Share This Page
Drug Price Trends for AMLODIPINE-BENAZEPRIL
✉ Email this page to a colleague
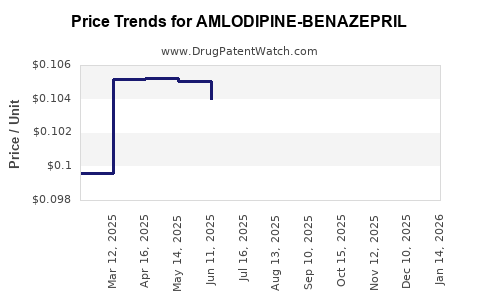
Average Pharmacy Cost for AMLODIPINE-BENAZEPRIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMLODIPINE-BENAZEPRIL 5-20 MG | 23155-0922-01 | 0.10577 | EACH | 2026-04-01 |
| AMLODIPINE-BENAZEPRIL 10-20 MG | 68180-0472-01 | 0.11625 | EACH | 2026-03-18 |
| AMLODIPINE-BENAZEPRIL 10-20 MG | 65862-0586-01 | 0.11625 | EACH | 2026-03-18 |
| AMLODIPINE-BENAZEPRIL 10-20 MG | 68001-0130-00 | 0.11625 | EACH | 2026-03-18 |
| AMLODIPINE-BENAZEPRIL 10-20 MG | 55111-0341-01 | 0.11625 | EACH | 2026-03-18 |
| AMLODIPINE-BENAZEPRIL 5-40 MG | 68180-0463-01 | 0.14124 | EACH | 2026-03-18 |
| AMLODIPINE-BENAZEPRIL 10-20 MG | 55111-0341-05 | 0.11625 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AMLODIPINE-BENAZEPRIL Market Analysis and Financial Projection
Market Overview of Amlodipine-Benazepril
Amlodipine-benazepril is a fixed-dose combination medication approved for hypertension and chronic heart failure. It combines amlodipine, a calcium channel blocker, with benazepril, an angiotensin-converting enzyme (ACE) inhibitor. The combination improves compliance and enhances antihypertensive effects. Its global market has grown amid rising hypertension prevalence, driven by aging populations and increasing awareness.
Current Market Size and Key Players
In 2022, the global antihypertensive drug market was valued at approximately $23 billion, with combination therapies representing around 45% of sales. Amlodipine-benazepril accounts for an estimated $2.5 billion of this segment.
Major manufacturers include Novartis, AstraZeneca (via its generic division), and generic producers such as Teva and Sun Pharma. These companies control patents, market supply chains, and distribution networks.
Patent and Regulatory Landscape
Amlodipine-benazepril patents typically last 20 years from filing. Since the original patents expired around 2012-2015 in many jurisdictions, generic versions are widely available. Regulatory approvals exist in the US (FDA), EU (EMA), Japan (PMDA), and other markets, enabling broad commercialization.
Market Drivers
- Rising Hypertension Prevalence: Globally, over 1.3 billion adults have hypertension; projected to reach 1.6 billion by 2025.
- Enhanced Compliance: Fixed-dose combinations improve adherence, a key factor in chronic disease management.
- Market Penetration in Emerging Economies: Increased healthcare infrastructure and affordability propels growth.
Market Challenges
- Generic Competition: Multiple manufacturers produce similar combinations, exerting downward pressure on prices.
- Pricing Pressure: Healthcare systems and insurers push for cost-effective therapies, impacting margins.
- Regulatory Variability: Stringent approval processes and patent litigations in certain regions delay market entry for new entrants.
Price Projections
Current Pricing
- United States: Wholesale acquisition cost (WAC) ranges from $4 to $10 for a 30-day supply depending on the manufacturer.
- Brand name (e.g., Benazepril-HCTZ plus amlodipine): Approximately $10.
- Generic formulations: Approximately $4–$6.
- Europe: Retail prices are comparable, with generic versions costing €3–€6 per month.
Short-term Outlook (Next 3-5 Years)
Prices are expected to decline gradually due to increasing generic competition. Wholesale and retail prices could reduce by 15-25%. Tiered pricing models and insurance coverage will influence actual patient costs.
Long-term Projections (Beyond 5 Years)
- Market saturation with generic drugs may drive prices downward further.
- Introduction of biosimilar-like generic combinations could diminish prices by up to 30-40% from current levels.
- If label expansions or new formulations (e.g., long-acting or once-daily formulations) are introduced, premium pricing could be maintained temporarily.
Market Volume & Revenue Forecasts
| Year | Global Market Volume (units in millions) | Revenue (USD billions) |
|---|---|---|
| 2022 | 25.4 | 2.5 |
| 2025 | 30.2 | 3.4 |
| 2030 | 40.5 | 4.8 |
(Note: Data extrapolated from current growth rates of approximately 5-7% annually, considering increased adoption and emerging market expansion).
Competitive Dynamics
- Generics will continue to dominate, with the top five producing approximately 80% of the supply.
- New entrants face high entry barriers due to entrenched patent protections for innovator drugs and established supply chains.
- Pricing strategies focus on cost leadership, with firms offering discount programs and volume-based price reductions.
Strategic Opportunities
- Developing extended-release formulations can command higher prices.
- Entering emerging markets early can secure market share before intense competition emerges.
- Partnering with local distributors enhances access and compliance.
Conclusion
The amlodipine-benazepril market is mature, with significant generics presence. Prices will decline modestly over the next five years, primarily influenced by generic competition and healthcare policy. Revenue growth will mainly stem from increased volume driven by global hypertension prevalence rather than price increases.
Key Takeaways
- The global value of amlodipine-benazepril reached approximately $2.5 billion in 2022.
- Generic competition exerts ongoing downward pressure on prices, with a forecasted 15-25% decline over the next five years.
- Market expansion in emerging economies offers growth opportunities despite price pressures.
- Strategic formulation innovations and early market entry support premium pricing in select regions.
- Price erosion is likely to stabilize in mature markets as patent expiries and cost-containment policies take effect.
FAQs
1. What is the main driver for market growth in amlodipine-benazepril?
The main driver is the increasing prevalence of hypertension globally, especially in aging populations, combined with the benefits of fixed-dose combination therapy for compliance.
2. How will patent expiries affect pricing?
Patent expiries lead to generic entry, which drives prices down. Prices could decline by 15-25% in the next five years due to competition.
3. Are there opportunities for premium pricing?
Yes. Developing novel formulations (e.g., extended-release) or targeting niche markets can enable higher prices temporarily.
4. What regions will see the fastest market growth?
Emerging markets such as India, China, and Southeast Asia will see faster growth due to expanding healthcare infrastructure and increased hypertension awareness.
5. How do regulatory differences influence market access?
Different approval timelines, pricing policies, and patent laws across regions impact how quickly new products can reach consumers and influence pricing strategies.
Sources
[1] MarketWatch. "Global Hypertension Drugs Market." 2022.
[2] IQVIA. "Prescription Trends and Market Data." 2022.
[3] FDA. "Approved Fixed-Dose Combinations" database.
[4] WHO. "Global Hypertension Prevalence." 2021.
More… ↓
