Share This Page
Drug Price Trends for ASPIRIN EC
✉ Email this page to a colleague
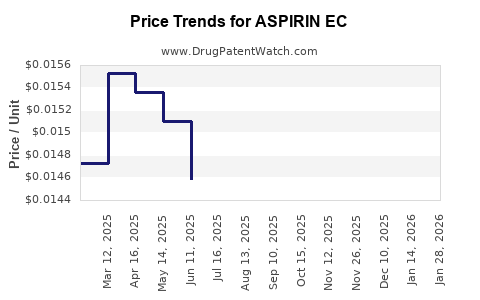
Average Pharmacy Cost for ASPIRIN EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ASPIRIN EC 81 MG TABLET | 82568-0003-01 | 0.01466 | EACH | 2026-04-29 |
| ASPIRIN EC 81 MG TABLET | 82568-0003-02 | 0.01466 | EACH | 2026-04-29 |
| ASPIRIN EC 325 MG TABLET | 00536-1232-01 | 0.02268 | EACH | 2026-04-22 |
| ASPIRIN EC 325 MG TABLET | 46122-0596-02 | 0.02268 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Aspirin EC: Market Analysis and Price Projections
What is Aspirin EC and its Current Market Position?
Aspirin EC, or enteric-coated aspirin, is a modified-release formulation of acetylsalicylic acid designed to dissolve in the small intestine rather than the stomach. This coating protects the stomach lining from the corrosive effects of aspirin, reducing the incidence of gastrointestinal side effects such as ulcers and bleeding.
The primary indications for Aspirin EC are pain relief, fever reduction, and anti-inflammatory effects, similar to standard aspirin. However, its protected-release mechanism makes it a preferred choice for long-term, low-dose use in cardiovascular prevention, including secondary prevention of myocardial infarction and stroke. This application accounts for a significant portion of the global Aspirin EC market.
Globally, the market for Aspirin EC is mature, characterized by widespread availability of generic formulations. The total market size for aspirin, including all forms, was estimated to be approximately $2.6 billion in 2022 [1]. While specific market data for the enteric-coated segment alone is not granularly reported by all market research firms, it represents a substantial sub-segment within the broader aspirin market, driven by its established efficacy and safety profile for chronic use.
Key market players include major pharmaceutical manufacturers and numerous generic producers. The competitive landscape is intense, with price being a significant differentiating factor for over-the-counter (OTC) products. Prescription-level sales are also present for specific cardiovascular indications.
What are the Key Drivers and Restraints in the Aspirin EC Market?
Drivers:
- Cardiovascular Prevention Demand: The established role of low-dose aspirin in preventing cardiovascular events remains a primary market driver. Guidelines from organizations like the American Heart Association and the European Society of Cardiology continue to recommend aspirin for specific patient populations [2].
- Aging Global Population: The increasing prevalence of cardiovascular diseases and age-related pain conditions in aging demographics globally supports sustained demand for effective analgesics and prophylactic cardiovascular treatments like Aspirin EC.
- Gastrointestinal Tolerance: The enteric coating's ability to mitigate stomach irritation is crucial for patient adherence in chronic treatment regimens, driving preference over standard aspirin for long-term use.
- OTC Accessibility: Aspirin EC is widely available as an over-the-counter medication in most countries, facilitating broad consumer access and market penetration for self-medication of minor ailments and preventive care.
- Cost-Effectiveness: Compared to newer therapeutic agents for cardiovascular protection or pain management, Aspirin EC remains a highly cost-effective option, making it attractive for healthcare systems and individuals with budget constraints.
Restraints:
- Emergence of Novel Therapeutics: In cardiovascular medicine, newer anticoagulant and antiplatelet agents, while often more expensive, offer alternative or adjunctive therapies that may limit the market share growth for aspirin in certain high-risk patient groups.
- Bleeding Risk Concerns: Despite the enteric coating, aspirin therapy is associated with an increased risk of bleeding, particularly gastrointestinal bleeding. This inherent risk, coupled with growing awareness of its side effects, can lead to physician caution in prescribing and patient reluctance for certain individuals.
- Generic Competition and Price Erosion: The market is heavily dominated by generic manufacturers, leading to significant price competition and downward pressure on profit margins for both branded and generic products.
- Evolving Clinical Guidelines: Periodic updates to clinical guidelines, particularly regarding the initiation or discontinuation of aspirin therapy in primary cardiovascular prevention, can impact overall demand. For instance, recent guidelines have refined recommendations for primary prevention [3].
- Perception of Older Drug: In an era of rapid pharmaceutical innovation, aspirin, despite its proven efficacy, may be perceived by some as an older, less advanced treatment option, potentially impacting its adoption in certain patient segments.
What is the Competitive Landscape for Aspirin EC?
The competitive landscape for Aspirin EC is fragmented and highly competitive, driven by the drug's long history and patent expirations.
Major Global Players:
- Bayer AG: Holds a prominent position with its flagship brand, Aspirin. Bayer actively markets various Aspirin formulations, including enteric-coated versions, globally. Their focus often includes brand building and marketing for specific indications.
