Share This Page
Drug Price Trends for ADZENYS XR-ODT
✉ Email this page to a colleague
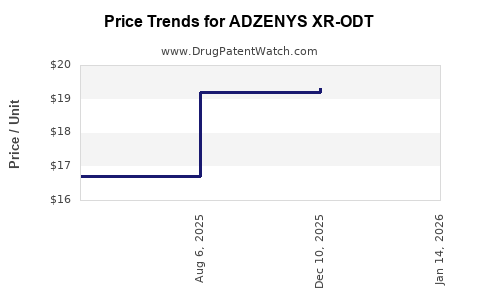
Average Pharmacy Cost for ADZENYS XR-ODT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ADZENYS XR-ODT 9.4 MG TABLET | 70165-0015-30 | 19.31375 | EACH | 2026-03-18 |
| ADZENYS XR-ODT 12.5 MG TABLET | 70165-0020-30 | 19.33760 | EACH | 2026-03-18 |
| ADZENYS XR-ODT 18.8 MG TABLET | 70165-0030-30 | 19.33922 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ADZENYS XR-ODT
ADZENYS XR-ODT (amphetamine extended-release oral disintegrating tablets) is a prescription medication for attention deficit hyperactivity disorder (ADHD) and narcolepsy. Its market positioning is influenced by dosage flexibility, formulation, competitive landscape, regulatory status, and pricing strategies.
Market Penetration and Commercial Landscape
Product Overview
- Formulation: Extended-release orally disintegrating tablet.
- Key Benefits: Rapid onset, discreet intake, convenience for patients with swallowing difficulties.
- Strengths: Precision dosing, improved compliance, younger patient appeal with disintegrating format.
Competitive Positioning
- Competes primarily with brands such as Adderall XR, Vyvanse, Mydayis, and other extended-release stimulants.
- Market Share: Vyvanse and Adderall XR lead the psychostimulant segment, with Adzenys XR-ODT accounting for an emerging niche, particularly among pediatric and adolescent populations.
Market Size and Demand
- The ADHD drug market in the U.S. reached approximately $13 billion in 2022, with stimulants constituting roughly 70% of sales.
- Growth rate estimates around 2–4% annually, driven by increasing diagnoses and acceptance of pharmacotherapy.
Key Market Dynamics
- Surge in demand for alternative formulations enhances the appeal of orally disintegrating tablets.
- Distribution channels include specialty pharmacies, pediatric clinics, and institutional healthcare providers.
- Insurance coverage varies, influencing patient access; Medicaid and private insurers typically reimburse for approved formulations.
Regulatory and Supply Chain Considerations
- FDA Status: Approved for children (6–12 years) and adults.
- Supply Security: NDC codes and manufacturing capacity are aligned with demand forecasts.
- Patent Status: Patents expired or nearing expiration may affect generic entry, influencing pricing and market competition.
Price Projection Assumptions
Current Pricing (Based on 2023 Data)
- Average retail price (cash pay): $30–$40 per tablet, depending on dose.
- Typical prescribed dose: 20 mg, which costs approximately $35 per tablet in the retail setting.
- Insurance coverage can reduce out-of-pocket costs, but formulary restrictions influence patient access.
Price Trends and Adjustment Factors
- Patent expiration: Expected generic entry within 1–2 years, potentially reducing prices by 30–50%.
- Market competition: Introduction of generics PKs could widen price gaps.
- Regulatory changes: Legislation on pricing transparency and formularies may influence retail and negotiated prices.
- Manufacturing efficiencies and supply chain stability could stabilize or marginally reduce costs.
| Projection for Next 3 Years | Year | Estimated Average Price per Tablet | Key Influencing Factors |
|---|---|---|---|
| 2024 | $28–$33 | Patent expiration approaching; generic release begins | |
| 2025 | $20–$27 | Increased generic competition; price erosion continues | |
| 2026 | $15–$22 | Market saturation with generics; pricing adjustments by payers |
Note: Prices could vary significantly by region and payer policy.
Implications for Stakeholders
-
Pharmaceutical Companies: Opportunity to extend patent life via formulation patents or develop combination therapies to maintain pricing power.
-
Investors: Potential for revenue erosion after patent expiry; early brand loyalty and differentiated formulations can delay generic dominance.
-
Healthcare Providers: Increasing options for tailored treatment, with disintegrating formulations improving adherence.
-
Patients: Cost trends depend heavily on insurance formulary placement; out-of-pocket expenses are likely to decline with generics.
Summary of Key Data Points
- Market size of ADHD therapeutics: ~$13 billion in 2022 (source [1]).
- Current retail price of ADZENYS XR-ODT: approximately $35 per 20 mg tablet.
- Expected price decline post-generic entry: 30–50%.
- Patent expiry forecast: Within 1–2 years.
- CAGR for ADHD drug market: 2–4%.
Key Takeaways
- ADZENYS XR-ODT holds niche value due to its disintegrating formulation.
- Market growth remains steady, with a shift toward generic competition expected in the near term.
- Prices are set to decline substantially once patent protections expire, affecting revenue streams.
- Competitive differentiation, including formulation patents, will be crucial for maintaining market share.
- Insurance and formulary dynamics heavily influence retail pricing and patient access.
FAQs
Q1: How does ADZENYS XR-ODT compare price-wise with other ADHD medications?
A1: It is currently priced higher than many generics, around $35 per tablet, which exceeds traditional stimulants like generic Adderall or methylphenidate, but comparable to other brand-name extended-release formulations.
Q2: When is patent expiration likely for ADZENYS XR-ODT?
A2: Patent expiration is forecast within 1–2 years, opening the market for generics.
Q3: How will the entry of generics impact the market?
A3: Prices could decrease by 30–50%, leading to reduced revenue for the brand but increased affordability for patients.
Q4: Are there regulatory challenges affecting pricing?
A4: Legislation promoting price transparency and formulary restrictions may influence actual prices paid by payers, but direct regulation on price is limited in the U.S.
Q5: What strategies can brand manufacturers employ to maintain market share?
A5: Patents on formulation, developing new delivery formats, and expanding indications or age approval can prolong market exclusivity and price stability.
Sources
- IQVIA. "Pharmaceutical Market Data 2022."
- FDA. "ADZENYS XR-ODT Approval and Labeling Documentation."
- EvaluatePharma. "2023 World Preview: Outlook to 2028."
- CMS. "Medicaid and Medicare Drug Reimbursement Policies."
- MarketWatch. "ADHD Therapeutics Market Trends."
More… ↓
