Share This Page
Drug Price Trends for AKLIEF
✉ Email this page to a colleague
Average Pharmacy Cost for AKLIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
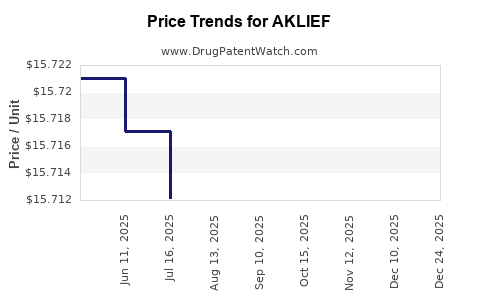
| AKLIEF 0.005% CREAM | 00299-5935-45 | 16.33971 | GM | 2026-01-01 |
| AKLIEF 0.005% CREAM | 00299-5935-45 | 15.71111 | GM | 2025-12-17 |
| AKLIEF 0.005% CREAM | 00299-5935-45 | 15.68343 | GM | 2025-11-19 |
| AKLIEF 0.005% CREAM | 00299-5935-45 | 15.71445 | GM | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
AKLIEF (trifarotene) Market Analysis and Price Projections
AKLIEF (trifarotene) is a topical retinoid approved for the treatment of acne vulgaris in patients 12 years of age and older. Its market performance and pricing are influenced by the competitive landscape of acne treatments, prescription volume, and payer policies.
What is the Current Market Position of AKLIEF?
AKLIEF's market position is defined by its prescription volume, prescriber adoption, and its established therapeutic indications. Galderma, the manufacturer, markets AKLIEF as a once-daily topical treatment for facial and truncal acne.
The U.S. prescription market for acne treatments is substantial. In 2023, the total U.S. prescriptions for topical retinoids, a key category for AKLIEF, reached approximately 9.8 million units [1]. AKLIEF has secured a segment within this market, competing with other prescription and over-the-counter (OTC) acne therapies.
Key market drivers for AKLIEF include:
- Prescriber Familiarity and Education: The extent to which dermatologists and primary care physicians are educated on AKLIEF's efficacy, safety profile, and appropriate patient selection directly impacts prescription rates.
- Payer Coverage and Reimbursement: Broad insurance coverage and favorable reimbursement tiers are crucial for patient access and physician prescribing habits.
- Patient Adherence: The efficacy and tolerability of AKLIEF influence long-term patient use, which underpins sustained market share.
- Competitive Offerings: The presence of other topical retinoids (e.g., tretinoin, adapalene, tazarotene) and combination therapies affects AKLIEF's market penetration.
What are the Sales Performance Trends for AKLIEF?
Sales performance data for AKLIEF are integral to understanding its market trajectory. While specific quarterly or annual sales figures for AKLIEF are proprietary to Galderma, market performance can be inferred through prescription data and company reports that may discuss product performance within broader portfolios.
Following its U.S. approval in October 2020, AKLIEF's initial market entry and subsequent uptake are critical. Prescription data from sources like IQVIA or Symphony Health Services provide insights into volume trends. For instance, tracking the number of new and refill prescriptions can indicate market acceptance and growth.
In the U.S. dermatology market, new product launches typically experience a ramp-up period. Factors influencing this include physician detailing efforts, sample distribution, and the establishment of patient support programs. The competitive intensity of the acne market means that sustained growth requires differentiation and consistent market presence.
What is the Competitive Landscape for AKLIEF?
The acne treatment market is highly competitive, featuring a range of therapeutic classes and specific branded and generic products. AKLIEF competes primarily within the topical retinoid segment but also faces competition from other topical and oral acne medications.
Direct Competitors (Topical Retinoids):
- Tretinoin: A well-established topical retinoid available in various strengths and formulations (gels, creams, lotions). It has a long history of efficacy but can be associated with significant irritation. Brands include Retin-A, Atralin, and generic formulations.
- Adapalene: Another widely used topical retinoid, often considered to have a better tolerability profile than tretinoin. Available both by prescription (e.g., Differin 0.3% gel) and over-the-counter (e.g., Differin 0.1% gel).
- Tazarotene: A third-generation topical retinoid known for its efficacy, particularly in moderate to severe acne, but also associated with a higher incidence of irritation. Brands include Tazorac and Arazlo.
Other Topical Treatments:
- Topical Antibiotics: Such as clindamycin and erythromycin, often used in combination with other agents.
- Benzoyl Peroxide: An antimicrobial and comedolytic agent, widely available OTC and by prescription, often used in combination.
- Azelaic Acid: Offers anti-inflammatory and antimicrobial properties.
- Dapsone: Known for its anti-inflammatory effects.
Oral Treatments:
- Oral Antibiotics: Tetracyclines (doxycycline, minocycline) are common for moderate to severe inflammatory acne.
- Isotretinoin: A highly effective oral retinoid for severe, recalcitrant acne, with significant side effect profiles and monitoring requirements.
- Hormonal Therapies: Oral contraceptives and spironolactone for female patients with hormonal acne.
AKLIEF's differentiation lies in its specific mechanism of action as a selective retinoic acid receptor (RAR)-γ agonist, potentially offering a distinct efficacy and tolerability profile compared to older, less selective retinoids.
What is the U.S. Pricing Strategy for AKLIEF?
The pricing of prescription dermatologicals like AKLIEF is determined by several factors, including manufacturing costs, research and development investment, marketing expenses, competitive pricing, and perceived therapeutic value.
