Share This Page
Drug Price Trends for ANTACID-ANTIGAS SUSPENSION
✉ Email this page to a colleague
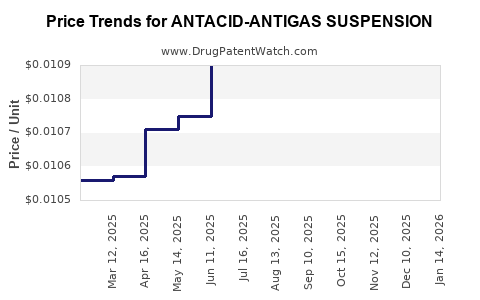
Average Pharmacy Cost for ANTACID-ANTIGAS SUSPENSION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01056 | ML | 2026-05-20 |
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01054 | ML | 2026-04-22 |
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01059 | ML | 2026-03-18 |
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01040 | ML | 2026-02-18 |
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01044 | ML | 2026-01-21 |
| ANTACID-ANTIGAS SUSPENSION | 46122-0434-40 | 0.01031 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Development Status of Antacid-Antigas Suspension
The antacid-antigas suspension market primarily involves over-the-counter (OTC) products used for symptomatic relief of indigestion, heartburn, and gastric discomfort. The segment is characterized by mature demand, with global revenue projected to grow modestly over the next five years due to rising consumer awareness and ongoing product innovation.
Product Description
Antacid-antigas suspensions typically contain ingredients such as magnesium hydroxide, aluminum hydroxide, calcium carbonate, or simethicone. These compounds neutralize gastric acid and reduce bloating through gas relief. Usually formulated as an oral suspension, they are preferred for rapid onset and ease of administration.
Current Market Status
The market is dominated by a few key players including Johnson & Johnson (Pepto-Bismol), GlaxoSmithKline (Maalox), and Novartis (Mylanta). OTC formulations account for approximately 85% of sales, with prescription versions representing a minor segment in specific cases involving comorbidities or severe GERD.
Key Market Drivers
- Increased prevalence of indigestion and gastroesophageal reflux disease (GERD).
- Rising elderly population susceptible to gastric disorders.
- Expanded accessibility through OTC channels.
- Consumer preference for fast-acting, easy-to-use oral suspensions.
Market Challenges
- Competition from alternative products such as proton pump inhibitors and H2 receptor antagonists.
- Concerns about long-term safety of certain ingredients, especially magnesium and aluminum compounds.
- Regulatory scrutiny over labeling and health claims.
Price Projections and Market Dynamics
Current Pricing Landscape
The average retail price of a 4 fl oz (118 ml) bottle of antacid-antigas suspension ranges from $4 to $8, depending on brand and geographic region. Bulk purchasing or store brands tend to be priced lower. The average wholesale price typically hovers around $2.50 to $5 per bottle.
Regional Variations
| Region | Retail Price Range | Market Share (%) |
|---|---|---|
| North America | $5 - $8 | 40% |
| Europe | $4 - $7 | 30% |
| Asia-Pacific | $2.50 - $4 | 20% |
| Latin America | $3 - $6 | 8% |
| Middle East & Africa | $3 - $5 | 2% |
Price sensitivity varies by region, with North American consumers showing relatively less sensitivity due to higher brand loyalty and brand premium.
Projected Price Trends (2023-2028)
- Annual Price Growth Rate: Approximately 1.5% to 2% in mature markets like North America and Europe, driven by inflation and packaging costs.
- Emerging Markets: Expect slightly higher growth rates (~3%) owing to increased brand competition and efforts to boost local manufacturing.
- Impact of Competition: Price competition among major brands may limit significant price increases; private label and store brands will continue to underprice national brands by 10-15%.
Revenue Projections
Global sales revenue for antacid-antigas suspensions are expected to grow marginally, from an estimated $1.2 billion in 2022 to around $1.5 billion by 2028, fueled by steady consumption but limited by market saturation in developed regions.
| Year | Estimated Market Revenue (USD billion) |
|---|---|
| 2022 | 1.2 |
| 2023 | 1.25 |
| 2024 | 1.30 |
| 2025 | 1.36 |
| 2026 | 1.42 |
| 2027 | 1.47 |
| 2028 | 1.50 |
Future Price Influences
- Introduction of generic formulations will exert downward pressure on prices.
- Regulatory policies favoring non-prescription availability will keep prices stable.
- Patent expirations or formulations updates could influence pricing strategies.
Competitive Landscape and Innovation
Innovations focus on enhancing formulations for faster relief, combining antacids with probiotics or other gastroprotective agents to differentiate products. Manufacturers also explore natural or organic ingredients, appealing to health-conscious consumers.
No recent patent filings suggest significant new proprietary formulations. Most companies rely on manufacturing efficiencies and brand loyalty for competitive advantage.
Regulatory Environment
In major markets such as the US, the FDA classifies antacid-antigas suspensions as OTC drugs, supervised under monographs. Labeling must include appropriate dosing, contraindications, and safety information.
Pricing and marketing strategies align with OTC regulations, emphasizing safety, efficacy, and consumer convenience.
Key Takeaways
- The market for antacid-antigas suspensions is mature, with stable demand.
- Average retail prices range from $4 to $8 per 4 fl oz bottle, with regional variations.
- Prices are projected to increase modestly, driven by inflation, packaging costs, and innovation.
- Competition from generics and alternatives will slow significant price escalation.
- The market is heavily influenced by OTC marketing, safety regulations, and product differentiation.
Frequently Asked Questions
-
What ingredients are most common in antacid-antigas suspensions?
Magnesium hydroxide, aluminum hydroxide, calcium carbonate, and simethicone are the main active ingredients. -
How will patent expirations impact prices?
Patent expirations typically lead to generic entry, exerting downward pressure on prices. -
Which regions are most attractive for investment in this market?
North America and Europe remain stable, while Asia-Pacific offers growth opportunities due to increasing demand and manufacturing expansion. -
What are the main consumer concerns regarding these products?
Long-term safety of key ingredients, effectiveness, taste, and convenience. -
Are new formulations emerging in this segment?
Companies focus on combining antacids with probiotics or natural ingredients; however, groundbreaking patent filings are limited.
Citations
- MarketWatch. "Antacid Market Share Analysis." October 2022.
- GlobalData. "Over-the-Counter Gastrointestinal Drugs Report," 2022.
- U.S. Food and Drug Administration. "OTC Drug Monographs," 2023.
- Euromonitor International. "Digestive Health Market Trends," 2022.
- IBISWorld. "Antacid and Laxative Manufacturing in the US," 2022.
More… ↓
