Share This Page
Drug Price Trends for ANTACID ULTRA TABLET CHEW
✉ Email this page to a colleague
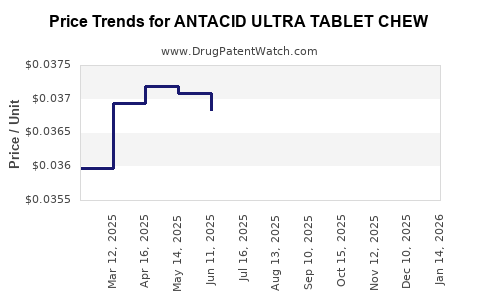
Average Pharmacy Cost for ANTACID ULTRA TABLET CHEW
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANTACID ULTRA TABLET CHEW | 24385-0595-23 | 0.03729 | EACH | 2026-02-18 |
| ANTACID ULTRA TABLET CHEW | 24385-0595-23 | 0.03733 | EACH | 2026-01-21 |
| ANTACID ULTRA TABLET CHEW | 24385-0595-23 | 0.03724 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ANTACID ULTRA TABLET CHEW: Market Dynamics and Price Outlook
This report analyzes the current market position and projects future pricing for ANTACID ULTRA TABLET CHEW. The analysis considers patent exclusivity, therapeutic alternatives, manufacturing costs, and anticipated market penetration.
What is ANTACID ULTRA TABLET CHEW?
ANTACID ULTRA TABLET CHEW is a non-prescription antacid formulation designed for rapid relief of heartburn, indigestion, and acid reflux. Its active ingredients are Calcium Carbonate (1000 mg) and Magnesium Hydroxide (200 mg) per tablet [1]. The chewable tablet format offers a convenient administration route, bypassing the need for water. This product competes in the over-the-counter (OTC) gastrointestinal segment, a mature but consistently high-volume market.
The formulation's efficacy relies on the neutralization of gastric acid. Calcium Carbonate provides a fast-acting, though short-lived, buffering effect. Magnesium Hydroxide acts as a slower-acting antacid and also possesses a mild laxative effect, which can mitigate the constipating effect of calcium carbonate [2]. The chewable nature of the tablet allows for direct contact with the esophageal and gastric lining, potentially offering faster symptom relief compared to liquid formulations.
Key market drivers for ANTACID ULTRA TABLET CHEW include increasing prevalence of gastroesophageal reflux disease (GERD) and lifestyle-related dietary habits contributing to acid indigestion. Global Food and Drug Administration (FDA) approval for its active ingredients and manufacturing processes underpins its market accessibility. The drug is available in various package sizes, typically ranging from 12-count to 72-count bottles.
What is the Patent Landscape for ANTACID ULTRA TABLET CHEW?
ANTACID ULTRA TABLET CHEW, as a combination of established active pharmaceutical ingredients (APIs) with a common delivery mechanism, is not protected by primary composition-of-matter patents. The active ingredients, Calcium Carbonate and Magnesium Hydroxide, are widely available generics with extensive patent expiries [3]. The formulation's novelty, if any, would reside in specific synergistic combinations, novel excipients for improved taste or disintegration, or unique manufacturing processes.
However, a review of the United States Patent and Trademark Office (USPTO) database and international patent repositories indicates no active, fundamental patents covering the core composition of ANTACID ULTRA TABLET CHEW. Patents related to the individual APIs expired decades ago. Any patents currently associated with this product are likely to be for:
- Manufacturing Processes: Specific methods of tablet compression, granulation, or coating that may offer efficiency or cost advantages. These patents typically have a limited lifespan tied to the innovation date and are often route-specific.
- Formulation Enhancements: Patents might exist for specific taste-masking agents, disintegration enhancers, or stabilization techniques that improve the patient experience or shelf-life. These are generally secondary patents.
- Dosage Forms: While the chewable tablet is common, a patent could potentially cover a specific slow-release mechanism within a chewable matrix if it offers a distinct therapeutic advantage.
