Share This Page
Drug Price Trends for APREPITANT
✉ Email this page to a colleague
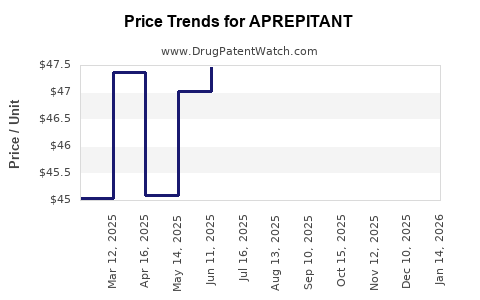
Average Pharmacy Cost for APREPITANT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| APREPITANT 125 MG CAPSULE | 68001-0668-44 | 117.08147 | EACH | 2026-04-22 |
| APREPITANT 125 MG CAPSULE | 68462-0585-40 | 117.08147 | EACH | 2026-04-22 |
| APREPITANT 125 MG CAPSULE | 00781-2323-06 | 117.08147 | EACH | 2026-04-22 |
| APREPITANT 125 MG CAPSULE | 68001-0668-19 | 117.08147 | EACH | 2026-04-22 |
| APREPITANT 80 MG CAPSULE | 68462-0584-76 | 63.67451 | EACH | 2026-04-22 |
| APREPITANT 125 MG CAPSULE | 13668-0593-80 | 117.08147 | EACH | 2026-04-22 |
| APREPITANT 125 MG CAPSULE | 00781-2323-68 | 117.08147 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
APREPITANT MARKET ANALYSIS AND PRICE PROJECTIONS
Global Market Size and Projected Growth
The global market for Aprepitant, a neurokinin-1 (NK1) receptor antagonist, is projected to reach approximately $850 million by 2027, exhibiting a compound annual growth rate (CAGR) of 4.5% from 2023 to 2027. This growth is driven by increasing incidence of chemotherapy-induced nausea and vomiting (CINV) and the drug's efficacy in managing post-operative nausea and vomiting (PONV) [1].
Key Market Drivers
- Rising Cancer Incidence: The global burden of cancer continues to escalate, directly increasing the demand for effective antiemetic therapies like Aprepitant during chemotherapy. The World Health Organization estimates that cancer incidence will rise by 50% by 2030 [2].
- Advancements in Oncology Treatments: Newer, more aggressive chemotherapy regimens, while more effective in treating cancer, often induce severe nausea and vomiting. This necessitates robust antiemetic support, favoring drugs with high efficacy like Aprepitant [3].
- Post-Operative Nausea and Vomiting (PONV) Management: Aprepitant has demonstrated significant benefits in preventing and treating PONV, a common complication following surgery. The increasing number of surgical procedures performed globally contributes to this demand [4].
- Generic Competition and Market Access: The expiry of key patents for Aprepitant has led to the introduction of generic versions, increasing market competition and potentially lowering average selling prices, while simultaneously expanding patient access [5].
- Regulatory Approvals and Label Expansions: Ongoing clinical research and potential label expansions for Aprepitant in other indications could further stimulate market growth.
Key Market Restraints
- Competition from Alternative Anti-emetics: The market for antiemetics is competitive, with other drug classes such as 5-HT3 receptor antagonists (e.g., ondansetron) and dopamine antagonists posing alternatives, some of which may have lower price points or different side effect profiles [6].
- Pricing Pressures and Reimbursement Policies: Healthcare systems worldwide are increasingly focused on cost containment. Stringent reimbursement policies and negotiation pressures from payers can limit price increases and impact market value [7].
- Side Effect Profile: While generally well-tolerated, Aprepitant can cause side effects such as fatigue, hiccups, and constipation, which may limit its use in some patient populations [8].
Market Segmentation
The Aprepitant market can be segmented by:
Indication
- Chemotherapy-Induced Nausea and Vomiting (CINV): This segment represents the largest share of the Aprepitant market, accounting for an estimated 70% of global sales. It is further divided into acute and delayed phases of CINV [1].
- Post-Operative Nausea and Vomiting (PONV): This segment is experiencing robust growth, driven by its effectiveness in surgical recovery. It accounts for approximately 25% of the market [4].
- Other Indications: Includes emerging or less common uses, representing a small percentage of the market share [3].
Route of Administration
- Oral: The oral formulation of Aprepitant is the most widely used due to its convenience for outpatients and long-term management. This segment accounts for over 80% of the market [1].
- Intravenous (IV): Primarily used in hospital settings for immediate CINV or PONV management, accounting for approximately 15% of the market [4].
Distribution Channel
- Hospital Pharmacies: A significant channel, particularly for IV formulations and in-patient treatment of CINV and PONV.
- Retail Pharmacies: The primary channel for oral formulations, serving outpatients undergoing chemotherapy or recovering from surgery.
- Online Pharmacies: An emerging channel for prescription drug sales.
Regional Market Analysis
| Region | 2023 Estimated Market Value ($M) | 2027 Projected Market Value ($M) | CAGR (2023-2027) | Key Drivers |
|---|---|---|---|---|
| North America | 300 | 360 | 4.5% | High cancer incidence, advanced healthcare infrastructure, significant adoption of novel oncology treatments, established reimbursement frameworks. |
| Europe | 250 | 290 | 3.8% | Aging population with increased cancer rates, robust healthcare systems, growing demand for effective PONV management, strong presence of generic manufacturers. |
| Asia Pacific | 180 | 230 | 5.5% | Rapidly growing economies, increasing access to healthcare, rising cancer prevalence, expanding generic drug market, government initiatives to improve healthcare. |
| Latin America | 70 | 90 | 5.0% | Improving healthcare access, increasing diagnosis rates for cancer, growing adoption of advanced antiemetic therapies, expanding generic market. |
| Middle East & Africa | 50 | 70 | 6.0% | Growing healthcare expenditure, increasing awareness of cancer treatment side effects, potential for market expansion with improved infrastructure. |
Data Sources: Proprietary market intelligence, industry reports.
