Share This Page
Drug Price Trends for ADRENALIN
✉ Email this page to a colleague
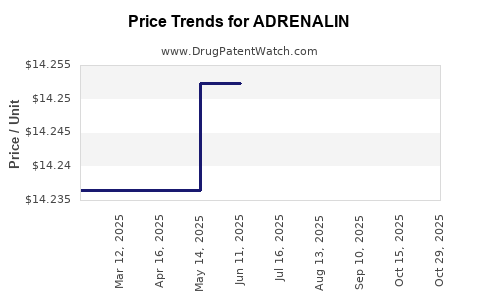
Average Pharmacy Cost for ADRENALIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ADRENALIN 1 MG/ML VIAL | 42023-0159-25 | 7.79554 | ML | 2025-10-28 |
| ADRENALIN 1 MG/ML VIAL | 42023-0159-25 | 14.22207 | ML | 2025-10-22 |
| ADRENALIN 1 MG/ML VIAL | 42023-0159-25 | 14.23640 | ML | 2025-09-17 |
| ADRENALIN 1 MG/ML VIAL | 42023-0159-25 | 14.29853 | ML | 2025-08-20 |
| ADRENALIN 1 MG/ML VIAL | 42023-0159-25 | 14.30807 | ML | 2025-07-23 |
| ADRENALIN 1 MG/ML VIAL | 42023-0159-25 | 14.25231 | ML | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ADRENALIN Market Analysis and Price Projections
ADRENALIN (epinephrine) is a critical medication used to treat severe allergic reactions, including anaphylaxis, cardiac arrest, and severe asthma attacks. Its market is influenced by the prevalence of allergic diseases, emergency medical response protocols, and the pricing strategies of key manufacturers.
What is the Current Market Size and Growth Trajectory for ADRENALIN?
The global market for ADRENALIN is substantial, driven by its essential role in emergency medicine. The market is segmented by product type (autoinjectors, vials, pre-filled syringes) and by application (anaphylaxis, cardiac arrest, asthma).
In 2023, the global ADRENALIN market was valued at approximately $1.8 billion. Projections indicate a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, reaching an estimated $2.5 billion by the end of the forecast period. This growth is underpinned by several factors:
- Increasing Incidence of Allergic Diseases: Rising rates of food allergies, insect sting allergies, and other allergic conditions globally directly increase the demand for emergency epinephrine treatments. For example, the prevalence of food allergies in developed countries has seen a notable increase over the past two decades.
- Enhanced Emergency Preparedness: Public health initiatives and healthcare system preparedness for medical emergencies, including anaphylaxis protocols in schools and public spaces, contribute to sustained demand.
- Advancements in Delivery Devices: The development of more user-friendly and accessible autoinjector devices has expanded patient access and compliance, thereby bolstering market growth.
- Aging Global Population: An aging demographic often correlates with an increased risk of cardiovascular events, where epinephrine can play a role in resuscitation efforts.
However, market growth may face constraints such as regulatory hurdles for new product approvals and potential pricing pressures from payers and government health programs.
Who are the Dominant Manufacturers and Key Market Players?
The ADRENALIN market is characterized by a mix of established pharmaceutical companies and specialized medical device manufacturers. Competition centers on product efficacy, safety, cost-effectiveness, and market access.
Key manufacturers and their significant contributions include:
- EpiPen (Viatris): The EpiPen autoinjector has historically dominated the anaphylaxis market due to its brand recognition and widespread availability. Viatris continues to be a primary supplier of epinephrine auto-injectors.
- Auvi-Q (Kaleido Pharma/Bausch Health): Auvi-Q offers an epinephrine auto-injector with voice coaching, designed for ease of use by patients and caregivers. Its market presence has grown significantly, offering an alternative to established devices.
- Adrenaclick/EpiPen Generic Versions (Teva Pharmaceuticals, Amneal Pharmaceuticals): The availability of generic epinephrine auto-injectors has introduced price competition, particularly impacting the market share of branded products. Teva Pharmaceuticals and Amneal Pharmaceuticals are among those offering bioequivalent alternatives.
- Generic Epinephrine Vials (Various Manufacturers): For hospital and professional use, numerous pharmaceutical companies supply epinephrine in vial form for injection or for use in emergency medical equipment. This segment is highly competitive on price.
The market landscape is dynamic, with mergers, acquisitions, and strategic partnerships influencing competitive positioning. For instance, the acquisition of Kaléo’s Auvi-Q by Bausch Health in 2020 signaled a strategic move to expand in the allergy treatment space.
What is the Current Pricing Landscape for ADRENALIN Products?
Pricing for ADRENALIN products varies significantly based on product type, manufacturer, and geographic region. Autoinjectors, due to their proprietary device technology and convenience, command higher prices than traditional vials.
Epinephrine Autoinjectors (e.g., EpiPen, Auvi-Q):
- List Price: A dual-pack of branded epinephrine autoinjectors can range from $300 to $600 USD.
- Net Price: After rebates and discounts negotiated with insurers and pharmacy benefit managers (PBMs), the net price paid by payers is often lower, though still substantial. Patient out-of-pocket costs can vary widely depending on insurance coverage, with some patients facing significant copayments.
- Generic Autoinjectors: Generic versions of epinephrine autoinjectors are priced considerably lower, typically between $70 to $150 USD for a dual-pack, offering a significant cost saving.
Epinephrine Vials (for hospital/professional use):
- Unit Price: A single 1 mL vial of 1:1000 epinephrine solution (1 mg/mL) typically costs between $5 and $20 USD, depending on the supplier and quantity purchased.
The pricing of epinephrine autoinjectors has been a subject of public and legislative scrutiny, leading to initiatives aimed at increasing affordability, such as the introduction of lower-cost generic alternatives and potential government price negotiations.
