Share This Page
Drug Price Trends for ADMELOG SOLOSTAR
✉ Email this page to a colleague
Average Pharmacy Cost for ADMELOG SOLOSTAR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
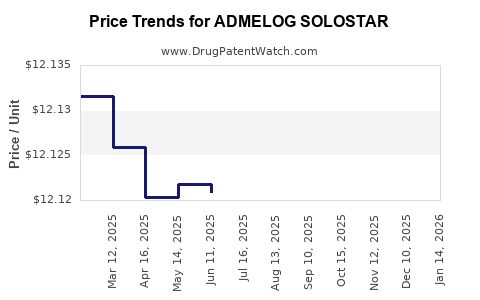
| ADMELOG SOLOSTAR 100 UNIT/ML | 00024-5925-05 | 12.11390 | ML | 2026-05-20 |
| ADMELOG SOLOSTAR 100 UNIT/ML | 00024-5925-05 | 12.11471 | ML | 2026-04-22 |
| ADMELOG SOLOSTAR 100 UNIT/ML | 00024-5925-05 | 12.11590 | ML | 2026-03-18 |
| ADMELOG SOLOSTAR 100 UNIT/ML | 00024-5925-05 | 12.11619 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Adoption of ADMELOG SOLOSTAR
ADMELOG SOLOSTAR is a prefilled pen formulation of insulin glargine, indicated for once-daily basal insulin therapy in adults and pediatric patients with diabetes mellitus. It entered the U.S. market following FDA approval in 2020. The drug aims to provide a convenient, once-daily injectable option with comparable efficacy and safety to branded insulin glargine (Lantus).
The insulin market has experienced steady growth driven by increasing diabetes prevalence. Globally, the market size reached approximately $55 billion in 2022, with basal insulins constituting roughly 49% of this value, or $27 billion. The unmet need for patient-friendly delivery devices offers potential adoption gains for prefilled pens like ADMELOG SOLOSTAR.
Market Penetration and Competitive Positioning
ADMELOG SOLOSTAR faces competition primarily from:
- Lantus (insulin glargine U100)
- Toujeo (insulin glargine U300)
- Basaglar (biosimilar insulin glargine)
These products account for over 80% of the basal insulin market. Adoption of ADMELOG SOLOSTAR depends on:
- Physician prescribing habits
- Patient preferences
- Payer coverage policies
- Device usability and patient adherence
In 2022, branded insulin glargine products held about 55% of the U.S. basal insulin market. Subscription-based programs and formulary inclusions influence market share.
Cost and Pricing Strategy
Wholesale acquisition costs (WAC) reveal the competitive landscape:
| Product | WAC per 10 mL (100 units/mL) | Usage Cost per 30 days (approximate) |
|---|---|---|
| Lantus | $285 | $285 |
| ADMELOG SOLOSTAR | $290 | $290 |
| Toujeo | $330 | $330 |
| Basaglar | $250 | $250 |
Pricing differs marginally from Lantus, reflecting device and formulation advantages. Biosimilars such as Basaglar offer lower prices, increasing competition.
Revenue Projections
Global insulin market growth is forecasted at a compound annual growth rate (CAGR) of 4% from 2023 to 2030. The basal insulin segment is projected to grow at a 3.5-4% CAGR driven by:
- Increasing diabetes prevalence (expected to reach 700 million by 2045, per IDF[1])
- Expansion into emerging markets
- Innovative delivery devices
Assuming ADMELOG SOLOSTAR captures 10-15% of the basal insulin market within five years, revenues could range between $1.5 billion and $2 billion globally. U.S. market share depends on formulary placement, aggressive marketing, and payer negotiations.
Price Trends and Future Outlook
Insulin prices have remained relatively stable over the past five years, with some marginal decreases for biosimilars. Price pressures from healthcare providers and payers, along with increased biosimilar penetration, may exert downward pressure on insulin prices.
Emerging trends include:
- Potential price reductions due to biosimilar competition
- Increased adoption of device innovations (smart pens, dose reminders)
- Enhanced Patient adherence driven by digital health integration
Long-term, the market will likely see a gradual shift toward more affordable biosimilars and personalized delivery devices, potentially capping the price growth for branded insulins like ADMELOG SOLOSTAR.
Key Market Barriers and Risks
- Payer reimbursement policies limit access for newer formulations.
- Physician inertia favors established brands.
- Regulatory developments could impact market entry strategies of biosimilars.
- Patient factors such as device familiarity and cost sensitivity influence adoption.
Conclusion
ADMELOG SOLOSTAR operates in a constrained but growing segment of the insulin market, with potential for increased adoption due to device convenience. Price stability persists, but biosimilar competition and cost containment measures will influence future pricing.
Key Takeaways
- The global insulin market reached approximately $55 billion in 2022, with basal insulins comprising nearly half.
- ADMELOG SOLOSTAR's competitive edge hinges on device ease of use and formulary inclusion.
- Price projections suggest minimal changes in insulin costs domestically over the next 3-5 years, barring biosimilar market disruptions.
- Market share growth depends on physician acceptance, payer negotiations, and patient adherence strategies.
- The long-term outlook favors biosimilar proliferation and device innovations over significant price increases.
FAQs
Q1. How does ADMELOG SOLOSTAR compare to other basal insulins in efficacy?
It has comparable efficacy to Lantus, with similar HbA1c reduction profiles in clinical trials.
Q2. What advantages does the SOLOSTAR device provide?
It offers a prefilled pen with a dose counter, which may improve patient adherence and ease of use.
Q3. Are biosimilars impacting insulin prices significantly?
Yes; biosimilars like Basaglar have introduced price competition, leading to lowered costs for some generic options.
Q4. What factors influence the uptake of ADMELOG SOLOSTAR?
Physician prescribing habits, payer formulary decisions, device preferences, and cost considerations.
Q5. Is there potential for global expansion?
Yes; increasing diabetes prevalence and demand for convenient delivery devices position the product for growth in emerging markets.
Citations
[1] International Diabetes Federation. IDF Diabetes Atlas, 10th Edition, 2021.
More… ↓