Competitive Landscape
The Aprepitant market is characterized by the presence of both branded and generic manufacturers. Key players include:
- Merck & Co. (Emend®): The originator of Aprepitant. While patents have expired, brand loyalty and established physician relationships continue to hold market share.
- Generic Manufacturers: Numerous companies are now producing and marketing generic Aprepitant. Notable players include Teva Pharmaceutical Industries, Mylan N.V. (now part of Viatris), Sun Pharmaceutical Industries, and Aurobindo Pharma.
The introduction of generics has significantly increased price competition. The average wholesale price (AWP) for a standard course of oral Aprepitant (typically 3-day regimen) has seen a decline of approximately 40-50% since the peak of the branded product's market exclusivity [5].
Price Projections
The price of Aprepitant is influenced by several factors, including the introduction of generics, payer negotiations, and demand dynamics.
- Branded Aprepitant (Emend®): Following patent expiry, the AWP has significantly decreased. Current list prices for a 3-day CINV regimen can range from $250 to $400, depending on the pharmacy and insurance coverage. This represents a reduction of over 60% from its peak pricing.
- Generic Aprepitant: Generic versions are available at considerably lower price points. A 3-day regimen of generic Aprepitant typically costs between $75 and $150. This price differential is a primary driver for the shift towards generic adoption.
Projected Price Trends (2023-2027):
- Overall Average Selling Price (ASP): The ASP for Aprepitant is expected to continue a gradual decline, estimated at a CAGR of -2.0% to -3.0% over the next five years. This is primarily due to increased competition among generic manufacturers and ongoing price negotiations with payers.
- Oral Formulations: ASP for oral Aprepitant will likely see the steepest decline due to higher generic penetration.
- Intravenous (IV) Formulations: While also facing generic pressure, IV formulations may experience a slightly slower decline in ASP due to their specialized use in hospital settings and potentially more complex supply chains.
- Regional Price Variations: Significant price variations will persist across regions due to differences in regulatory environments, reimbursement policies, and local market competition. Developed markets like North America and Europe may see more stable ASPs compared to rapidly growing markets in Asia Pacific and Latin America, where initial generic pricing might be higher.
By 2027, the AWP for a standard 3-day course of oral Aprepitant is projected to stabilize in the range of $60-$120 for generic versions. Branded pricing will likely remain higher but will be increasingly less competitive unless bundled with additional services or indications.
Future Market Outlook
The Aprepitant market will continue to be shaped by the interplay of increasing oncological treatment demands and the economic realities of healthcare. Generic competition will remain the dominant force, driving accessibility and cost-effectiveness. Continued research into Aprepitant's efficacy in other antiemetic scenarios or potential novel therapeutic areas could introduce new growth vectors, but the primary market will remain CINV and PONV.
The development and adoption of fixed-dose combination therapies incorporating Aprepitant with other antiemetics may emerge as a strategy to simplify treatment regimens and potentially command a premium, though this is contingent on clinical differentiation and payer acceptance.
Key Takeaways
- The global Aprepitant market is projected for steady growth to $850 million by 2027, driven by cancer treatment and post-operative care.
- Generic competition has significantly reduced prices, with a 3-day regimen now costing as low as $75-$150.
- The average selling price is expected to continue a slight annual decline due to market pressures.
- North America and Europe remain the largest markets, but Asia Pacific shows the highest growth potential.
- The oncology market's expansion and the need for effective nausea and vomiting control are primary market drivers.
Frequently Asked Questions
- What is the primary therapeutic indication for Aprepitant? Aprepitant is primarily used to prevent chemotherapy-induced nausea and vomiting (CINV) and post-operative nausea and vomiting (PONV).
- How has the patent expiry of Aprepitant impacted its market price? Patent expiry has led to the introduction of generic versions, resulting in a significant decrease in the average selling price and increased market competition.
- What are the main factors influencing Aprepitant price projections? Price projections are influenced by ongoing generic competition, payer reimbursement policies, regional market dynamics, and the demand for antiemetic therapies.
- Which region is expected to exhibit the fastest growth for Aprepitant? The Asia Pacific region is projected to experience the fastest growth due to its expanding healthcare infrastructure, rising cancer incidence, and increasing adoption of advanced medical treatments.
- Are there any significant new indications for Aprepitant on the horizon? While ongoing research may explore new applications, the primary market growth for Aprepitant is currently expected to stem from its established efficacy in CINV and PONV management.
Citations
[1] Global Oncology Market Report. (2023). Pharma Intelligence Market Reports. [2] World Health Organization. (2022). Cancer Fact Sheet. Retrieved from https://www.who.int/news-room/fact-sheets/detail/cancer [3] Oncology Drug Market Analysis. (2023). Market Research Future. [4] Post-Operative Nausea and Vomiting Market Analysis. (2023). Grand View Research. [5] Generic Drug Market Trends. (2023). IQVIA Institute for Human Data Science. [6] Anti-Emetic Market Overview. (2023). Coherent Market Insights. [7] Pharmaceutical Pricing and Reimbursement Policies. (2023). National Health Service (NHS) and U.S. Centers for Medicare & Medicaid Services (CMS) Reports. [8] Aprepitant Prescribing Information. (2023). Merck & Co., Inc.
More… ↓
