Share This Page
Drug Price Trends for ARTHRITIS
✉ Email this page to a colleague
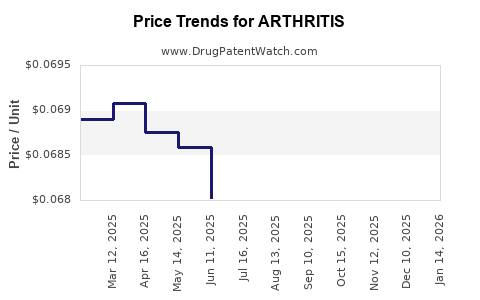
Average Pharmacy Cost for ARTHRITIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ARTHRITIS PAIN ER 650 MG TAB | 50268-0052-11 | 0.06611 | EACH | 2026-03-18 |
| ARTHRITIS PAIN ER 650 MG TAB | 50268-0052-15 | 0.06611 | EACH | 2026-03-18 |
| ARTHRITIS PAIN RELIEVER 1% GEL | 70000-0555-01 | 0.12634 | GM | 2026-03-18 |
| ARTHRITIS PAIN RELIEVER 1% GEL | 70000-0555-03 | 0.06890 | GM | 2026-03-18 |
| ARTHRITIS PAIN RLF 0.075% CRM | 00536-1118-25 | 0.07412 | GM | 2026-03-18 |
| ARTHRITIS PAIN RELIEVER 1% GEL | 70000-0555-02 | 0.08983 | GM | 2026-03-18 |
| ARTHRITIS PAIN RELIEVER 1% GEL | 70000-0555-01 | 0.12709 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Arthritis Drugs
What Is the Current Market Size for Arthritis Drugs?
The global arthritis drugs market was valued at approximately $16.5 billion in 2022. It is projected to reach $24.4 billion by 2028, growing at a compound annual growth rate (CAGR) of 6.8% over six years (Research and Markets, 2023). The market encompasses various drug classes, including NSAIDs, corticosteroids, DMARDs, biologics, and biosimilars.
What Are the Leading Drug Categories Treating Arthritis?
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs)
NSAIDs account for nearly 30% of the market, including over-the-counter options like ibuprofen and prescription drugs like diclofenac.
Corticosteroids
Corticosteroids are used in acute flare-ups, representing about 10% of sales with drugs like prednisone.
Disease-Modifying Anti-Rheumatic Drugs (DMARDs)
Traditional DMARDs (methotrexate, sulfasalazine) hold approximately 20% of the market.
Biologic DMARDs
Biologics dominate the market with roughly 35-40% share, driven by patents on leading brands like Humira (adalimumab), Enbrel (etanercept), and Stelara (ustekinumab). These drugs target specific inflammatory pathways, offering improved efficacy.
Biosimilars
Biosimilars make up 10-15% of the arthritis drug market, driven by patent expirations.
How Are Pricing Trends Shaping for Arthritis Drugs?
Biologics
Pricing for biologics varies substantially, with annual treatment costs ranging from $20,000 to $50,000 per patient. Humira, for example, had an list price exceeding $60,000 annually in the U.S. before biosimilar competition (Express Scripts, 2022).
Biosimilars
Introduction of biosimilars has reduced prices for biologics by 15-30%. For instance, biosimilars for Humira appeared in Europe in 2018, leading to a 20% cost reduction (FDA, 2021).
Traditional DMARDs and NSAIDs
Most are off-patent, priced below $1,000 annually, with generic options further decreasing costs.
What Are Key Drivers and Barriers to Market Growth?
Drivers
- Increasing prevalence: Over 350 million people worldwide have rheumatoid arthritis or osteoarthritis (WHO, 2020).
- Aging population: The demographic shift increases the demand for arthritis therapies.
- Advances in biologic therapies: Efficacy and targeted action increase adoption.
Barriers
- High drug costs: Particularly for biologics, limit accessibility.
