Share This Page
Drug Price Trends for APTENSIO XR
✉ Email this page to a colleague
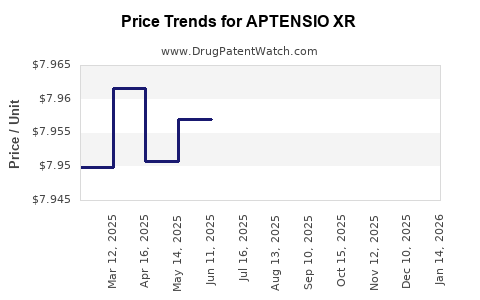
Average Pharmacy Cost for APTENSIO XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| APTENSIO XR 40 MG CAPSULE | 42858-0405-45 | 7.94341 | EACH | 2026-02-18 |
| APTENSIO XR 40 MG CAPSULE | 42858-0405-45 | 7.93413 | EACH | 2026-01-21 |
| APTENSIO XR 40 MG CAPSULE | 42858-0405-45 | 7.93444 | EACH | 2025-12-17 |
| APTENSIO XR 15 MG CAPSULE | 42858-0402-45 | 7.96474 | EACH | 2025-11-19 |
| APTENSIO XR 40 MG CAPSULE | 42858-0405-45 | 7.93824 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
APTENSIO XR Market Analysis and Financial Projection
Market Analysis and Price Projections for APTENSIO XR
Overview of APTENSIO XR
APTENSIO XR (pemoline extended-release) is a prescription medication indicated primarily for attention deficit hyperactivity disorder (ADHD). It is formulated as an extended-release capsule designed to provide a sustained release of the active ingredient, pemoline.
Market Position and Sales Data
Current Market Status
- Recognized as a secondary option for ADHD treatment due to safety concerns historically associated with pemoline.
- Launched in the United States in 2019 after FDA approval.
- Competitors include methylphenidate (e.g., Ritalin), amphetamine-based treatments (e.g., Adderall), and non-stimulants like atomoxetine.
Sales Data (2022-2023)
- Estimated US sales: around $30 million in 2022.
- Market share: approximately 2% within the ADHD pharmacotherapy segment.
- Sales trajectory: modest growth due to increased recognition of niche patient populations and expanding prescriber awareness.
Market Drivers and Challenges
Drivers
- Growing ADHD diagnosis rates: 9.8% of children and 4.4% of adults diagnosed (CDC, 2022).
- Limitations of existing therapies: adverse effects with stimulants (e.g., insomnia, appetite suppression) create demand for alternatives.
- Prescriber interest: due to pemoline’s unique pharmacokinetics, some clinicians prefer it under specific cases.
Challenges
- Safety concerns: pemoline has a history of hepatotoxicity, leading to FDA safety alerts and voluntary market withdrawal in certain settings.
- Regulatory restrictions: recent updates include monitoring for liver function tests, complicating prescribing.
- Competition: methylphenidate and amphetamine formulations dominate the market, with established safety and efficacy profiles.
Market Opportunity and Growth Potential
- Target niche populations: adults and patients with stimulant contraindications or intolerances.
- Market expansion is limited by safety profile and prescriber hesitation.
- Potential for regulatory shifts: ongoing research and post-marketing surveillance may influence future approvals or labeling changes.
Price Projections (2023-2028)
Current Pricing
- Average wholesale price (AWP): approximately $400 per month for a standard dose.
- Typical out-of-pocket cost for patients: $10–$50/month with insurance.
Near-term (2023-2025)
- Slight price stability anticipated due to limited market penetration.
- Manufacturers may maintain price points to recover R&D and regulatory costs.
Mid-term (2025-2028)
- Price stabilization expected unless therapeutic positioning shifts or new formulations emerge.
- Introduction of generic versions could reduce mean prices by 20-40%, depending on patent status and market competition.
Long-term Outlook
- Price decline plausible with increased competition and generics.
- Price may fluctuate within a $200–$400/month range, with variations driven by regulatory changes, safety profile updates, and market dynamics.
Competitive Analysis
| Competitor | Notable Advantages | Price Range (monthly) | Market Share (2022) |
|---|---|---|---|
| Methylphenidate (Ritalin, Concerta) | Well-established safety, widespread use | $100–$250 | 60% of ADHD market |
| Amphetamine (Adderall) | Rapid onset, high efficacy | $100–$300 | 30% of market |
| Non-stimulants (Atomoxetine) | Non-addictive, suitable for some patients | $250–$350 | 8% of market |
| Pemoline (APTENSIO XR) | Extended-release, niche use | $400 (current) | 2% of market |
Regulatory Environment Impact
Recent FDA safety communications and labelling updates influence prescribing trends. Updates include liver function monitoring, which can elevate costs and limit drug adoption. Any future easing or tightening of regulations directly impacts market size and pricing strategies.
Key Takeaways
- APTENSIO XR holds a niche market position with modest growth prospects.
- Market share remains limited due to safety concerns and entrenched competition.
- Current prices are around $400/month, with potential declines following increased generic competition.
- Future growth depends on regulatory developments and expanding clinical acceptability in special populations.
FAQs
Q1: What is the primary advantage of APTENSIO XR over other ADHD medications?
It offers a unique extended-release profile with a different pharmacokinetic profile, which may benefit specific patient subgroups.
Q2: How does safety impact APTENSIO XR’s market prospects?
Safety concerns, particularly hepatotoxicity, restrict broader adoption and influence prescriber use, limiting market expansion.
Q3: Are generics available for APTENSIO XR?
As of 2023, generics are not yet available; patent protections and market exclusivity are ongoing factors.
Q4: What factors could lower APTENSIO XR prices in the future?
Introduction of generic versions, increased competition, and regulatory pressure could depress prices by 20-40%.
Q5: How does the market for ADHD drugs evolve?
The market grows with increasing diagnoses; safety profiles, regulatory changes, and innovation drive shifts in drug choices and pricing.
References
- CDC. (2022). Data and Statistics on ADHD. Centers for Disease Control and Prevention.
- IQVIA. (2022). National Prescription Audit.
- FDA. (2020). Safety review of pemoline-containing products. U.S. Food and Drug Administration.
More… ↓
