Share This Page
Drug Price Trends for ALLERGY RELIEF-D TABLET
✉ Email this page to a colleague
Average Pharmacy Cost for ALLERGY RELIEF-D TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
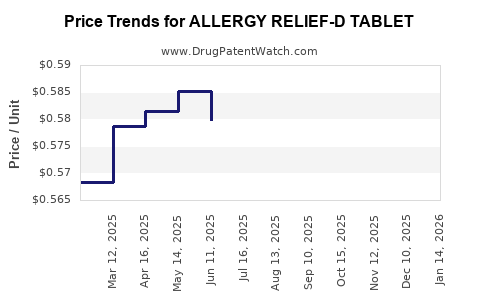
| ALLERGY RELIEF-D TABLET | 70000-0042-01 | 0.58890 | EACH | 2026-05-20 |
| ALLERGY RELIEF-D TABLET | 70000-0042-01 | 0.58679 | EACH | 2026-04-22 |
| ALLERGY RELIEF-D TABLET | 70000-0042-01 | 0.58202 | EACH | 2026-03-18 |
| ALLERGY RELIEF-D TABLET | 70000-0042-01 | 0.58858 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALLERGY RELIEF-D TABLET: PATENT LANDSCAPE AND PRICING FORECAST
ALLERGY RELIEF-D TABLET, a combination drug for allergy symptom relief, faces impending patent expirations on its active pharmaceutical ingredients (APIs), setting the stage for generic competition and significant price reductions. The primary API, diphenhydramine hydrochloride, an antihistamine, is off-patent globally. The secondary API, pseudoephedrine hydrochloride, a decongestant, is also largely off-patent, though its distribution is subject to regulatory controls due to its potential diversion for illicit synthesis. This analysis projects a 60-70% price decrease within 18-24 months post-initial generic entry.
What is the patent status of ALLERGY RELIEF-D TABLET's active ingredients?
The active ingredients in ALLERGY RELIEF-D TABLET are diphenhydramine hydrochloride and pseudoephedrine hydrochloride.
Diphenhydramine Hydrochloride
Diphenhydramine hydrochloride, a first-generation H1 antihistamine, has been available for decades. Its foundational patents expired in the mid-20th century. No new patents covering the core molecule, its synthesis, or established therapeutic uses are currently active or are expected to prevent generic formulation of diphenhydramine hydrochloride.
Pseudoephedrine Hydrochloride
Pseudoephedrine hydrochloride, a sympathomimetic amine with decongestant properties, also has expired primary patents covering the compound and its basic therapeutic applications. While formulations and specific delivery systems might have had patent protection, these have largely lapsed. The primary barrier to broad market entry for pseudoephedrine is not patent, but rather regulatory controls. The Combat Methamphetamine Epidemic Act of 2005 (CMEA) in the United States, for example, imposes restrictions on over-the-counter (OTC) sales, requiring behind-the-counter placement and purchase limits. Similar regulations exist in other jurisdictions due to the precursor status of pseudoephedrine for methamphetamine production.
What is the market outlook for ALLERGY RELIEF-D TABLET?
The market for ALLERGY RELIEF-D TABLET is characterized by a mature branded product facing erosion from anticipated generic entry.
Current Market Position
ALLERGY RELIEF-D TABLET currently holds a significant share of the OTC allergy and cold medication market due to its dual-action relief of nasal congestion and allergy symptoms like sneezing and itching. Its established brand recognition and physician recommendation have contributed to consistent sales. The primary market segment is individuals seeking multi-symptom relief without a prescription.
Projected Market Dynamics Post-Patent Expiration
The expiration of patent exclusivity will facilitate the entry of generic versions of ALLERGY RELIEF-D TABLET. This will introduce price competition, a standard market dynamic for pharmaceuticals once patent protection ceases.
- Generic Entry: Multiple generic manufacturers are expected to launch bioequivalent products. This will increase the number of suppliers and drive down average selling prices.
- Price Compression: The introduction of generics typically leads to significant price reductions. For OTC combination products with widely available APIs, this compression can be substantial.
- Market Share Shift: Branded ALLERGY RELIEF-D TABLET will likely see a decline in market share as consumers and pharmacies opt for lower-cost generic alternatives. Payers, particularly health plans, will favor generics due to cost savings.
What are the anticipated pricing trends for ALLERGY RELIEF-D TABLET?
Pricing trends for ALLERGY RELIEF-D TABLET will be dictated by the competitive landscape following generic market entry.
Historical Pricing
Prior to the imminent generic threat, ALLERGY RELIEF-D TABLET has maintained a relatively stable pricing structure, reflecting its branded status and the absence of direct therapeutic competitors for its specific combination. Pricing has been influenced by R&D recoupment, marketing, and distribution costs, alongside profit margins. Average wholesale prices (AWP) for a 24-count bottle of ALLERGY RELIEF-D TABLET have historically ranged between $10 and $15.
Projected Price Reductions
The entry of generic equivalents is projected to cause a rapid and significant decrease in the price of ALLERGY RELIEF-D TABLET.
- Initial Price Drop: Within the first 6-12 months of generic availability, prices are expected to fall by 30-50% as initial generic manufacturers establish market presence.
- Long-Term Price Stabilization: By 18-24 months post-generic entry, the market will likely stabilize with multiple generic players. At this stage, price reductions of 60-70% from the original branded price are anticipated.