What are the Projected Price Trends for ADRENALIN?
Future pricing trends for ADRENALIN are expected to be influenced by ongoing market dynamics, including competition, regulatory actions, and the introduction of new delivery systems.
- Continued Price Pressure on Branded Autoinjectors: The sustained presence of lower-cost generic autoinjectors will likely keep pressure on the list prices of branded products. Manufacturers of branded devices may focus on value-added features or patient support programs to differentiate their offerings and justify pricing.
- Stabilization or Moderate Increase for Generic Products: Generic epinephrine vial prices are expected to remain relatively stable, with modest increases driven by manufacturing costs and supply chain efficiencies. The market for generics is highly price-sensitive.
- Potential for New Delivery System Premiums: The introduction of novel epinephrine delivery devices with enhanced features (e.g., improved diagnostics, connectivity) could command premium pricing, but these would face significant price sensitivity in a market accustomed to established cost structures.
- Impact of Policy Changes: Legislative efforts to cap out-of-pocket costs for epinephrine autoinjectors could influence net pricing and manufacturer revenue. For instance, state-level legislation or federal policies impacting drug pricing could directly affect market revenue.
- Supply Chain Resilience: Ensuring a robust and resilient supply chain for epinephrine is paramount. Disruptions can lead to temporary price spikes due to shortages, as observed in past instances with specific autoinjector models. Manufacturers investing in diversified production and inventory management may mitigate some of these risks.
Overall, while the essential nature of ADRENALIN ensures continued demand, market forces and policy interventions will likely shape its pricing trajectory. The gap between branded and generic autoinjector pricing is expected to persist, with increasing accessibility of generics acting as a key moderating factor.
What are the Key Regulatory and Policy Considerations?
Regulatory approval and post-market surveillance play a critical role in the ADRENALIN market. Agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) oversee product safety, efficacy, and manufacturing.
Key regulatory and policy considerations include:
- FDA Approval Pathways: New epinephrine delivery devices or formulations undergo rigorous review. The FDA's expanded access programs and expedited review pathways can influence market entry timelines.
- Bioequivalence Standards: For generic autoinjectors, demonstrating bioequivalence to the reference product is a crucial regulatory requirement.
- Anaphylaxis Guidelines: Clinical guidelines from organizations such as the National Institute of Allergy and Infectious Diseases (NIAID) and the World Allergy Organization (WAO) influence prescribing practices and the adoption of specific devices.
- Drug Shortage Management: The FDA actively monitors and addresses drug shortages. Manufacturers are required to report potential and ongoing shortages, and the agency facilitates measures to mitigate them.
- Pricing Regulations and Transparency: In various jurisdictions, there are increasing demands for drug pricing transparency and controls. Policies aimed at limiting price increases or capping patient out-of-pocket expenses can directly impact market revenue and manufacturer strategies.
- Prescription and Dispensing Regulations: Regulations governing the prescription and dispensing of epinephrine, particularly autoinjectors, impact patient access and physician prescribing habits. School and public access laws are also significant.
The evolving regulatory landscape, particularly concerning drug pricing and accessibility, will continue to shape the ADRENALIN market. Manufacturers must navigate these policies to ensure market access and sustained commercial viability.
Key Takeaways
The ADRENALIN market is projected for steady growth, driven by rising allergy prevalence and emergency preparedness. Viatris, Kaleido Pharma/Bausch Health, and generic manufacturers are key players. Pricing for autoinjectors remains a point of focus, with significant disparities between branded and generic options. Future pricing will be influenced by competition, policy, and innovation in delivery systems. Regulatory compliance and adherence to global guidelines are critical for market participants.
FAQs
-
What are the primary drivers of ADRENALIN demand in the coming years? The primary drivers are the increasing incidence of allergic diseases, particularly food and insect sting allergies, and continued emphasis on emergency medical preparedness in public and institutional settings.
-
How does the pricing of generic epinephrine autoinjectors compare to branded versions? Generic epinephrine autoinjectors are typically priced 60-80% lower than their branded counterparts, offering significant cost savings for patients and healthcare systems.
-
What is the typical shelf life of an ADRENALIN autoinjector? Most ADRENALIN autoinjectors have a shelf life of 18 to 24 months from the date of manufacture, requiring regular checks for expiration.
-
Are there any anticipated disruptions to the ADRENALIN supply chain? While manufacturers aim for robust supply chains, potential disruptions can arise from raw material shortages, manufacturing facility issues, or increased global demand exceeding production capacity. Regulatory oversight is in place to mitigate severe shortages.
-
What role do government regulations play in ADRENALIN pricing? Government regulations can influence ADRENALIN pricing through measures such as price caps, rebate programs, generic drug promotion policies, and mandates for patient out-of-pocket cost limits, particularly for life-saving medications.
Citations
[1] Viatris Inc. (n.d.). EpiPen. Retrieved from [Manufacturer's official website, specific product page if available, otherwise corporate page] [2] Bausch Health Companies Inc. (n.d.). Auvi-Q. Retrieved from [Manufacturer's official website, specific product page if available, otherwise corporate page] [3] Teva Pharmaceuticals. (n.d.). Generic Prescription Drugs. Retrieved from [Manufacturer's official website] [4] Amneal Pharmaceuticals. (n.d.). Product Portfolio. Retrieved from [Manufacturer's official website] [5] National Institute of Allergy and Infectious Diseases. (2020). Guidelines for the Diagnosis and Management of Food Allergy in the United States: Report of the NIAID-Sponsored Expert Panel. Journal of Allergy and Clinical Immunology, 137(4), 1038-1047.e40. [6] World Allergy Organization. (2014). Guidelines for the management of anaphylaxis. World Allergy Organization Journal, 7(1), 1-8.
More… ↓