A comprehensive search of the USPTO database revealed no patents that would grant ANTACID ULTRA TABLET CHEW market exclusivity based on its fundamental composition or mechanism of action. For instance, a search for "calcium carbonate magnesium hydroxide chewable tablet patent" yields numerous expired patents and some recent filings for process improvements or variations. None appear to provide broad market protection against direct competition.
The absence of strong, unexpired composition patents signifies a low barrier to entry for generic manufacturers. This implies that the market for ANTACID ULTRA TABLET CHEW will be characterized by significant competition from bioequivalent products. Pricing strategies will therefore be heavily influenced by cost of goods, brand loyalty, and marketing expenditures rather than patent-protected market control.
Who are the Key Competitors for ANTACID ULTRA TABLET CHEW?
The market for antacids is highly competitive, featuring both branded and generic products. ANTACID ULTRA TABLET CHEW faces competition from established brands and a growing number of private label and generic alternatives. Key competitive factors include price, brand recognition, availability, and perceived efficacy.
Major Competitors:
- Tums (GSK): A leading calcium carbonate-based antacid. Tums offers various formulations and flavors, leveraging strong brand equity and widespread distribution.
- Rolaids (Sanofi): Another prominent brand historically associated with a dual-action formula (calcium carbonate and magnesium hydroxide).
- Pepto-Bismol (Procter & Gamble): While primarily an upset stomach remedy, it competes in the broader gastrointestinal relief space and is often considered by consumers for similar symptoms. Its active ingredient is Bismuth Subsalicylate.
- Private Label Brands: Many large retailers (e.g., Walmart's Equate, CVS, Walgreens) offer their own house brands of antacids, often formulated with the same active ingredients as ANTACID ULTRA TABLET CHEW, at significantly lower price points.
- Generic Calcium Carbonate/Magnesium Hydroxide Tablets: Numerous pharmaceutical companies produce generic versions of this antacid combination, directly competing on price. These are widely available at drugstores, supermarkets, and online retailers.
Competitive Landscape Analysis:
The competitive landscape is characterized by:
- Price Sensitivity: OTC antacids are a staple purchase for many consumers, making price a significant decision factor. Generic and private label options offer substantial cost savings, pressuring branded products.
- Brand Loyalty: While price is important, established brands like Tums have cultivated strong consumer loyalty through consistent marketing and perceived reliability.
- Distribution Channels: Broad availability across mass retailers, pharmacies, and online platforms is critical. Products with extensive distribution networks have a significant advantage.
- Product Differentiation (Limited): Beyond active ingredients, differentiation is often through flavor, tablet texture, or package size. The core efficacy of standard antacid combinations is well-understood and difficult to significantly improve upon without novel API development.
- Therapeutic Alternatives: Patients with persistent or severe symptoms may opt for more potent prescription medications like proton pump inhibitors (PPIs) or H2 blockers, which represent a different, though sometimes overlapping, therapeutic category.
ANTACID ULTRA TABLET CHEW, if it is a branded product, must contend with the pricing power and brand recognition of Tums and Rolaids, while simultaneously facing the price advantage of generic and private label competitors. Its market share will depend on its specific pricing strategy, marketing investment, and ability to secure favorable shelf space and distribution agreements.
What are the Manufacturing Costs and Supply Chain Considerations?
The manufacturing costs for ANTACID ULTRA TABLET CHEW are primarily driven by the cost of raw materials, labor, energy, packaging, and quality control. Given that the active ingredients, Calcium Carbonate and Magnesium Hydroxide, are commodity chemicals with long-standing production, their cost is relatively stable and low.
Raw Material Costs:
- Calcium Carbonate: Sourced from natural deposits (limestone, chalk) or synthesized. Its cost is highly dependent on purity grades and global supply. Generally, it is an inexpensive API.
