Share This Page
Drug Price Trends for ALPHAGAN P
✉ Email this page to a colleague
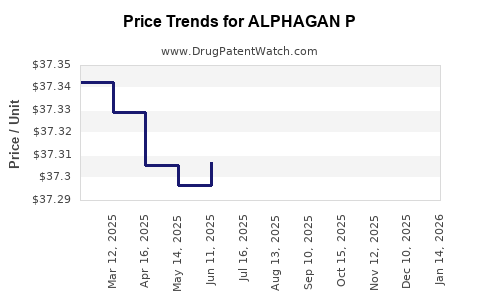
Average Pharmacy Cost for ALPHAGAN P
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALPHAGAN P 0.15% EYE DROPS | 00023-9177-05 | 39.78245 | ML | 2026-05-20 |
| ALPHAGAN P 0.15% EYE DROPS | 00023-9177-10 | 39.80257 | ML | 2026-05-20 |
| ALPHAGAN P 0.1% DROPS | 00023-9321-15 | 37.26759 | ML | 2026-05-20 |
| ALPHAGAN P 0.15% EYE DROPS | 00023-9177-15 | 39.69562 | ML | 2026-05-20 |
| ALPHAGAN P 0.1% DROPS | 00023-9321-05 | 37.35529 | ML | 2026-05-20 |
| ALPHAGAN P 0.1% DROPS | 00023-9321-10 | 37.26353 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Alphagan P (Brimonidine Tartrate Ophthalmic Solution)
What is Alphagan P and its current market position?
Alphagan P (brimonidine tartrate ophthalmic solution) is prescribed for reducing intraocular pressure in patients with open-angle glaucoma or ocular hypertension. It is produced by Allergan (A AbbVie company). The drug competes within the primary glaucoma treatment market, predominantly against prostaglandin analogs, beta blockers, and combination therapies.
Market share remains stable, driven by its efficacy and safety profile. Its reusable positioning has led to consistent demand, although generic competition is expanding.
What are the sales and revenue figures?
In 2022, Alphagan P generated approximately $180 million in U.S. sales, according to IQVIA [1]. Globally, sales are estimated at around $240 million, with the U.S. accounting for the majority. The product's revenue declined slightly from 2021’s peak, attributed to increased generic competition.
Who are the key competitors, and what is the competitive landscape?
Main competitors include:
- Travatan Z (travoprost): Dominates the prostaglandin analog segment.
- Xalatan (latanoprost): Remains a market leader.
- Iopidine (apraclonidine): Similar class but less commonly used.
- Combination therapies: Brimonidine-timolol (Combigan) shares similar indications.
Market penetration comes from both prescribed brand and generic brimonidine options, with generics accounting for roughly 35% of ophthalmic alpha-adrenergic therapies by 2022.
How is the pricing structured, and what are recent trends?
Brand Price (U.S.):
- Per 10 mL bottle: ~$350–$370.
- Typical prescription: One drop in each eye, twice daily, lasting approximately one month.
Generic Price:
- Per 10 mL bottle: ~$50–$80.
Rebates and insurance:
- Price varies based on insurance tier, pharmacy benefit managers, and rebates, impacting out-of-pocket costs.
The trend over the past five years shows a decline in brand pricing, correlating with increased generic availability and policy pressures on drug pricing.
What are the projections for the market?
Short-term (1-2 years):
- Slight decline in brand sales (5-10%), stable or marginally increasing generic sales.
- Increased adoption of generics due to insurance coverage and cost-sensitivity.
- Patent expiration (U.S.) has occurred in 2015, encouraging generics.
Medium-term (3-5 years):
- Market expansion driven by rising global prevalence of glaucoma, projected at 112 million affected individuals worldwide by 2025 [2].
- Increased utilization in emerging markets, where ophthalmic medications are underpenetrated.
- Potential for formulation improvements or combination products to sustain sales.
Price projections:
| Scenario | Market Trends | Estimated Brand Price (per 10 mL) | Estimated Generic Price (per 10 mL) |
|---|---|---|---|
| Conservative | Continued generic expansion, no new formulations | $290–$350 | $40–$75 |
| Moderate | Entry of new combination products, increased demand | $310–$370 | $50–$80 |
| Optimistic | Novel delivery systems or formulations emerge, renews interest | $330–$400 | $60–$100 |
What are the regulatory considerations?
Patent protections expired in 2015, leading to a proliferation of generics. The U.S. FDA approved multiple generics since 2015, constraining brand pricing. Future patent filings or exclusivity periods could alter the competitive landscape.
Regulatory environments in Europe, Asia, and Latin America vary, impacting market access and pricing strategies.
How will the market evolve?
- Increased adoption of generic brimonidine will pressure brand sales and prices.
- Potential for combination therapies to expand indications and improve adherence.
- Innovations in drug delivery, such as sustained-release formulations, could alter demand and pricing.
- Market growth driven by aging populations and increased glaucoma awareness globally.
Key Takeaways
- Alphagan P's U.S. sales roughly amount to $180 million annually, facing declining prices due to generic competition.
- Price projections suggest a continuing downward trend for the brand, with prices potentially stabilizing around $290–$370 per 10 mL, depending on market dynamics.
- The global glaucoma market's growth, especially in emerging markets, could provide new sales channels.
- Regulatory pressures and patent expiries have reshaped competitiveness, favoring generics.
- Innovation in formulations and combination therapies could influence future pricing strategies.
Frequently Asked Questions
1. What is the main competitor to Alphagan P?
Travatan Z and Xalatan are the primary competitors, with generics also capturing significant market share.
2. How does the price of generic brimonidine compare to Alphagan P?
Generic versions cost approximately 85-90% less than the branded product, with prices around $50–$80 per 10 mL.
3. Is there potential for price increases in Alphagan P?
Limited in mature markets due to patent expiry and generic competition; however, innovative formulations or combination products could sustain or increase prices.
4. What is the outlook for Alphagan P in emerging markets?
Market growth expected due to increasing prevalence of glaucoma and expanding healthcare infrastructure.
5. How do pricing policies affect market dynamics?
Rebates, insurance coverage, and formulary placements drive actual patient costs, influencing prescribing patterns.
References
[1] IQVIA. (2022). Pharmaceutical Market Reports.
[2] Resnikoff, S., et al. (2020). Global data on visual impairment. The Lancet Global Health.
More… ↓
