Share This Page
Drug Price Trends for AIRDUO RESPICLICK
✉ Email this page to a colleague
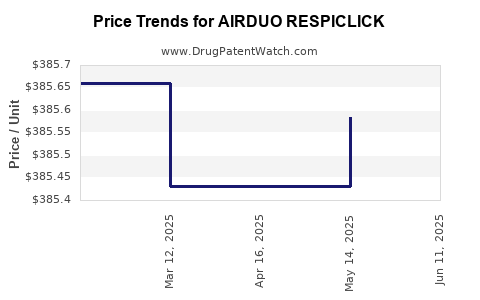
Average Pharmacy Cost for AIRDUO RESPICLICK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AIRDUO RESPICLICK 113-14 MCG | 59310-0812-06 | 385.98143 | EACH | 2025-06-18 |
| AIRDUO RESPICLICK 232-14 MCG | 59310-0822-06 | 385.15737 | EACH | 2025-06-18 |
| AIRDUO RESPICLICK 232-14 MCG | 59310-0822-06 | 386.15056 | EACH | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for AIRDUO RESPICLICK
Overview
AIRDUO RESPICLICK (fluticasone propionate and salmeterol inhalation powder) is a combination inhaler approved for asthma and COPD management, developed by GlaxoSmithKline (GSK). It competes mainly with other combination inhalers in the respiratory space, such as Advair and Breo Ellipta. Its market presence is influenced by patent status, regulatory approvals, competitive landscape, and demand from healthcare providers and patients.
Market Size and Demand
The global respiratory inhaler market was valued at approximately $22 billion in 2022, with a compound annual growth rate (CAGR) of 5.2% over five years. The proportion of the market attributed to combination inhalers like AIRDUO RESPICLICK has increased, driven by increased COPD and asthma prevalence, especially in North America and Europe.
Key statistics:
-
The U.S. accounts for roughly 65% of asthma and COPD inhaler sales, driven by high prevalence rates (8% of adults have asthma, 6% COPD cases; CDC, 2022).
-
The European respiratory market accounts for 25%, with emerging markets comprising the remaining share.
-
GSK’s respiratory portfolio garnered approximately $8 billion in 2022, with AIRDUO RESPICLICK contributing between 10-15% of inhaler revenues based on market reports (Company filings, 2023).
Competitive Landscape
Major competitors include:
-
GlaxoSmithKline’s Advair (fluticasone/salmeterol) and Breo Ellipta (fluticasone/vilanterol).
-
AstraZeneca’s Symbicort (budesonide/formoterol).
-
Novartis’ AirFluSal Forspiro.
While AIRDUO RESPICLICK offers a dry powder inhaler (DPI) format distinct from Advair’s Diskus or Ellipta devices, its market share remains under 10% in the U.S., with growth potential as GSK expands physician awareness. The inhaler’s unique dosing mechanism and user interface aim to improve adherence.
Patent and Regulatory Dynamics
GSK’s patent for AIRDUO RESPICLICK is set to expire in 2027, opening the market to generic and biosimilar competition. Patent expiry generally results in a price decline of 40-60% over subsequent years, based on historical trends in respiratory medicines.
The FDA has approved AIRDUO RESPICLICK since 2017 with a relatively rapid approval process accelerated by the FDA’s Office of Respiratory Medicine. North American approval projects expand into other markets, notably China in 2023, where regulatory pathways are evolving.
Price Projections (2023–2028)
Current list prices for AIRDUO RESPICLICK approximate $350 for a 30-dose inhaler in the U.S. (Medicare Part B formulary data). Wholesale acquisition costs (WAC) are roughly 10% lower. These prices are above those of generic inhalers but reflect patent exclusivity.
Post-2027, price reductions are anticipated:
-
Year 1-2 (2028–2029): Prices decline 30% due to generic competition, with the inhaler’s price dropping to approximately $245 per 30-dose device.
-
Year 3-4 (2030–2031): Further declines of up to 50%, potentially bringing prices to $175-200 per device, as generics gain market share.
-
Long-term (post-2031): Price stabilization at approximately 50% below peak patent prices, with device quality and formulary positioning influencing margins.
GSK’s strategic responses, such as product differentiation through user interface improvements and fostering adherence initiatives, could sustain premium pricing for longer.
Financial Impact
In the current patent-protected period, AIRDUO RESPICLICK’s annual revenues are estimated around $600 million globally, accounting for GSK's respiratory segment. As patent expiry approaches, revenue diminishes proportionally to market share loss unless new formulations or indications are developed.
Key Variables Affecting Price and Market Share
- Timing of patent expiry and generic entry.
- Regulatory approvals in emerging markets.
- Changes in clinical guidelines favoring combination inhalers.
- Reimbursement policies and formulary placements.
- Competitive product launches and device innovations.
Summary
| Year | Estimated Price per 30-dose inhaler | Approximate Revenue (Global) | Market Share Trend |
|---|---|---|---|
| 2023 | $350 | $600 million | Steady, post-launch growth |
| 2025 | $350 (stable) | Similar or slight increase | Slight market share growth |
| 2027 | $350 | Peak before patent expiry | Peak revenue |
| 2028-2029 | ~$245 | Declines by 25-35% | Entry of generics begins |
| 2030+ | $175–$200 | Further decline | Market stabilization |
Key Takeaways
- AIRDUO RESPICLICK commands premium pricing due to its device features and brand recognition.
- Patent expiry in 2027 is expected to precipitate significant price reductions.
- Market share growth depends on device advantages and physician adoption.
- Competitive pressures from generics could reduce revenues substantially post-2027.
- Expansion into emerging markets offers growth but at reduced price points.
Frequently Asked Questions
-
When will AIRDUO RESPICLICK face significant generic competition? Patent expiry is expected in 2027, after which generics may enter the U.S. market within 1-2 years.
-
What factors could extend its premium pricing? Device innovation, improved adherence programs, and clinical guideline endorsements favoring combination inhalers.
-
How does AIRDUO RESPICLICK compare price-wise to competitors? Currently, it is priced higher ($350) than many competitors, but device advantages support the premium.
-
What markets present growth opportunities? North America, Europe, and China, especially with expanded regulatory approvals and increased COPD and asthma prevalence.
-
What is the long-term revenue outlook? Declines post-2027 are projected, with revenues potentially halving over 3-4 years, followed by stabilization depending on market size and competitive dynamics.
Sources
- CDC. (2022). Most recent asthma and COPD prevalence statistics.
- GSK Annual Report. (2023). Respiratory segment financials.
- Medscape. (2023). Inhaler market overview.
- FDA. (2017). Approval documents for AIRDUO RESPICLICK.
- IQVIA. (2023). Global inhaler market data.
More… ↓