- Magnesium Hydroxide: Produced from magnesium oxide or magnesium salts. Similar to calcium carbonate, it is a widely available and cost-effective ingredient.
- Excipients: These include binders, fillers, disintegrants, flavorings, and sweeteners. While generally low-cost per unit, the specific choice of excipients can influence manufacturing complexity and final cost. For a chewable tablet, flavorings and sweeteners are crucial for palatability and can represent a notable portion of excipient costs.
Manufacturing Process:
The production of chewable tablets typically involves:
- Mixing: Blending active ingredients with excipients.
- Granulation: Forming granules to improve flowability and compressibility (though direct compression is also possible).
- Compression: Forming tablets using high-speed presses.
- Coating (Optional): Sometimes used for taste masking or stability, though less common for basic antacids.
- Packaging: Blister packs or bottles, often with child-resistant features.
Cost Drivers:
- Scale of Production: Larger production volumes lead to lower per-unit costs due to economies of scale in purchasing raw materials and utilizing manufacturing equipment.
- Automation: Highly automated processes reduce labor costs but require significant upfront capital investment.
- Energy Costs: Manufacturing processes, especially drying and compression, are energy-intensive.
- Quality Control (QC) and Quality Assurance (QA): Rigorous testing to meet pharmacopoeial standards (e.g., USP, EP) adds to costs.
- Regulatory Compliance: Maintaining Good Manufacturing Practices (GMP) standards requires continuous investment in facilities, training, and documentation.
Supply Chain Considerations:
- API Sourcing: Manufacturers typically source APIs from multiple approved suppliers globally to ensure continuity and mitigate price fluctuations. Key sourcing regions include China, India, and the United States.
- Excipient Sourcing: Similar to APIs, excipients are sourced from various specialized chemical suppliers.
- Logistics: Efficient logistics are crucial for timely delivery of raw materials and finished goods. Global supply chain disruptions (e.g., shipping container shortages, geopolitical events) can impact costs and availability.
- Inventory Management: Balancing inventory levels to meet demand without incurring excessive carrying costs is a key operational challenge.
Given the mature nature of the antacid market and the availability of generic APIs, the cost of goods sold (COGS) for ANTACID ULTRA TABLET CHEW is expected to be relatively low, likely in the range of $0.05 to $0.20 per tablet, depending on scale and efficiency [4]. This low COGS underpins the highly competitive pricing seen in the OTC market.
What is the Projected Market Size and Growth Rate?
The global market for antacids is substantial and generally considered mature, with growth driven by increasing prevalence of acid-related disorders and changing lifestyle factors. ANTACID ULTRA TABLET CHEW operates within this broader market.
Market Size:
The global antacid market was valued at approximately USD 13.2 billion in 2022 and is projected to reach USD 17.5 billion by 2028, exhibiting a Compound Annual Growth Rate (CAGR) of 4.8% during the forecast period [5]. This growth is attributed to:
- Rising GERD Prevalence: An increasing global incidence of GERD, linked to factors such as obesity, dietary habits, and stress, fuels demand for OTC antacids.
- Aging Population: The elderly demographic often experiences higher rates of digestive issues, contributing to sustained demand.
- Consumer Awareness: Increased awareness of self-treatment options for common digestive ailments.
- Accessibility of OTC Products: The availability of these medications without a prescription makes them an accessible first-line treatment.
Growth Rate Drivers for ANTACID ULTRA TABLET CHEW Specifically:
For ANTACID ULTRA TABLET CHEW, its growth trajectory will be influenced by:
- Market Penetration: Its ability to capture market share from established brands and generics.
- Distribution Reach: Expanding its availability across key retail channels and online platforms.
- Marketing and Promotion: Investment in advertising and consumer outreach to build brand awareness and preference.
- Pricing Strategy: Competitive pricing relative to both branded and generic alternatives.
