Share This Page
Drug Price Trends for ALFUZOSIN HCL ER
✉ Email this page to a colleague
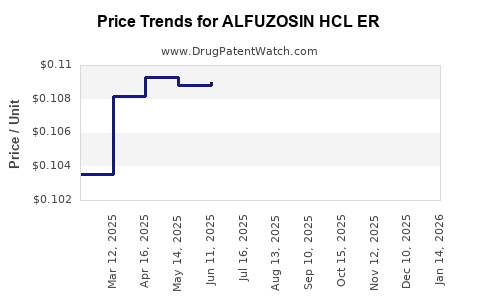
Average Pharmacy Cost for ALFUZOSIN HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALFUZOSIN HCL ER 10 MG TABLET | 76282-0302-05 | 0.11112 | EACH | 2026-03-18 |
| ALFUZOSIN HCL ER 10 MG TABLET | 57237-0114-90 | 0.11112 | EACH | 2026-03-18 |
| ALFUZOSIN HCL ER 10 MG TABLET | 76282-0302-01 | 0.11112 | EACH | 2026-03-18 |
| ALFUZOSIN HCL ER 10 MG TABLET | 29300-0155-01 | 0.11112 | EACH | 2026-03-18 |
| ALFUZOSIN HCL ER 10 MG TABLET | 60505-2850-09 | 0.11112 | EACH | 2026-03-18 |
| ALFUZOSIN HCL ER 10 MG TABLET | 29300-0155-19 | 0.11112 | EACH | 2026-03-18 |
| ALFUZOSIN HCL ER 10 MG TABLET | 76282-0302-10 | 0.11112 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALFUZOSIN HCL ER: Patent Landscape and Market Projections
Alfuzosin HCl ER, a selective alpha-1 adrenergic receptor antagonist, is primarily used for the treatment of symptomatic benign prostatic hyperplasia (BPH). Its efficacy in improving urine flow rate and reducing BPH symptoms underpins its market presence. Understanding the patent expiry and generic entry timeline is critical for competitive positioning and investment strategy.
What is the current patent status of Alfuzosin HCl ER?
The primary composition of matter patent for alfuzosin hydrochloride has expired in major markets. For instance, in the United States, U.S. Patent No. 4,535,186, which claimed the compound itself, expired in 2006. However, secondary patents, particularly those related to extended-release formulations and manufacturing processes, have extended market exclusivity for specific branded products.
- United States: The foundational patent has expired. However, branded products have relied on formulation patents, such as those covering specific extended-release mechanisms. For example, U.S. Patent No. 5,716,983, covering an extended-release dosage form, had an expiry date that facilitated generic competition for specific formulations. Key generic launches for alfuzosin HCl ER occurred following the expiry of these formulation patents.
- Europe: Similar patent expiry timelines have been observed. Core compound patents are long expired. Specific extended-release formulations have also seen patent expiries, enabling generic market entry. The European Patent EP0608499, for example, relates to pharmaceutical compositions and has seen its term expire.
- Japan: The patent situation mirrors that of the US and Europe, with original compound patents having expired, allowing for generic penetration after secondary patent expiries.
The landscape is characterized by the early expiry of the core active pharmaceutical ingredient (API) patent, followed by a period of protection for specific delivery technologies and formulations. This has historically allowed branded manufacturers to maintain market share for a defined period before significant generic competition emerges.
When did generic versions of Alfuzosin HCl ER become available?
The availability of generic alfuzosin HCl ER varies by region, dependent on specific patent expiries and any subsequent litigation or regulatory hurdles.
- United States: Generic versions of alfuzosin HCl ER began appearing in the U.S. market around 2008-2009, following the expiration of key formulation patents and successful patent challenges. Major generic manufacturers obtained FDA approval for their AB-rated bioequivalent products during this period.
- Europe: Generic entry in European countries has been staggered, with significant generic penetration occurring in the early to mid-2010s across major pharmaceutical markets. Countries typically see generic launches following the expiry of relevant national or European patents and the cessation of any marketing exclusivities granted to the originator product.