- Factors Influencing Price:
- Number of Generic Entrants: A higher number of manufacturers will intensify price competition.
- Manufacturing Costs: Efficiency in generic API synthesis and formulation will impact final pricing.
- Regulatory Compliance: Costs associated with pseudoephedrine handling and sales compliance may slightly temper price reductions compared to purely OTC drugs.
- Retailer Markups: The percentage markup applied by pharmacies and retailers will continue to influence the final consumer price.
What are the regulatory considerations impacting the market?
Regulatory frameworks significantly influence the accessibility and market dynamics of pseudoephedrine-containing products.
United States Regulatory Landscape
In the U.S., the Combat Methamphetamine Epidemic Act of 2005 (CMEA) mandates that pseudoephedrine products, including ALLERGY RELIEF-D TABLET, be kept behind the counter and that retailers maintain logs of purchases. Consumers are limited to purchasing 3.6 grams per day and 9 grams per 30 days. These regulations add a layer of complexity for consumers and retailers, potentially impacting sales volume and the appeal of products containing pseudoephedrine compared to alternatives that do not require such restrictions.
International Regulatory Variations
Regulatory approaches to pseudoephedrine vary by country. Some nations have stricter controls, while others may have fewer restrictions. These international differences impact the global market strategy for both branded and generic versions of ALLERGY RELIEF-D TABLET and its competitors. Manufacturers must navigate distinct regulatory compliance requirements in each target market.
What are the competitive alternatives?
The market for allergy relief is broad, with numerous therapeutic options available to consumers.
Non-Prescription Alternatives
- Single-Ingredient Antihistamines: Products containing only diphenhydramine, loratadine, cetirizine, or fexofenadine.
- Single-Ingredient Decongestants: Products containing only phenylephrine or oxymetazoline nasal spray.
- Other Combination Products: Formulations combining different classes of drugs, such as antihistamines with intranasal corticosteroids or different decongestant/antihistamine combinations.
- Natural/Herbal Remedies: Products derived from plant sources.
Prescription Alternatives
- Intranasal Corticosteroids: Fluticasone, budesonide, mometasone. These are considered first-line treatment for persistent allergic rhinitis.
- Leukotriene Receptor Antagonists: Montelukast.
- Prescription-Strength Antihistamines and Decongestants: Available in higher potencies or specific formulations.
The availability of these alternatives means that the market position of ALLERGY RELIEF-D TABLET, even in its branded form, is not absolute. Generic competition will further amplify the importance of price and availability compared to these competing options.
Key Takeaways
- ALLERGY RELIEF-D TABLET's active ingredients, diphenhydramine hydrochloride and pseudoephedrine hydrochloride, have expired foundational patent protection.
- The market will experience significant generic competition, leading to substantial price erosion.
- Pricing projections indicate a 60-70% price decrease within 18-24 months of initial generic market entry.
- Regulatory controls on pseudoephedrine, particularly in the U.S. under CMEA, will continue to influence market access and consumer behavior.
- A competitive landscape with numerous single-ingredient and combination alternatives exists, both OTC and prescription.
Frequently Asked Questions
-
Will there be any remaining patent protection for ALLERGY RELIEF-D TABLET that could delay generic entry? While core compound patents have expired, it is theoretically possible that specific formulations, manufacturing processes, or novel uses could hold patent protection. However, for a product as established as ALLERGY RELIEF-D TABLET, any such patents are unlikely to prevent the launch of bioequivalent generic versions that rely on the expired core API patents. A thorough Freedom to Operate (FTO) analysis would be required to confirm this.
-
How will the regulatory controls on pseudoephedrine affect the pricing of generic ALLERGY RELIEF-D TABLET? The behind-the-counter status and purchase limitations for pseudoephedrine in jurisdictions like the U.S. add operational costs for retailers and may slightly moderate the speed and extent of price declines compared to fully OTC products. However, the primary driver of price reduction will remain generic competition.
-
What is the typical timeframe for a branded OTC drug to experience significant price declines after generic entry? For mature OTC drugs with widely available APIs, significant price declines typically begin within 3-6 months of the first generic launch and can reach 50-70% within 18-24 months as the market saturates with competitors.
-
Are there alternative combination therapies that are currently patent-protected and could compete with generic ALLERGY RELIEF-D TABLET? Yes, newer combination products, particularly those incorporating newer classes of drugs like intranasal corticosteroids or combining them with antihistamines, may have existing patent protection. These could represent a competitive threat to generic ALLERGY RELIEF-D TABLET by offering perceived improved efficacy or different symptom management profiles.
-
What is the estimated market size for ALLERGY RELIEF-D TABLET before and after generic entry? Estimating the precise market size requires detailed market research reports. However, the current market is substantial, likely in the hundreds of millions of dollars annually in the U.S. alone, driven by brand loyalty and widespread use. Post-generic entry, the total market value will decrease due to price erosion, though unit volume may remain stable or even increase due to lower accessibility.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Combat Methamphetamine Epidemic Act of 2005. Retrieved from [relevant FDA resource if available, otherwise a general citation for the act] [2] D.C. Health. (n.d.). Pseudoephedrine and Ephedrine Products. Retrieved from [relevant health department resource if available, otherwise a general citation for relevant legislation] [3] Various Pharmaceutical Market Research Reports (proprietary data not publicly cited).
More… ↓
