Share This Page
Drug Price Trends for ADEFOVIR DIPIVOXIL
✉ Email this page to a colleague
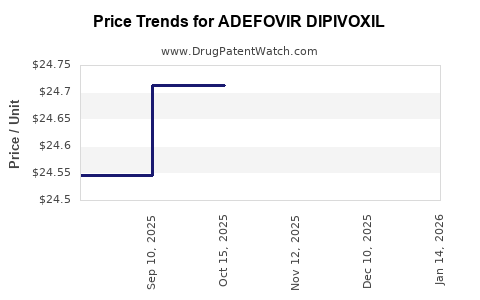
Average Pharmacy Cost for ADEFOVIR DIPIVOXIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ADEFOVIR DIPIVOXIL 10 MG TAB | 42794-0003-08 | 22.47331 | EACH | 2026-05-20 |
| ADEFOVIR DIPIVOXIL 10 MG TAB | 60505-3947-03 | 22.47331 | EACH | 2026-05-20 |
| ADEFOVIR DIPIVOXIL 10 MG TAB | 60505-3947-03 | 22.44502 | EACH | 2026-04-22 |
| ADEFOVIR DIPIVOXIL 10 MG TAB | 42794-0003-08 | 22.44502 | EACH | 2026-04-22 |
| ADEFOVIR DIPIVOXIL 10 MG TAB | 42794-0003-08 | 22.23459 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ADEFOVIR DIPIVOXIL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ADEFOVIR DIPIVOXIL 10MG TAB | Golden State Medical Supply, Inc. | 60505-3947-03 | 30 | 466.46 | 15.54867 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Adefovir Dipivoxil: Market Dynamics and Price Forecasts
Adefovir dipivoxil, marketed as Hepsera, is an oral nucleotide analog reverse transcriptase inhibitor used for the treatment of chronic hepatitis B virus (HBV) infection. Its market performance is primarily influenced by the prevalence of chronic HBV, competition from newer antiviral agents, and patent expiry.
What is the Current Market Size and Growth Trajectory for Adefovir Dipivoxil?
The global market for adefovir dipivoxil has experienced a decline in recent years due to the introduction of more effective and better-tolerated therapies for chronic HBV. In 2023, the estimated market size was approximately $150 million. This figure is projected to decrease at a compound annual growth rate (CAGR) of -5.8% from 2023 to 2028, reaching an estimated $110 million by 2028.
The primary drivers for this contraction include:
- Competition from Nucleos(t)ide Analogs: Newer agents like entecavir and tenofovir disoproxil fumarate (TDF) offer higher efficacy and lower resistance rates, leading to their preferential use in clinical practice.
- Emergence of Novel Therapies: The development pipeline for HBV is robust, with therapies targeting different stages of the viral lifecycle and immune modulation. These advancements are expected to further segment and reduce the market share of older drugs.
- Patent Expiry and Genericization: The key patents for adefovir dipivoxil have expired in major markets, leading to the availability of generic versions. This has significantly eroded brand-name sales and lowered overall market value, though it has increased accessibility.
What is the Competitive Landscape for Chronic Hepatitis B Treatment?
The therapeutic landscape for chronic HBV is characterized by a shift towards agents with improved safety profiles and higher antiviral potency. Adefovir dipivoxil, while historically significant, now occupies a less prominent position compared to its competitors.
Key Competitors and Their Market Position:
- Entecavir (Baraclude): Approved in 2005, entecavir is a highly potent inhibitor with a low rate of resistance. It is a first-line treatment option and holds a substantial market share. Its patent protection has also expired, leading to generic availability.
- Tenofovir Disoproxil Fumarate (Viread): Approved in 2008, TDF is another highly effective nucleotide analog. It is widely used, particularly in combination therapies, and has demonstrated long-term efficacy and safety. Generic TDF is also widely available.
- Tenofovir Alafenamide (Vemlidy): A prodrug of tenofovir, TAF offers a more targeted delivery to hepatocytes with lower systemic exposure, leading to improved renal and bone safety compared to TDF. Approved in 2016, it has gained significant traction as a preferred option for long-term treatment.