- Japan: The Japanese market experienced generic alfuzosin HCl ER launches subsequent to the expiry of its secondary patents, generally in the late 2000s and early 2010s.
The introduction of generics has consistently led to significant price erosion for the drug. The competitive pressure from multiple generic suppliers has been a primary driver of price reduction post-exclusivity.
What are the key therapeutic advantages of Alfuzosin HCl ER?
Alfuzosin HCl ER offers distinct therapeutic advantages for patients with symptomatic benign prostatic hyperplasia (BPH).
- Selective Alpha-1 Blockade: Alfuzosin selectively blocks alpha-1 adrenergic receptors in the prostate and bladder neck. This blockade relaxes the smooth muscle in these tissues, reducing the obstruction of the urethra and improving urine flow.
- Once-Daily Dosing: The extended-release formulation allows for convenient once-daily administration. This improves patient compliance compared to immediate-release formulations that may require more frequent dosing.
- Improved Urinary Flow Rate: Clinical studies demonstrate a statistically significant improvement in peak urine flow rate in patients treated with alfuzosin HCl ER compared to placebo.
- Reduced BPH Symptoms: The drug effectively alleviates lower urinary tract symptoms (LUTS) associated with BPH, including hesitancy, weak stream, incomplete emptying, and nocturia.
- Cardiovascular Safety Profile: Alfuzosin HCl ER is generally considered to have a favorable cardiovascular safety profile, particularly regarding orthostatic hypotension, compared to some older alpha-blockers. This is attributed to its uroselectivity.
- No Significant Impact on Blood Pressure (for most patients): While orthostatic hypotension can occur, alfuzosin HCl ER typically has less of a systemic impact on blood pressure compared to non-selective alpha-blockers. This is crucial for elderly patients who often have co-existing cardiovascular conditions.
These advantages have supported its sustained use in BPH management, even with the advent of newer treatment modalities.
Who are the major generic manufacturers of Alfuzosin HCl ER?
Several pharmaceutical companies have successfully developed and marketed generic versions of alfuzosin HCl ER. The presence of these manufacturers indicates a competitive market.
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a broad portfolio, including alfuzosin HCl ER.
- Sun Pharmaceutical Industries: Another major player in the generic pharmaceutical market, offering alfuzosin HCl ER in various regions.
- Apotex Inc.: A Canadian-based pharmaceutical company that has manufactured and distributed generic alfuzosin HCl ER.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with a significant generic drug presence, including alfuzosin HCl ER.
- Mylan N.V. (now Viatris): Has historically offered generic versions of alfuzosin HCl ER.
- Aurobindo Pharma: An Indian pharmaceutical company that produces a wide range of generics, including alfuzosin HCl ER.
The competitive landscape is populated by multiple authorized generic manufacturers and numerous independent generic players, contributing to price competition.
What is the projected market trajectory for Alfuzosin HCl ER?
The market trajectory for alfuzosin HCl ER is characterized by mature generic competition, with stable but declining prices driven by market saturation and ongoing cost pressures.
- Current Market Size: The global market for alfuzosin HCl ER is a subset of the broader BPH therapeutics market, which is valued in the billions of dollars. Specific market size data for alfuzosin HCl ER is influenced by regional pricing and the extent of generic penetration. Estimates place the annual global sales of branded and generic alfuzosin HCl ER in the hundreds of millions of dollars, though precise figures are proprietary.
- Price Trends: Following generic entry, prices for alfuzosin HCl ER have experienced significant reductions, often exceeding 70-80% compared to the peak branded price. The ongoing competitive intensity among generic manufacturers continues to exert downward pressure on prices. Prices are projected to remain stable at low levels, with minor fluctuations driven by supply-demand dynamics and tender bidding in institutional markets.
- Volume Trends: While prices decline, the volume of alfuzosin HCl ER prescribed is expected to remain relatively stable or experience modest growth. This is due to its established efficacy, favorable safety profile, and cost-effectiveness as a treatment option for a chronic condition affecting a large and aging male population. The increasing prevalence of BPH globally due to aging demographics will likely sustain demand.
