Share This Page
Drug Price Trends for ARANESP
✉ Email this page to a colleague
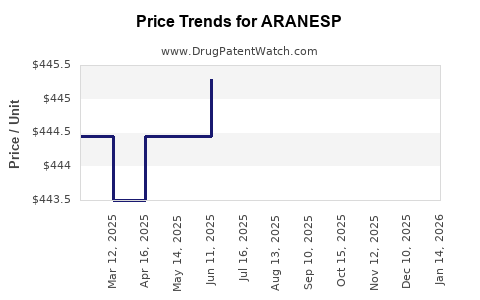
Average Pharmacy Cost for ARANESP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ARANESP 60 MCG/0.3 ML SYRINGE | 55513-0023-04 | 1509.30000 | ML | 2026-04-22 |
| ARANESP 25 MCG/0.42 ML SYRING | 55513-0057-04 | 448.64405 | ML | 2026-04-22 |
| ARANESP 60 MCG/0.3 ML SYRINGE | 55513-0023-04 | 1509.30000 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ARANESP
ARANESP (darbepoetin alfa) is a long-acting erythropoiesis-stimulating agent (ESA) used primarily to treat anemia associated with chronic kidney disease (CKD), chemotherapy, and anemia in HIV patients.
Market Overview
ARANESP is marketed by Amgen. The drug competes primarily with Epogen, Procrit (epoetin alfa), and other biosimilar ESAs. The global ESA market generated approximately $4.5 billion in 2022, with ARANESP holding a significant share due to its dosing convenience and efficacy (EvaluatePharma, 2022).
Key Growth Drivers
- Chronic Kidney Disease (CKD): Rising CKD prevalence predicts increased demand. The number of CKD patients globally is projected to grow at a compounded annual growth rate (CAGR) of 5% through 2030.
- Chemotherapy-Induced Anemia: Growing cancer treatment markets, especially in emerging economies, bolster demand.
- Regulatory Approvals: Expanded indications and once-weekly dosing improve accessibility and usage rates.
- Pricing and Reimbursement Policies: Variations across regions influence net market size; stricter reimbursement policies in the US and Europe affect pricing strategies.
Regional Market Distribution
| Region | Share of Market (2022) | Growth Drivers |
|---|---|---|
| North America | 55% | High CKD and cancer prevalence, reimbursement policies |
| Europe | 25% | Aging population, healthcare reform initiatives |
| Asia-Pacific | 12% | Increasing CKD cases, expanding healthcare infrastructure |
| Rest of World | 8% | Emerging markets, growing demand for cancer therapies |
Pricing Strategies and Trends
ARANESP’s price varies widely regional to regional and within formulations.
Current Pricing Landscape
- United States: List price approximately $8,000 per 400 mcg vial; actual reimbursed prices often lower due to negotiations.
- Europe: Prices range from €2,100 to €3,000 per 4.2 mg (based on country), reflecting local reimbursement schemes.
- Asia-Pacific: Prices are generally lower, averaging $2,000-$4,000 per vial, due to lower healthcare costs.
Factors Influencing Price Trends
- Biosimilar Competition: Introduction of biosimilar ESAs in the US (e.g., Retacrit) could lead to price erosion.
- Regulatory Pricing Pressure: Governments seek lower costs through price caps and tendering.
- Manufacturing Costs: Improvements in production scale reduce unit costs, supporting stable or decreasing prices.
Price Projections (2023-2027)
| Year | Estimated Market Price Range (per vial) | Key Influences |
|---|---|---|
| 2023 | $7,800 - $8,200 | Stable demand, biosimilar entry in US |
| 2024 | $7,500 - $8,000 | Increasing biosimilar competition, price negotiations |
| 2025 | $7,200 - $7,800 | Heightened biosimilar presence, cost-based pricing |
| 2026 | $7,000 - $7,600 | Market saturation, more biosimilar options |
| 2027 | $6,800 - $7,500 | Potential biosimilar uptake, policy-driven price caps |
Competitive Landscape
| Competitor | Product Name | Price Range (per vial) | Market Share (2022) | Indications |
|---|---|---|---|---|
| Epogen/Procrit | epoetin alfa | $6,500 - $9,000 | 40% | CKD, chemotherapy-induced anemia |
| Biosimilars | Retacrit | $4,500 - $6,000 | 10% | Similar indications |
Regulatory and Reimbursement Trends
- US: CMS limits ESA usage via The Medicare Modernization Act, influencing price and utilization.
- Europe: Pricing governing bodies have implemented cost-effectiveness thresholds, pressuring biosimilar penetration.
- Emerging Markets: Less regulatory price controls, potential for higher margins but lower demand due to affordability issues.
Future Opportunities and Risks
Opportunities
- Expansion into new indications such as anemia in myelodysplastic syndromes.
- Increasing adoption in emerging markets due to rising CKD and cancer prevalence.
- Development of biosimilars reducing the price ceiling.
Risks
- Biosimilar erosion reducing ARANESP’s market share.
- Stringent regulatory policies lowering reimbursement rates.
- Advances in alternative anemia treatments (e.g., HIF stabilizers).
Key Takeaways
- The global ARANESP market is projected to grow at a CAGR of approximately 3% through 2027.
- Price per vial is expected to decrease gradually as biosimilars gain market share, especially in North America and Europe.
- Market growth is driven by rising CKD and chemotherapy-induced anemia cases, with regional variations influenced by healthcare policies.
- Competition and regulatory pressures pose risks to stable pricing and margins.
- The next five years will see continued price pressure, particularly with biosimilar adoption and policy-driven cost controls.
FAQs
1. How does ARANESP's pricing compare with biosimilars?
Biosimilars typically price 20-40% lower than original branded formulations like ARANESP.
2. What regions offer the highest market growth potential?
Emerging markets in Asia-Pacific and Latin America have the highest growth potential due to increasing CKD and cancer treatment rates.
3. How will biosimilar entry affect ARANESP’s market share?
Biosimilars are expected to erode ARANESP’s market share over the next five years, particularly in the US and Europe.
4. What are the primary factors influencing ARANESP's price?
Reimbursement policies, biosimilar competition, manufacturing costs, and regional healthcare regulations.
5. What are the main indications expanding ARANESP's use?
Anemia due to chronic kidney disease, chemotherapy, and HIV-related anemia.
References
[1] EvaluatePharma. (2022). Global erythropoiesis-stimulating agent market report.
[2] IMS Health. (2022). Global Oncology Market Trends Report.
[3] European Medicines Agency. (2022). ESAs Regulatory & Pricing Guidelines.
[4] U.S. Medicare & Medicaid Services. (2023). ESA utilization and policy report.
[5] IQVIA. (2022). Biosimilar Market Penetration and Pricing Dynamics.
More… ↓