- Johnson & Johnson: Through its subsidiary McNeil Consumer Healthcare, Johnson & Johnson offers Tylenol and other pain relief products. While Tylenol is acetaminophen-based, J&J also has a presence in the broader analgesic market and may offer aspirin products in certain regions.
- GSK (GlaxoSmithKline): Offers Excedrin, a combination product that often includes aspirin, and may have separate aspirin formulations.
- Generic Manufacturers: A vast number of generic pharmaceutical companies worldwide produce Aspirin EC. These include large multinational corporations and regional players such as:
- **Te
- va Pharmaceuticals Industries Ltd.**
- Mylan N.V. (now part of Viatris)
- Hikma Pharmaceuticals PLC
- Sun Pharmaceutical Industries Ltd.
- Zydus Lifesciences Ltd. (formerly Cadila Healthcare)
Key Competitive Factors:
- Price: For OTC Aspirin EC, price is a paramount factor. Generic products compete aggressively on cost.
- Distribution Channels: Extensive retail pharmacy presence, supermarket availability, and online sales platforms are critical.
- Brand Recognition: For branded Aspirin EC, established brand equity and consumer trust play a role, particularly in developed markets.
- Product Quality and Formulation: Consistency in enteric coating efficacy and tablet integrity is important for patient safety and efficacy.
- Regulatory Approvals: Compliance with pharmaceutical regulations in target markets.
The market is characterized by high sales volumes and low profit margins per unit due to intense generic competition. The focus for many players is on optimizing manufacturing costs and achieving economies of scale.
What are the Price Trends and Projections for Aspirin EC?
Current Price Trends:
The pricing of Aspirin EC is largely bifurcated:
- Over-the-Counter (OTC) Products: These are subject to significant retail competition. Prices for standard bottle sizes (e.g., 100-200 count) of generic Aspirin EC typically range from $5 to $15 USD, depending on the brand, strength, and retailer. Branded Aspirin EC, like Bayer Aspirin, commands a premium, often ranging from $10 to $25 USD for similar quantities.
- Prescription Products: When prescribed by a physician, particularly for low-dose cardiovascular prevention (e.g., 81 mg or 100 mg), the cost to the patient or insurance payer is also influenced by pharmacy markups and insurance formularies. Prices can vary but generally fall within a similar range to OTC products on a per-tablet basis, especially for generic versions.
Factors Influencing Pricing:
- Active Pharmaceutical Ingredient (API) Costs: The cost of sourcing acetylsalicylic acid and enteric-coating materials.
- Manufacturing and Packaging Costs: Economies of scale and production efficiency are key.
- Distribution and Retail Markups: Significant factors in the final consumer price.
- Competition: The number of generic manufacturers and their pricing strategies directly impact market prices.
- Regulatory Compliance: Costs associated with maintaining quality standards and regulatory approvals.
- Brand Premium: Established brands can command higher prices due to marketing and perceived quality.
Price Projections:
The Aspirin EC market is expected to experience stable to slightly declining price trends in real terms over the next five years (2024-2029).
- Generic Aspirin EC: Prices are projected to remain highly competitive. Significant price increases are unlikely due to the mature market and ongoing generic competition. Slight downward pressure may persist as manufacturers optimize production and new generic entrants emerge in specific regional markets. Inflationary pressures on raw materials and manufacturing could offset some of these declines, leading to overall price stability.
- Branded Aspirin EC: While branded products will likely maintain a price premium over generics, this premium is expected to be challenged. Manufacturers may engage in targeted marketing to justify higher prices based on formulation quality or specific patient benefits, but significant price hikes are improbable given the availability of low-cost generic alternatives.
Projected Average Price Ranges (USD per 100 tablets, 81mg/325mg):
| Product Type | Current Range (2023) | Projected Range (2027) | Projected Change |
|---|---|---|---|
| Generic Aspirin EC | $5 - $15 | $5 - $14 | -2% to -5% |
| Branded Aspirin EC | $10 - $25 | $10 - $23 | -2% to -5% |
These projections assume no major disruptions in API supply chains, continued regulatory stability, and no significant shifts in cardiovascular prevention guidelines that would drastically alter demand for aspirin therapy. The increasing global focus on healthcare cost containment also reinforces the expectation of price stabilization or decline.
What are the Regulatory Considerations for Aspirin EC?
The regulatory landscape for Aspirin EC is governed by national and regional health authorities. Key considerations include:
- Drug Approval and Marketing Authorization: Manufacturers must obtain marketing authorization from regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and similar agencies in other countries. This involves submitting comprehensive dossiers detailing manufacturing processes, quality control, non-clinical and clinical data demonstrating safety and efficacy.
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to strict GMP guidelines to ensure product quality, purity, and consistency. Regular inspections by regulatory authorities are common.
- Labeling and Packaging Requirements: Regulations dictate the information that must be included on product labels and packaging, including active ingredient, strength, dosage instructions, warnings, contraindications, and expiration dates. For OTC products, clear and concise labeling for consumer understanding is paramount.