- Competitive Landscape: The market is highly competitive, dominated by generic manufacturers. The absence of significant patent barriers for new entrants means that competition is primarily based on manufacturing efficiency, supply chain reliability, and market access. The emergence of new, highly differentiated BPH treatments could potentially impact market share for older drugs like alfuzosin, but its established position and low cost provide a significant defense.
- Geographic Considerations: Developed markets (North America, Europe) are characterized by high generic penetration and mature pricing. Emerging markets may see continued growth in generic adoption as healthcare access expands and cost-consciousness increases.
The overall outlook is for a mature, highly competitive generic market. Growth will be driven by volume demand in aging populations rather than significant price appreciation.
What are the key manufacturing considerations for Alfuzosin HCl ER?
Manufacturing alfuzosin HCl ER involves specific considerations related to API synthesis, extended-release formulation, quality control, and regulatory compliance.
- API Synthesis: The synthesis of alfuzosin hydrochloride requires a multi-step chemical process. Manufacturers must ensure robust supply chains for raw materials and intermediates. Process optimization for yield, purity, and cost-effectiveness is critical for competitive generic production. Key synthetic routes have been published and are well-understood by the industry.
- Extended-Release Formulation: Achieving the desired extended-release profile is a core manufacturing challenge. This typically involves specialized technologies such as:
- Matrix Systems: Embedding the drug in a polymer matrix that slowly releases the API over time.
- Coating Technologies: Applying specialized coatings to tablets or pellets that control drug release.
- Osmotic Pump Systems: While less common for alfuzosin, these could theoretically be employed for precise release. The choice of formulation technology impacts manufacturing complexity and cost. Bioequivalence studies are essential to demonstrate that the generic formulation delivers the drug at the same rate and extent as the reference listed drug.
- Quality Control and Assurance: Stringent quality control measures are paramount. This includes:
- API Purity Testing: Ensuring the absence of impurities and degradation products.
- Dissolution Testing: Verifying the extended-release profile meets specifications.
- Content Uniformity: Guaranteeing consistent drug dosage in each unit.
- Stability Studies: Assessing the shelf-life of the finished product under various storage conditions.
- Regulatory Compliance: Manufacturing facilities must adhere to Good Manufacturing Practices (GMP) as mandated by regulatory authorities such as the FDA (U.S.), EMA (Europe), and PMDA (Japan). This includes rigorous documentation, validation of processes, and regular inspections.
- Cost Management: For generic manufacturers, cost of goods sold (COGS) is a critical determinant of profitability. Efficient manufacturing processes, economies of scale, and competitive sourcing of raw materials are essential.
What is the competitive impact of alternative BPH treatments?
While alfuzosin HCl ER holds a significant place in BPH treatment, alternative therapies and newer drug classes exert competitive pressure.
- Other Alpha-Blockers: While alfuzosin is a selective alpha-1 blocker, other drugs in this class, such as tamsulosin, silodosin, and doxazosin, also compete. Tamsulosin, in particular, is widely prescribed and has a similar market position. Silodosin offers higher uroselectivity.
- 5-Alpha Reductase Inhibitors (5-ARIs): Drugs like finasteride and dutasteride work by shrinking the prostate gland. They are typically used for larger prostates and have a slower onset of action than alpha-blockers. They are often used in combination with alpha-blockers.
- Phosphodiesterase-5 (PDE5) Inhibitors: Sildenafil, tadalafil, and vardenafil, primarily known for erectile dysfunction, are also approved for BPH symptoms. Tadalafil, in particular, is indicated for both BPH and erectile dysfunction and offers once-daily dosing.
- Antimuscarinics: For patients with predominant storage symptoms (frequency, urgency, nocturia), antimuscarinic agents may be considered, sometimes in combination with alpha-blockers.
- Minimally Invasive and Surgical Interventions: Procedures like transurethral resection of the prostate (TURP), laser prostatectomy, and newer techniques such as prostatic urethral lift (PUL) or water vapor thermal therapy (Rezum) offer definitive or less invasive treatment options, particularly for moderate to severe BPH. These can reduce the long-term reliance on pharmacological therapy.