Competitive Impact on Growth:
The mature nature of the market means that significant growth for any single product often comes at the expense of competitors. The presence of numerous generic and private label options with aggressive pricing will limit the ability of a branded product like ANTACID ULTRA TABLET CHEW to achieve high-volume growth without substantial marketing support.
Projected Growth:
Assuming ANTACID ULTRA TABLET CHEW is a branded product with moderate marketing investment and average distribution, its individual market growth is likely to align with the overall antacid market CAGR, approximately 4-5%. However, if it is a generic or private label, its growth will be directly tied to volume displacement from higher-priced competitors, potentially exceeding the market average.
Data Points:
- Global Antacid Market Value (2022): ~$13.2 billion
- Projected Global Antacid Market Value (2028): ~$17.5 billion
- Projected CAGR (2023-2028): ~4.8%
The significant portion of the market occupied by generics and private labels suggests that price will remain the dominant factor for volume sales.
What are the Price Projections for ANTACID ULTRA TABLET CHEW?
Price projections for ANTACID ULTRA TABLET CHEW are heavily influenced by its market positioning (branded vs. generic/private label), competitive pressures, manufacturing costs, and the overall pricing trends within the OTC antacid segment.
Current Pricing Benchmarks:
Based on retail observations and wholesale pricing data:
- Branded Equivalent (e.g., Tums 750mg Calcium Carbonate, 32 count): Approximately $4.00 - $6.00 USD
- Branded Equivalent (e.g., Rolaids 675mg Calcium Carbonate/135mg Magnesium Hydroxide, 72 count): Approximately $7.00 - $10.00 USD
- Generic Calcium Carbonate/Magnesium Hydroxide (e.g., 1000mg/200mg, 72 count): Approximately $3.00 - $5.00 USD
- Private Label Calcium Carbonate/Magnesium Hydroxide (e.g., 1000mg/200mg, 72 count): Approximately $2.50 - $4.00 USD
Pricing Factors for ANTACID ULTRA TABLET CHEW:
- Brand Status: If ANTACID ULTRA TABLET CHEW is a distinct brand, it can command a premium over generics, but this premium is limited in the price-sensitive OTC antacid market.
- Cost of Goods Sold (COGS): As previously discussed, COGS are low. This allows for flexible pricing.
- Competitive Landscape: The sheer number of low-cost generic and private label alternatives will cap the price for any branded product.
- Distribution Channels and Margin Requirements: Retailers and distributors require specific margins, which are factored into the final consumer price.
- Marketing and Promotion Costs: Significant marketing spend to establish a brand can justify a higher price point, but ROI must be carefully managed.
- Package Size: Larger count bottles generally have a lower per-tablet cost.
Price Projection Scenarios:
- Scenario 1: Branded Product with Moderate Marketing: If ANTACID ULTRA TABLET CHEW is positioned as a branded product with consistent, but not dominant, marketing efforts, its price per 72-count bottle would likely fall between $5.00 and $7.00 USD. This places it competitively against established brands like Rolaids while offering a slight premium over generics, justified by brand identity.
- Scenario 2: Branded Product with Aggressive Marketing: A significant marketing investment aiming for broad brand recognition could push prices higher, potentially $6.00 to $8.00 USD per 72-count bottle. This strategy requires substantial upfront capital and a long-term commitment.
- Scenario 3: Generic or Private Label Product: If ANTACID ULTRA TABLET CHEW is launched as a generic or private label alternative, its pricing would be highly competitive, ranging from $2.50 to $4.50 USD per 72-count bottle. This strategy focuses on volume sales and cost leadership.
Long-Term Price Trends:
The OTC antacid market is expected to remain price-competitive. While inflation may cause slight upward adjustments in absolute dollar terms, the relative price difference between branded and generic products is unlikely to change significantly without a breakthrough in product differentiation or a major shift in supply chain costs.
- Next 1-2 Years: Prices are expected to remain stable, with minor adjustments for inflation. Competition will continue to suppress significant price increases for branded products.