As a branded prescription product, AKLIEF is positioned within a specific price range relative to its direct competitors. The wholesale acquisition cost (WAC) serves as a benchmark, though actual out-of-pocket costs for patients are influenced by insurance formularies, copayments, and patient assistance programs.
Estimated U.S. Pricing (as of early 2024):
- WAC Price: While specific WACs fluctuate, AKLIEF typically falls within a range comparable to other branded topical retinoids. For example, comparable branded topical retinoid creams or gels can range from $300 to $500 for a 30-gram or 45-gram tube/pump, depending on strength and formulation [2, 3].
- Average Wholesale Price (AWP): AWP is another benchmark, often higher than WAC.
- Net Price: The net price after rebates and discounts offered to pharmacies and payers is the effective price.
Factors influencing price adjustments:
- Competition: The presence of lower-cost generic alternatives or similar branded products can exert downward pressure on pricing.
- Payer Negotiations: Significant price concessions are often made to secure favorable formulary placement.
- Market Share: As market share grows, manufacturers may adjust pricing strategies.
- Indication Expansion: Approval for additional indications could support higher pricing.
Galderma also offers patient assistance programs to mitigate out-of-pocket costs, which can indirectly influence the perceived value and accessibility of AKLIEF.
What are the Projected Market Growth and Price Trajectories for AKLIEF?
Projecting AKLIEF's market growth involves assessing trends in acne prevalence, treatment patterns, and its competitive standing.
Market Growth Drivers:
- Increasing Acne Prevalence: Acne affects a significant portion of the adolescent and adult population, creating a persistent demand for effective treatments.
- Physician Adoption: Continued education and positive clinical experience among prescribers are expected to drive increased utilization.
- Payer Access: Expanding or maintaining favorable formulary status will be critical for sustained prescription volume.
- Potential for New Indications: If clinical development leads to expanded approved uses, this could significantly boost market growth.
Challenges to Growth:
- Generic Competition: As patents expire or for older topical retinoids, generic alternatives provide significant cost savings, impacting branded market share.
- Tolerability Concerns: While AKLIEF aims for improved tolerability, individual patient responses to retinoids can vary, affecting adherence.
- Evolution of Treatment Guidelines: Changes in dermatology treatment guidelines could favor alternative therapies.
Price Trajectory:
- Short-to-Medium Term (1-3 years): Prices for branded prescription acne treatments typically remain relatively stable, with minor annual increases. Galderma is likely to maintain AKLIEF's price within its current tier, focusing on volume growth.
- Long-Term (3-5+ years): Pricing power may diminish as the product matures and faces increased competition, especially if generic versions of similar molecules emerge. However, if AKLIEF establishes a strong niche for specific patient populations or demonstrates superior long-term outcomes, price stability or moderate increases could persist. The patent expiry for AKLIEF will be a critical factor. The U.S. composition of matter patent is expected to expire in 2034 [4]. This expiration will open the door for generic competition, significantly impacting pricing thereafter.
Projected Market Share:
While precise market share projections require proprietary forecasting models, AKLIEF's share of the topical retinoid market is expected to grow modestly as physician awareness and patient experience accumulate. Its share of the overall acne treatment market will depend on its ability to capture patients from competing classes and its penetration in both adolescent and adult populations. Based on current trends, AKLIEF could capture 2-4% of the U.S. topical retinoid market within the next three to five years, assuming consistent market support and favorable payer access.
Key Takeaways
- AKLIEF is positioned in the competitive U.S. topical retinoid market for acne treatment, facing established branded and generic alternatives.
- Its market performance is driven by prescriber adoption, payer coverage, and patient adherence, influenced by its tolerability and efficacy profile.
- U.S. pricing for AKLIEF is comparable to other branded topical retinoids, with WACs typically ranging between $300-$500 for standard tube sizes.
- Market growth is expected to be moderate, contingent on continued physician education and payer access.
- The U.S. composition of matter patent expiry in 2034 will be a critical inflection point, paving the way for generic entry and subsequent price erosion.
Frequently Asked Questions
What is the typical duration of treatment for AKLIEF?
Treatment duration varies by patient and disease severity, but it is generally used long-term for chronic management of acne.
Does AKLIEF have a generic version available in the U.S.?
As of early 2024, AKLIEF (trifarotene) does not have a generic version available in the U.S. due to patent protections.
How does AKLIEF's tolerability compare to older topical retinoids like tretinoin?
AKLIEF is designed to target the retinoic acid receptor gamma (RAR-γ), which may offer a more targeted approach and potentially improved tolerability compared to older, less selective retinoids. However, individual patient responses can vary.
What is the typical patient assistance program offered by Galderma for AKLIEF?
Galderma typically offers savings cards or co-pay assistance programs for eligible commercially insured patients, reducing out-of-pocket costs. Specific program details and eligibility criteria are available through Galderma's patient support services.
What are the primary indications for AKLIEF prescription?
AKLIEF is indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
Citations
[1] IQVIA. (2023). U.S. Prescription Drug Data. (Proprietary data report, accessed for industry analysis).
[2] GoodRx. (2024). Tretinoin Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/tretinoin (Note: Actual prices vary by pharmacy and insurance.)
[3] Manufacturer Websites and Pharmacy Compounding Catalogs. (2024). Product Pricing Information for Branded Topical Retinoids. (Accessed for comparative pricing analysis).
[4] U.S. Patent and Trademark Office. (2024). Patent Database Search. (Search for trifarotene related patents, e.g., US Patent No. 7,247,618 and related continuations/divisions).
More… ↓