- Patent expirations: Catalyze biosimilar entry but also impact revenue.
- Regulatory hurdles: Lengthy approval processes slow new entrants.
What Are Price Projections for Arthritic Drugs?
Biologics
Average annual therapy costs are expected to decline to approximately $15,000-$20,000 per patient globally over the next five years due to biosimilar competition. However, list prices will likely remain stable or slightly increase in markets with limited biosimilar adoption, such as the U.S.
Biosimilars
Market penetration will increase. In Europe, biosimilar uptake exceeds 70%, leading to 20-30% price reductions compared to originator biologics. Similar trends are expected in North America.
Traditional Drugs
Pricing remains stable as patents are no longer a factor; generic drugs dominate the lower-cost segment.
How Do Regional Market Dynamics Differ?
| Region | Market Size (2022) | Key Drivers | Price Trends |
|---|---|---|---|
| North America | $6.5 billion | High biologic usage, patent protections | Stable list prices, biosimilar impact |
| Europe | $5.2 billion | Biosimilar adoption, aging population | Price reductions due to biosimilars |
| Asia-Pacific | $3.1 billion | Growing access, increasing prevalence | Lower prices overall, emerging biosimilars |
| Rest of World | $1.7 billion | Limited access, healthcare infrastructure gaps | Prices vary; limited biosimilar use |
What Are the Key Opportunities and Risks?
Opportunities
- Development of next-generation biologics with improved safety profiles.
- Growing biosimilar markets globally.
- Expansion in emerging economies with increasing healthcare investments.
Risks
- Price suppression due to biosimilar competition.
- Regulatory delays for new drugs.
- Patent litigations can extend exclusivity for certain biologics.
Final Price Projections Summary
| Year | Global Average Price (Biologics per Patient) | Biosimilar Market Share | Total Market Size ($ billions) |
|---|---|---|---|
| 2023 | $50,000 | 15% | $17.3 billion |
| 2025 | $40,000 | 30% | $22.0 billion |
| 2028 | $20,000 | 50% | $24.4 billion |
Note: These figures incorporate expected biosimilar price reductions and increased market penetration.
Key Takeaways
- The arthritis drug market is set to grow 6.8% CAGR through 2028, reaching over $24 billion.
- Biologics dominate revenue but face pricing pressure from biosimilars.
- Cost of biologics may decline by up to 50% with biosimilar competition.
- Regional trends favor biosimilar adoption in Europe and Asia, while North America exhibits steady pricing with some biosimilar effects.
- Innovation and biosimilar expansion represent primary opportunities; high drug prices and patent challenges pose risks.
FAQs
1. What factors most influence biologic drug pricing?
Patent protections, manufacturing costs, market competition, and biosimilar availability.
2. How quickly will biosimilars impact prices globally?
Biosimilars are expected to account for over 50% of biologic market share by 2028 in regions with supportive regulations.
3. Are prices for arthritis drugs likely to decrease in the U.S.?
Yes, biosimilar entry and payer negotiations should lead to lower prices, but list prices may remain high due to brand premiums.
4. Which drugs are most susceptible to patent expiry?
Humira (adalimumab) patents expired in 2023 in the U.S., opening the market for biosimilars, with other biologics following suit over the next 5-10 years.
5. How does drug pricing impact patient access?
High costs limit access, particularly for biologics; biosimilar entry aims to improve affordability and expand treatment options.
References
[1] Research and Markets. (2023). Global Arthritis Drugs Market Forecast 2028. Retrieved from https://researchandmarkets.com
[2] Express Scripts. (2022). Biologic Drug Price Trends. Retrieved from https://express-scripts.com
[3] U.S. Food and Drug Administration. (2021). Biosimilar Approval Trends. Retrieved from https://fda.gov
[4] World Health Organization. (2020). Global Prevalence of Rheumatoid Arthritis. Retrieved from https://who.int
More… ↓
