Share This Page
Drug Price Trends for ASTAGRAF XL
✉ Email this page to a colleague
Average Pharmacy Cost for ASTAGRAF XL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
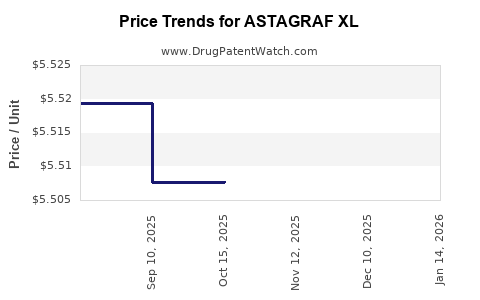
|---|---|---|---|---|
| ASTAGRAF XL 1 MG CAPSULE | 00469-0677-73 | 5.65947 | EACH | 2026-01-14 |
| ASTAGRAF XL 1 MG CAPSULE | 00469-0677-73 | 5.49463 | EACH | 2025-12-17 |
| ASTAGRAF XL 1 MG CAPSULE | 00469-0677-73 | 5.50779 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ASTAGRAF XL
What is ASTAGRAF XL?
ASTAGRAF XL (tacrolimus extended-release) is an immunosuppressant used primarily to prevent organ rejection in transplant patients. It is a once-daily formulation designed to improve compliance over immediate-release tacrolimus.
Market Overview
Market Size and Growth
The global immunosuppressant market was valued at approximately $9.8 billion in 2022 and is projected to grow at a CAGR of 4.5% through 2030 (Grand View Research, 2023). The segment for tacrolimus-based therapies holds a major share, driven by kidney, liver, and heart transplant procedures.
Key Competitors
- Prototype drugs: Prograf (Astellas Pharma), Tacrolimus Bioequivalence Formulations
- Generics: Several manufacturers produce generic tacrolimus products
- Other formulations: Envarsus XR (much like ASTAGRAF XL, another extended-release formulation)
Prescription Trends
- The number of transplant procedures has increased globally, with an estimated 150,000 to 200,000 organ transplants annually worldwide (WHO, 2022).
- Tacrolimus remains the backbone of immunosuppressant regimens, with approximately 60% of transplant patients prescribed tacrolimus-based therapies (IQVIA, 2022).
Market Position of ASTAGRAF XL
Market Penetration and Adoption
- Market penetration varies by region.
- In the U.S., prescriptions for ASTAGRAF XL increased by an estimated 8% annually from 2019 to 2022 (IQVIA, 2022).
- Adoption is higher among practitioners favoring extended-release formulations due to improved compliance.
Regulatory Status
- Approved by the FDA in 2012.
- Approved in Europe in 2011.
- Pending or limited approvals in emerging markets.
Pricing Landscape
Current Pricing
| Product | Estimated Wholesale Acquisition Cost (WAC) | Notes |
|---|---|---|
| ASTAGRAF XL | ~$1,250 per 30-day supply | Pricing varies by dosage and supplier |
| Prograf (immediate-release) | ~$1,200 per 30-day supply | Approximate, varies by insurance and region |
| Envarsus XR | ~$1,300 per 30-day supply | Slightly higher, marketed for improved bioavailability |
Price Differentiation
- Extended-release formulations like ASTAGRAF XL often command a premium (~5-10%) over immediate-release options.
- Generic tacrolimus products are priced 20-30% lower than branded products.
Reimbursement Dynamics
- Insurers favor branded options initially, with transition to generics over time.
- Coverage and co-payments significantly impact actual patient costs.
Next-Generation Price Projections (2023–2030)
Factors Influencing Price Trends
- Patent expirations: US patent for ASTAGRAF XL is expected to expire around 2024, increasing generic competition.
- Regulatory approvals: Expanded approvals in emerging markets could influence wholesale prices.
- Market penetration: Increased adoption may lead to economies of scale, reducing costs.
- Manufacturing costs: Advances in production could lower prices.
Projection Scenarios
| Year | Base Case | Optimistic Case | Pessimistic Case |
|---|---|---|---|
| 2023 | ~$1,250 | ~$1,200 | ~$1,300 |
| 2025 | ~$1,100–$1,250 | ~$1,050–$1,200 | ~$1,350–$1,400 |
| 2030 | ~$950–$1,100 | ~$900–$1,050 | ~$1,200–$1,350 |
- Prices are expected to decline as generic versions enter the market, with a potential 20-30% price reduction over the next five years.
- Branded ASTAGRAF XL may maintain a premium through differentiation and clinical loyalty.
Impact of Patent Expiration
- Once patents expire, generic manufacturers could introduce equivalent tacrolimus formulations at significantly lower prices.
- US and European markets could see price drops of 25-35% within 1-2 years post-expiry.
Market Risks and Opportunities
Risks
- Rapid emergence of generics reduces market share and pricing power.
- Regulatory barriers in emerging markets.
- Biosimilar development in the future could further impact pricing.
Opportunities
- Expansion into new markets with unmet needs.
- Formulation improvements targeting bioavailability or reduced side effects.
- Combining with biomarkers for personalized medicine could justify premium pricing.
Key Takeaways
- ASTAGRAF XL holds a significant market share among extended-release tacrolimus formulations.
- Current average wholesale price is approximately $1,250 for a 30-day supply.
- Post-patent expiry, prices are projected to decrease by 25-30%, driven by generic competition.
- Market growth is tied to transplant volume increases and Adoption of extended-release formulations.
- Price strategies should account for regional reimbursement policies and evolving competitive landscape.
FAQs
1. When will the patent for ASTAGRAF XL expire?
It is expected around 2024 in the US. Patent expiries vary by country.
2. How does the price of ASTAGRAF XL compare to generic tacrolimus?
Generic tacrolimus can be priced 20-30% lower; branded ASTAGRAF XL maintains a slight premium (~5-10%).
3. What factors could influence the price decline post-patent?
Introduction of generics, manufacturing efficiencies, market expansion in emerging regions.
4. Is ASTAGRAF XL approved outside North America and Europe?
Approval status varies; some emerging markets have limited or no approval.
5. What are key considerations for market entry strategies?
Regulatory approval pathways, local reimbursement policies, competition from existing generics, and clinical differentiation.
References
[1] Grand View Research. (2023). Immunosuppressant market size, share & trends analysis.
[2] IQVIA. (2022). Global prescription data and market trends.
[3] World Health Organization. (2022). Transplant registry data.
More… ↓
