Share This Page
Drug Price Trends for ALMOTRIPTAN MALATE
✉ Email this page to a colleague
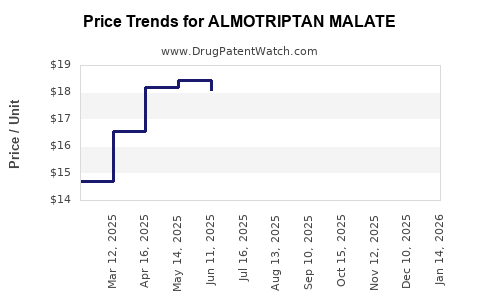
Average Pharmacy Cost for ALMOTRIPTAN MALATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALMOTRIPTAN MALATE 6.25 MG TAB | 27241-0041-68 | 19.07186 | EACH | 2026-05-20 |
| ALMOTRIPTAN MALATE 12.5 MG TAB | 00093-5261-19 | 17.92395 | EACH | 2026-05-20 |
| ALMOTRIPTAN MALATE 12.5 MG TAB | 00093-5261-29 | 17.92395 | EACH | 2026-05-20 |
| ALMOTRIPTAN MALATE 12.5 MG TAB | 00378-5246-85 | 17.92395 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is almotriptan malate?
Almotriptan malate is a selective serotonin receptor agonist used primarily for the acute treatment of migraines with or without aura. It was developed by Almirall and approved by the U.S. Food and Drug Administration (FDA) in 2007. It is sold under the brand name Axert in the United States and by various other companies globally. The drug is administered orally and is part of the triptan class, which includes drugs like sumatriptan and rizatriptan.
What is the current market landscape for almotriptan malate?
The global migraine medication market is projected to reach USD 4.5 billion by 2028, growing at a CAGR (compound annual growth rate) of approximately 4.5% from 2023 to 2028 [1]. Triptans account for a significant portion of this market, with almotriptan being one of the notable drugs due to its favorable safety profile and efficacy.
Market share and competition
- Sumatriptan: Holds roughly 45% of the triptan market, leading due to broader use and longer presence.
- Rizatriptan: Approximately 20%, popular because of rapid onset.
- Almotriptan: Estimated to have about 8-10% of the global triptan market. Its sales are concentrated in North America and Europe.
Key players
- Almirall (original developer): Manufactures Axert.
- Generic manufacturers: Several firms in India, China, and Europe produce generic almotriptan.
- Beyond Almirall, other pharmaceutical firms hold licensing rights or distribute in different regions.
Patent landscape
- Patent expiry: The original patent in the U.S. expired around 2014, leading to generic entry.
- Current patents: Some formulations and delivery mechanisms may still be under patent or exclusivity rights till 2030 [2].
Economic factors influencing price
The price of almotriptan malate varies widely by region and manufacturer. Key factors include:
- Brand versus generic pricing: Brand-name drugs like Axert tend to be priced between USD 30-50 per tablet, while generics cost USD 2-10 per tablet.
- Healthcare system differences: Countries with universal healthcare negotiate lower prices via tenders.
- Patent status: Patent protection maintains higher prices; generic entry drastically reduces prices.
Price projections and future trends
Short-term projections (2023-2025)
- In the U.S., the retail price for branded almotriptan is expected to remain around USD 30-50 per tablet.
- Generic prices in the U.S. are projected to stay below USD 10 per tablet, possibly declining further to USD 2-5 in the next two years.
- In Europe, prices for branded drugs are around EUR 25-40 per tablet, with generics costing EUR 2-8.
Long-term projections (2026-2030)
- Pricing for branded almotriptan is unlikely to decline significantly due to limited patent protections remaining.
- Generic market expansion will sustain low prices globally, especially in emerging markets.
- Market competition and potential biosimilar or new delivery formulations may influence price dynamics.
Impact of new formulations and competition
- Development of non-oral delivery (e.g., nasal sprays, auto-injectors) could influence market share.
- Availability of combination therapies aiming at migraine symptoms may dilute direct competition.
- Price erosion continues as biosimilars and generics expand, particularly across Asia and Latin America.
Regulatory and market factors affecting pricing
- Region-specific regulations impact drug approval, reimbursement, and pricing.
- Price controls in countries like Canada and several European nations limit maximum allowable prices.
- The U.S. Medicaid and Medicare pricing negotiations influence retail and pharmacy costs.
Key considerations for market entrants and investors
- Patent expiration is a critical juncture for generic pricing. Since the main patent expired in 2014, market entry of generics has saturated price reductions.
- Differentiation may focus on formulations, delivery systems, or combination therapies.
- Emerging markets offer growth opportunities due to increasing migraine prevalence and limited treatment options.
Summary table of pricing estimates
| Region | Branded Almotriptan (USD) per tablet | Generic Almotriptan (USD) per tablet |
|---|---|---|
| United States | 30 - 50 | 2 - 10 |
| Europe | 25 - 40 | 2 - 8 |
| Asia | 20 - 35 | 1 - 5 |
| Latin America | 15 - 30 | 1 - 4 |
Key Takeaways
- Almotriptan malate is a well-established migraine treatment with global sales concentrated in North America and Europe.
- Post-patent expiration has led to significant generic price reductions, shaping current market dynamics.
- Prices are expected to remain stable for branded versions but decline for generics, especially in emerging markets.
- Competition from other triptans and delivery methods will influence future pricing strategies.
- Regulatory policies and healthcare systems significantly impact retail prices and market access.
FAQs
1. How does almotriptan's efficacy compare with other triptans?
Almotriptan has comparable efficacy to other triptans such as sumatriptan and rizatriptan, with a favorable safety profile, particularly lacking vasospasm in some patients.
2. What factors influence the price of almotriptan in different markets?
Patent status, regional drug regulations, healthcare reimbursement policies, and market competition impact pricing across regions.
3. Are there any upcoming formulations or delivery systems for almotriptan?
Research is ongoing into non-oral delivery methods, including nasal sprays and auto-injectors, which may affect future pricing and market share.
4. How will patent expiration affect future market size?
Patent expiration increases generic competition, reducing prices and expanding access, but may suppress revenue for brand manufacturers.
5. What are the main barriers to market growth for almotriptan?
Market saturation by generics, competition from other triptans, and the emergence of new preventive therapies limit growth potential.
References
- Market Watch. "Global Migraine Drugs Market Size, Share & Trends Analysis Report," 2023.
- Pharmaceutical Patent Watch. "Almotriptan Patents," 2023.
More… ↓
