Share This Page
Drug Price Trends for APRISO ER
✉ Email this page to a colleague
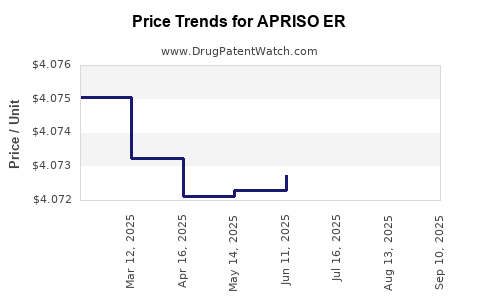
Average Pharmacy Cost for APRISO ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| APRISO ER 0.375 GRAM CAPSULE | 65649-0103-02 | 4.07102 | EACH | 2025-09-17 |
| APRISO ER 0.375 GRAM CAPSULE | 65649-0103-02 | 4.07198 | EACH | 2025-08-20 |
| APRISO ER 0.375 GRAM CAPSULE | 65649-0103-02 | 4.07234 | EACH | 2025-07-23 |
| APRISO ER 0.375 GRAM CAPSULE | 65649-0103-02 | 4.07275 | EACH | 2025-06-18 |
| APRISO ER 0.375 GRAM CAPSULE | 65649-0103-02 | 4.07230 | EACH | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for APRISO ER
What is APRISO ER?
APRISO ER (mesalamine extended-release) is a branded oral medication used to treat ulcerative colitis. It contains mesalamine, an anti-inflammatory agent that suppresses inflammation in the colon. Approved by the FDA for maintenance of remission in ulcerative colitis, APRISO ER is a product of Salix Pharmaceuticals, now part of Bausch Health.
Market Size and Current Position
Market Overview
The global inflammatory bowel disease (IBD) therapeutics market, which includes ulcerative colitis treatments like APRISO ER, was valued at approximately USD 4.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5.4% from 2023 to 2030 [1].
U.S. Market Specifics
The U.S. remains the largest market for ulcerative colitis drugs, representing roughly 70% of the global revenue in this area. The prevalence of ulcerative colitis in the U.S. affects about 907,000 people, with an increasing incidence rate that drives demand for maintenance therapies like APRISO ER [2].
Competitive Landscape
APRISO ER competes primarily with oral mesalamine formulations, including:
- Pentasa (Ferring Pharmaceuticals)
- Lialda (Viatris)
- Asacol (divided formulations; formerly part of Takeda)
- Generic mesalamine products
Despite multiple competitors, APRISO ER maintains a premium position due to its extended-release formulation and brand recognition.
Market Penetration and Revenue Data
Current Revenue
In 2022, APRISO ER generated approximately USD 250 million in worldwide sales, with the majority from the U.S. market [3].
Market Share
APRIso ER holds roughly 20-25% of the oral mesalamine market segment in the U.S., with its main competitors holding similar shares. The extended-release technology's differentiation supports premium pricing.
Price Analysis
Current Pricing
- Average Wholesale Price (AWP): Approximately USD 1,100 per month's supply (30 capsules).
- Insurance Reimbursement: Prices after co-pays range between USD 10-50 per month for insured patients, based on pharmacy benefit managers.
- Patient Out-of-Pocket: Typically USD 10-30 per month, influenced by insurance policies.
Price Trends
Over the past five years, the average wholesale price of APRISO ER has increased 3-5% annually, aligned with inflation and R&D costs. The entry of generics could pressure pricing downward; however, significant market share for APRISO ER has kept the price relatively stable.
Future Price Projections
Assuming patent exclusivity remains until 2028 and no significant market disruptions:
| Year | Projected Wholesale Price (USD) | Notes |
|---|---|---|
| 2023 | 1,100 | Current price |
| 2025 | 1,150 | Slight inflation, potential generic entry risk |
| 2028 | 1,200 | Post-patent expiry, likely price compression begins |
Post-2028, generic mesalamine products are expected to drive prices down substantially, potentially reducing AWP by 40-60%.
Patent and Regulatory Considerations
The primary patent for APRISO ER expired in 2023, with exclusivity until 2028. Patent cliffs usually lead to increased generic competition, affecting pricing and market share.
Key Market Dynamics
- Patent Expiration: Expected in 2028
- Generic Competition: Likely to enter within 1-2 years after patent expiry
- Market Growth: Driven by increasing ulcerative colitis prevalence and treatment adherence
- Pricing Power: Will diminish approaching 2028, with significant downward pressure expected
Investment and R&D Outlook
Bausch Health continues exploring pipeline drugs for ulcerative colitis, potentially impacting future market share of APRISO ER. Development of formulations offering better efficacy or reduced side effects could offer differentiation post-generic entry.
Key Takeaways
- Current market size for APRISO ER is USD 250 million globally, primarily in the U.S.
- Prices have increased modestly, with an average monthly wholesale price around USD 1,100.
- Market share remains stable amid competition due to brand recognition and formulation advantages.
- Patent expiry forecast for 2028 indicates imminent risk of generic entry, leading to potential price declines.
- Post-2028, prices are projected to fall 40-60%, aligning with typical generic pricing trends in the IBD segment.
FAQs
1. When does the patent for APRISO ER expire?
The primary patent is scheduled to expire in 2028, after which generics are expected to enter the market.
2. How does APRISO ER differ from other mesalamine products?
It uses an extended-release formulation designed for once-daily dosing, potentially improving adherence.
3. What are the key drivers for future revenue decline?
Patent expiration and the entry of generics are the main factors. Increasing market competition and price compression are expected post-2028.
4. How does pricing impact patient access?
Insurance coverage significantly reduces out-of-pocket expenses, enabling broader access despite high wholesale prices.
5. Are there any upcoming formulations or competitors that threaten APRISO ER?
Yes. Multiple generics and alternative formulations are under development or already marketed, which could further erode APRISO ER’s market share and pricing.
More… ↓