- Enteric Coating Standards: While not always a separate regulatory category, the efficacy and integrity of the enteric coating are critical to the drug's safety and performance. Regulators expect data demonstrating that the coating reliably delays dissolution until the small intestine. This may involve in vitro dissolution testing according to pharmacopoeial standards (e.g., USP, EP).
- Post-Marketing Surveillance: Manufacturers are obligated to monitor and report adverse events and product quality issues that arise after a drug is on the market.
- Generic Drug Equivalence: Generic Aspirin EC products must demonstrate bioequivalence to the reference listed drug (RLD) or the innovator product. This typically involves studies showing comparable rates and extent of absorption.
- Advertising and Promotion: Regulations govern the advertising and promotional activities for both OTC and prescription drugs, preventing misleading claims and ensuring that marketing is consistent with approved labeling.
Specific Regulatory Examples:
- FDA (USA): Aspirin is classified as an OTC drug, with specific monographs outlining requirements for its formulation and labeling. Low-dose aspirin for cardiovascular prevention may also be available as a prescription product, requiring a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics.
- EMA (Europe): Aspirin EC products undergo a centralized or national authorization procedure. The EMA provides guidelines on product quality, safety, and efficacy.
- Pharmacopoeial Standards: Aspirin EC formulations must generally comply with the monographs for Acetylsalicylic Acid Tablets or Enteric-Coated Tablets in major pharmacopoeias like the United States Pharmacopeia (USP), European Pharmacopoeia (EP), and Japanese Pharmacopoeia (JP). These monographs define physical and chemical characteristics, dissolution criteria, and impurity limits.
Key Takeaways
Aspirin EC is a mature pharmaceutical product with stable demand driven by its established role in cardiovascular prevention and its gastrointestinal safety profile compared to standard aspirin. The market is highly competitive, dominated by generic manufacturers, leading to price erosion and low profit margins. While branded products retain a premium, significant price increases are unlikely. Regulatory compliance, particularly regarding GMP and enteric coating efficacy, is critical for market access. Future price trends are projected to be stable to slightly declining in real terms, influenced by cost-effective manufacturing and ongoing generic competition.
Frequently Asked Questions
-
What is the typical lifespan of a patent for Aspirin EC? Aspirin itself is a well-established drug with expired primary patents. However, patents can be obtained for new formulations, methods of delivery, or specific therapeutic uses. For an enteric-coated formulation, a patent might cover the specific coating technology or a unique manufacturing process, typically lasting 20 years from the filing date, though market exclusivity can be extended through various regulatory pathways.
-
How does the enteric coating affect the cost of Aspirin EC production compared to standard aspirin? The enteric coating process adds complexity and cost to manufacturing. It requires specialized equipment, additional coating materials (polymers, plasticizers), and longer processing times, increasing per-unit production costs by approximately 10-25% compared to uncoated aspirin tablets.
-
Are there specific patient populations where Aspirin EC is contraindicated despite its coating? Yes, Aspirin EC is contraindicated in patients with known hypersensitivity to salicylates or NSAIDs, active peptic ulcers, a history of gastrointestinal bleeding, bleeding disorders (e.g., hemophilia), severe kidney or liver disease, and in the third trimester of pregnancy. The enteric coating mitigates but does not eliminate the risk of gastrointestinal bleeding in susceptible individuals.
-
What is the projected impact of new anticoagulant therapies on the Aspirin EC market for cardiovascular prevention? Newer direct oral anticoagulants (DOACs) and more potent antiplatelet agents are increasingly used, particularly in high-risk cardiovascular patients. This may lead to a gradual substitution of aspirin in some primary and secondary prevention scenarios, potentially leading to a modest decline in the volume growth of Aspirin EC for these specific indications. However, Aspirin EC is expected to remain a standard of care for many patients due to its established efficacy and cost-effectiveness.
-
Can Aspirin EC be used for acute pain relief, or is it primarily for chronic use? Aspirin EC can be used for acute pain relief, fever, and inflammation. However, due to the delayed onset of action caused by the enteric coating (which needs to dissolve in the intestine), it is less preferred for immediate pain relief compared to standard aspirin or acetaminophen. Its primary advantage and market driver for chronic use are the reduced gastrointestinal side effects.
Citations
[1] Grand View Research. (2023). Aspirin Market Size, Share & Trends Analysis Report By Product Type (Aspirin Tablets, Aspirin Capsules, Aspirin Powder), By Application (Pain Relief, Cardiovascular Disease Prevention, Anti-inflammatory), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online), By Region, And Segment Forecasts, 2023 - 2030. (Accessed via market research databases).
[2] He, J., Fillit, H. M., & Saeed, R. (2018). Aspirin in Primary Prevention of Cardiovascular Disease: A Review of Recent Guidelines and Evidence. The American Journal of Medicine, 131(12), 1472-1479. doi:10.1016/j.amjmed.2018.07.034
[3] U.S. Preventive Services Task Force. (2022). Aspirin Use for the Primary Prevention of Cardiovascular Disease: U.S. Preventive Services Task Force Recommendation Statement. U.S. Preventive Services Task Force.
More… ↓