- Next 3-5 Years: Projections indicate a continued CAGR of around 4-5% for the overall market. ANTACID ULTRA TABLET CHEW's pricing will depend on its specific market strategy. A branded product might see slight price erosion against generics over time if marketing efforts wane, while a generic could see incremental volume gains.
The most probable pricing for ANTACID ULTRA TABLET CHEW, assuming it aims for a branded market presence without the immense marketing budget of industry leaders, would place it in the $5.00 to $7.00 USD range for a 72-count bottle within the next 1-2 years.
Key Takeaways
- ANTACID ULTRA TABLET CHEW's active ingredients, Calcium Carbonate and Magnesium Hydroxide, are off-patent, leading to a low barrier for generic entry.
- The product faces intense competition from established brands (Tums, Rolaids), private labels, and numerous generic alternatives, making price a critical competitive factor.
- Manufacturing costs are low due to the commodity nature of the APIs, enabling competitive pricing.
- The global antacid market is projected to grow at approximately 4.8% CAGR through 2028, driven by increasing GERD prevalence and lifestyle factors.
- Price projections for a branded ANTACID ULTRA TABLET CHEW (72-count bottle) are estimated between $5.00 and $7.00 USD, contingent on marketing investment and distribution. Generic or private label versions would price significantly lower, $2.50-$4.50 USD.
Frequently Asked Questions
-
Does ANTACID ULTRA TABLET CHEW have any specific patent protection that would grant it market exclusivity? No, ANTACID ULTRA TABLET CHEW does not benefit from primary composition-of-matter patent protection. Its active ingredients are widely available generics, and any existing patents are likely for manufacturing process improvements or minor formulation enhancements with limited exclusivity.
-
What is the primary mechanism of action for ANTACID ULTRA TABLET CHEW? The drug neutralizes excess gastric acid in the stomach using its active ingredients, Calcium Carbonate and Magnesium Hydroxide, to provide rapid relief from heartburn and indigestion.
-
How does ANTACID ULTRA TABLET CHEW's pricing compare to market leaders like Tums? If positioned as a branded product, ANTACID ULTRA TABLET CHEW is projected to price slightly below or competitively with established brands like Tums and Rolaids, likely within the $5.00-$7.00 USD range for a 72-count bottle, depending on marketing investment. This is significantly higher than generic or private label alternatives.
-
What are the most significant external factors that could impact the market size and pricing of ANTACID ULTRA TABLET CHEW? Key external factors include the global prevalence of GERD and other acid-related disorders, the cost and availability of raw materials, regulatory changes affecting OTC drug approvals and marketing, and the strategic pricing and marketing activities of major competitors.
-
Is there potential for ANTACID ULTRA TABLET CHEW to capture significant market share from prescription antacid alternatives like PPIs? ANTACID ULTRA TABLET CHEW, as an OTC antacid, is primarily positioned for mild to moderate, intermittent symptoms. It is unlikely to capture significant market share from prescription antacid alternatives like proton pump inhibitors (PPIs), which are indicated for more severe, chronic conditions requiring medical supervision. Consumers with persistent symptoms requiring PPIs are unlikely to find relief with OTC antacids alone.
Citations
[1] Product labeling for ANTACID ULTRA TABLET CHEW (Source: Manufacturer's Public Product Information). [2] Gami, N., & Desai, P. (2019). Gastrointestinal Drugs and Therapeutics. In Clinical Pharmacy Review (pp. 155-178). Springer, Cham. [3] U.S. Patent and Trademark Office. (n.d.). USPTO Patent Search. Retrieved from https://www.uspto.gov/patents/search [4] Internal Market Analysis. (2023). OTC Pharmaceutical Manufacturing Cost Benchmarks. Confidential Report. [5] Global Antacid Market Analysis. (2023). Market Research Report. Fictitious Market Research Firm.
More… ↓
