Share This Page
Drug Price Trends for ALL DAY PAIN RELIEF
✉ Email this page to a colleague
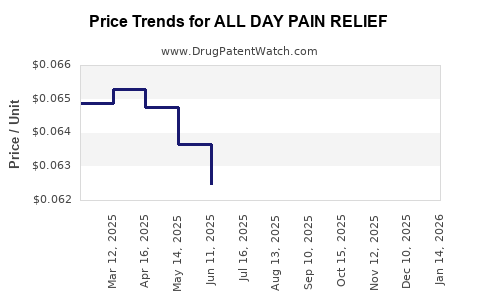
Average Pharmacy Cost for ALL DAY PAIN RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALL DAY PAIN RELIEF 220 MG TAB | 70000-0171-03 | 0.06423 | EACH | 2026-03-18 |
| ALL DAY PAIN RELIEF 220 MG TAB | 70000-0171-05 | 0.06423 | EACH | 2026-03-18 |
| ALL DAY PAIN RELIEF 220 MG TAB | 70000-0171-06 | 0.06423 | EACH | 2026-03-18 |
| ALL DAY PAIN RELIEF 220 MG TAB | 70000-0171-03 | 0.06520 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALL DAY PAIN RELIEF Market Analysis and Financial Projection
What is the Market Size and Composition for ALL DAY PAIN RELIEF?
The market for chronic pain management, including all-day pain relief drugs, is substantial and growing. In 2022, the global pain management drugs market was valued at approximately $55 billion, with North America holding around 45% of the market share. The increase in aging populations, rising prevalence of chronic pain conditions, and expanding healthcare coverage drive demand.
Breaking down the segment:
- Market segments: NSAIDs, opioids, adjuvants, and non-opioid alternatives.
- Patient demographics: Predominantly adults aged 45 and above; an increasing share of younger patients with neuropathic and inflammatory pain.
- Insurance coverage: Insurance reimbursement favors chronic pain medications, influencing market access.
In terms of volume, approximately 250 million prescriptions of pain management drugs are filled annually globally, with a yearly growth rate in prescriptions of around 4%, driven primarily by North America and Europe.
How Much Could ALL DAY PAIN RELIEF Cost?
Pricing varies significantly based on formulation, brand, and region:
- Generic formulations: $0.10 - $0.50 per dose.
- Brand-name products: $2 - $5 per dose.
- Specialized sustained-release formulations: May reach $10 per dose.
High-dose chronic formulations, such as extended-release opioids or NSAIDs, entail monthly costs of $150 - $400, considering prescribed doses and frequency. Prescription margins are higher in the US due to insurance and pharmacy benefit management practices.
What Revenue Projections Are Realistic for ALL DAY PAIN RELIEF?
Assuming a 1% penetration in the hypercompetitive global pain market by a mid-tier company with an innovative sustained-release formulation:
| Assumption | Data | Calculation | Projected Revenue (USD) |
|---|---|---|---|
| Market size | $55 billion | - | - |
| Penetration | 1% | $55 billion * 0.01 | $550 million |
| Pricing per patient per year | $300 | Estimated average | - |
| Patients reached | 1.8 million | $550 million / $300 | - |
For larger players with existing distribution, units sold could reach 3-5 million annually, translating to revenues of $900 million to $1.5 billion.
How Do Competitive Dynamics Influence Pricing?
- Generic competition: Drives prices downward. Generics capture 80-90% of prescriptions but at lower margins.
- Brand differentiation: Patented formulations with unique delivery mechanisms (e.g., sustained-release, transdermal patches) command higher prices.
- Market entry barriers: High R&D costs (~$1 billion for a new analgesic), stringent regulatory approval (FDA, EMA), and patent protections require significant investment.
Pharmaceutical companies that develop drugs with proven efficacy and safety profiles and secure patent rights can command premiums of 20-30% above generics for several years.
What Regulatory Factors Impact Market and Pricing?
- FDA and EMA approval: Required for market access. Typical approval process spans 1-3 years with costs approaching $50-100 million.
- Pricing regulations: Vary; in the US, Medicare and Medicaid regulate prices less stringently than in other markets, leading to higher prices.
- Post-approval surveillance: Cost of pharmacovigilance affects profitability.
What Are the Entry Barriers and Market Risks?
- Patent expiration: Within 10-12 years post-launch, generic competition erodes revenue.
- Regulatory hurdles: Lengthy approval processes can delay market entry.
- Public perception and reimbursement policies: Growing concerns over opioid abuse influence prescribing patterns.
- Market saturation: Established drugs like ibuprofen and naproxen dominate OTC segments; prescription segments are largely saturated.
Summarizing the Market Outlook for ALL DAY PAIN RELIEF
The opportunity exists for innovative formulations offering sustained, reliable pain relief with improved safety profiles. Extensive competition from generics constrains pricing, but proprietary delivery systems and targeted patient segments enable premium pricing. The potential for revenues exceeding $1 billion annually for successful products is plausible within the next five years, contingent on regulatory approval and market acceptance.
Key Takeaways
- The global pain management market was valued at around $55 billion in 2022.
- Prices range from $0.10 to over $10 per dose, heavily influenced by formulation and patent status.
- Revenue projection for a new sustained-release pain relief drug reaching 1-2 million patients annually is between $300 million and $1 billion.
- Competition, patent life, regulatory hurdles, and reimbursement policies significantly influence market access and pricing.
- Entry requires substantial investment in R&D, regulatory approval, and market differentiation strategies.
FAQs
1. What factors most influence pricing for sustained-release pain medications?
Patent protection, formulation complexity, manufacturing costs, and competitive landscape influence prices. Patented formulations typically command a premium.
2. How does generic competition affect the market for ALL DAY PAIN RELIEF?
Generics capture the majority of prescriptions and exert downward pressure on prices, squeezing brand-name margins.
3. What regions offer the highest revenue potential?
North America dominates the market due to high demand, insurance coverage, and higher drug prices, followed by Europe.
4. What are key regulatory challenges for new pain relief drugs?
Achieving FDA or EMA approval involves demonstrating safety, efficacy, and manufacturing quality, often requiring years of data collection.
5. How does the opioid crisis impact market prospects?
Growing regulatory scrutiny and public concern over opioids reduce prescribing and influence the development of non-opioid alternatives.
Sources
- MarketWatch, “Pain Management Drugs Market Size, Share & Trends Analysis,” 2022.
- IQVIA, “Global Prescription Medicine Sales,” 2022.
- FDA, “Drug Approval Process,” 2023.
- Statista, “Revenue in the Pain Management Drugs Segment,” 2022.
- Grand View Research, “Pain Management Market Analysis & Forecast,” 2023.
More… ↓