The choice of treatment depends on symptom severity, prostate size, patient comorbidities, and patient preference. Alfuzosin HCl ER's established efficacy, safety profile, and low cost make it a preferred option for many patients, particularly those with mild to moderate symptoms or when cost is a significant factor. However, the availability of these alternatives necessitates ongoing market evaluation.
Key Takeaways
- Alfuzosin HCl ER's core composition of matter patents have expired globally. Market exclusivity was extended through secondary patents covering specific extended-release formulations and manufacturing processes, with these also having expired in major markets.
- Generic versions of alfuzosin HCl ER became widely available in the U.S. around 2008-2009 and in Europe and Japan in the subsequent years, following patent expiries.
- Key therapeutic advantages include selective alpha-1 blockade, convenient once-daily dosing, improved urine flow, and a generally favorable cardiovascular safety profile.
- Major generic manufacturers include Teva Pharmaceuticals, Sun Pharmaceutical Industries, Apotex Inc., Dr. Reddy's Laboratories, and Aurobindo Pharma, among others.
- The market trajectory is characterized by mature, intense generic competition, leading to significant price erosion. Future growth will be volume-driven by the increasing prevalence of BPH in aging populations, with stable low pricing.
- Manufacturing considerations center on efficient API synthesis, specialized extended-release formulation technologies, stringent quality control, and adherence to global GMP standards.
- Alfuzosin HCl ER competes with other alpha-blockers, 5-ARIs, PDE5 inhibitors, antimuscarinics, and surgical interventions, with treatment choice depending on symptom severity and patient factors.
FAQs
1. Are there any new patents on alfuzosin HCl ER formulations or manufacturing processes that could extend market exclusivity?
As of current public patent databases, no significant new composition of matter or novel formulation patents for alfuzosin HCl ER are poised to grant substantial market exclusivity beyond the existing generic landscape. The innovation focus has largely shifted to therapeutic areas with unmet needs or to novel drug delivery systems for different drug classes.
2. How does the pricing of alfuzosin HCl ER compare to other alpha-blockers like tamsulosin or silodosin in the generic market?
In the generic market, alfuzosin HCl ER is generally competitively priced, often at the lower end of the spectrum for alpha-blockers. Tamsulosin HCl ER also has widespread generic availability and a similar low-price profile. Silodosin, which was a later entrant and has higher uroselectivity, may command a slightly higher price point even in its generic form, though this varies by region and manufacturer.
3. What is the typical patient profile that benefits most from alfuzosin HCl ER treatment?
Alfuzosin HCl ER is typically prescribed for adult males experiencing moderate to severe lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH). It is particularly beneficial for patients who value once-daily dosing convenience and require symptomatic relief of voiding dysfunction. Its favorable cardiovascular safety profile makes it a suitable option for many patients, including those with mild hypertension or at risk of orthostatic hypotension.
4. What are the primary regulatory hurdles for generic manufacturers entering the alfuzosin HCl ER market?
The primary regulatory hurdles involve demonstrating bioequivalence to the reference listed drug through rigorous pharmacokinetic and pharmacodynamic studies, meeting stringent quality control standards (e.g., impurity profiles, dissolution testing), and obtaining marketing authorization from regulatory bodies like the FDA, EMA, or PMDA. Facilities must also pass GMP inspections.
5. Can alfuzosin HCl ER be used in combination therapy for BPH?
Yes, alfuzosin HCl ER is frequently used in combination therapy. A common combination is with a 5-alpha reductase inhibitor (5-ARI) such as finasteride or dutasteride for men with larger prostates and LUTS. It can also be used in combination with antimuscarinic agents for patients experiencing both voiding and storage symptoms, or with PDE5 inhibitors for men with both BPH and erectile dysfunction.
Citations
[1] U.S. Patent No. 4,535,186. (1985). 2-Amino-5-(2-aminopropyl)-6-chloroquinoline and derivatives thereof.. United States Patent and Trademark Office. [2] U.S. Patent No. 5,716,983. (1998). Extended release pharmaceutical dosage form.. United States Patent and Trademark Office. [3] European Patent EP0608499. (1995). Pharmaceutical compositions.. European Patent Office.
More… ↓
