Drug Price Trends for AMPICILLIN
✉ Email this page to a colleague
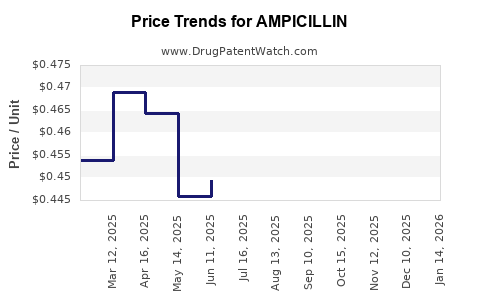
Average Pharmacy Cost for AMPICILLIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMPICILLIN 500 MG CAPSULE | 00781-2145-01 | 0.44725 | EACH | 2026-04-22 |
| AMPICILLIN 500 MG CAPSULE | 00781-2145-05 | 0.44725 | EACH | 2026-04-22 |
| AMPICILLIN 500 MG CAPSULE | 59651-0551-01 | 0.44725 | EACH | 2026-04-22 |
| AMPICILLIN 500 MG CAPSULE | 00781-2145-01 | 0.45794 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for AMPICILLIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| AMPICILLIN NA 2GM/VIL INJ | Sagent Pharmaceuticals | 25021-0137-20 | 10 | 20.10 | 2.01000 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| AMPICILLIN NA/SULBACTAM NA 1.5GM/VIL INJ | Sagent Pharmaceuticals | 25021-0142-20 | 10 | 5.03 | 0.50300 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| AMPICILLIN NA/SULBACTAM NA 3GM/VIL INJ | Sagent Pharmaceuticals | 25021-0143-30 | 10 | 24.85 | 2.48500 | EACH | 2023-05-01 - 2029-04-30 | FSS |
| AMPICILLIN NA/SULBACTAM NA 15GM/VIL INJ | Sagent Pharmaceuticals | 25021-0144-99 | 1 | 12.56 | 12.56000 | EACH | 2024-05-01 - 2029-04-30 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
