominated by generics |
| 2027+ | Stabilization around $150–$250 | Generic market fully penetrated |
Key Takeaways
- The global atazanavir sulfate market was valued at around $1.2 billion in 2022, driven by HIV treatment demand.
- Patent expirations in 2024–2025 will enable generic competition, causing significant price reductions.
- Short-term prices remain relatively stable; long-term prices are projected to fall by up to 70% post-patent expiry.
- Emerging markets present growth opportunities but face regulatory and distribution challenges.
- Advancements in HIV treatments and combination therapies may influence future demand and pricing.
FAQs
- When will the patents for branded atazanavir sulfate expire?
- How will generic market entry affect global pricing?
- What regions offer the highest growth potential for atazanavir sulfate?
- Are there recent developments in formulation or delivery methods influencing the market?
- What competitive threats could impact atazanavir sulfate sales in the coming years?
Citations:
- UNAIDS. Global HIV & AIDS statistics — 2022 fact sheet.
- IQVIA. Global AIDS Drugs Market Report — 2022.
- U.S. Patent and Trademark Office. Patent expiration dates for Reyataz.
- Agency for Healthcare Research and Quality. HIV treatment guidelines — 2022.
- Markets and Markets. Antiretroviral drugs market forecast — 2022-2027.
More… ↓