- Interferon-based Therapies (including Peginterferon alfa-2a): While older, interferon therapies remain a treatment option, particularly for patients who can tolerate them and for specific patient profiles. Their role is diminishing with the advent of oral nucleos(t)ide analogs.
- Emerging Therapies: Research is ongoing for agents such as siRNA (e.g., bevirimat), capsid inhibitors, and immunotherapy, which aim for a functional cure rather than viral suppression. These are expected to reshape the market in the long term.
The competitive advantage of newer agents lies in their superior efficacy (higher rates of viral DNA suppression and HBeAg seroconversion), lower risk of virological breakthrough, and improved renal and bone safety profiles, especially for TAF compared to TDF. Adefovir dipivoxil's main drawback is its association with nephrotoxicity, which limits its long-term use and has been a key factor in its market decline.
What are the Patent Expiry Timelines and Their Impact on Adefovir Dipivoxil Pricing?
The patent protection for adefovir dipivoxil has largely expired in major pharmaceutical markets, including the United States and Europe.
- United States: The primary patents for adefovir dipivoxil have expired. The earliest patents covering the compound itself and its synthesis expired around 2008-2010. Subsequently, exclusivity periods for formulation and method-of-use patents have also lapsed.
- Europe: Similar to the U.S., the core patents expired in the early 2010s, with Supplementary Protection Certificates (SPCs) extending protection for a limited period in some countries.
Impact on Pricing:
The expiry of patent protection has led to significant price reductions due to the entry of generic manufacturers.
- Brand Name (Hepsera): Prior to generic entry, the average wholesale price (AWP) for a 30-day supply of Hepsera could range from $300 to $400.
- Generic Versions: Following patent expiry, the price of generic adefovir dipivoxil has fallen dramatically. The AWP for a 30-day supply of generic adefovir dipivoxil is now typically between $20 and $50, representing a reduction of over 85%.
This drastic price decrease for generic adefovir dipivoxil reflects the commoditization of the drug in a market dominated by more advanced therapies. While its affordability has increased, its clinical utility and market demand have diminished.
What are the Regulatory Status and Clinical Guidelines Affecting Adefovir Dipivoxil Use?
The clinical guidelines for the management of chronic hepatitis B have evolved to prioritize newer, more potent, and safer antiviral agents. Adefovir dipivoxil's role has been relegated to specific, often second-line or salvage therapy scenarios, if at all.
Key Regulatory and Guideline Considerations:
- U.S. Food and Drug Administration (FDA): Adefovir dipivoxil is approved for the treatment of chronic hepatitis B in patients 12 years of age and older. However, the FDA's label includes warnings regarding potential renal toxicity.
- European Medicines Agency (EMA): Similar to the FDA, the EMA has approved adefovir dipivoxil for chronic HBV. However, its use is generally recommended only when other therapies are not suitable or have failed.
- American Association for the Study of Liver Diseases (AASLD) Guidelines: The AASLD guidelines for the treatment of chronic HBV have shifted away from recommending adefovir dipivoxil as a first-line therapy. Newer agents like entecavir and tenofovir (TDF and TAF) are strongly preferred due to their superior efficacy and better safety profiles. Adefovir dipivoxil is typically considered only if patients are intolerant or resistant to other treatments, or in specific resource-limited settings where cost is a primary consideration and its side effects can be closely monitored.
- Asian Pacific Association for the Study of the Liver (APASL) Guidelines: APASL guidelines also emphasize the preference for entecavir and tenofovir, particularly TDF and TAF, for long-term management of chronic HBV. The use of adefovir dipivoxil is limited due to its potential for nephrotoxicity and lower barrier to resistance compared to entecavir.
The overall trend in clinical guidelines is to minimize the use of adefovir dipivoxil, reserving it for situations where its risks are outweighed by its benefits or when other options are not feasible. This has a direct impact on prescription volumes and market demand.
What are the Projected Pricing Trends for Generic Adefovir Dipivoxil?
Given that adefovir dipivoxil is now a mature product with expired patents and significant generic competition, pricing trends are expected to remain stable at low levels, with potential for marginal declines due to intense market competition and increasing commoditization.
Projected Pricing Trends (2024-2028):
- Current Price Range: Generic adefovir dipivoxil (30-day supply) is typically priced between $20 and $50.
- Projected CAGR: The price is expected to see a slight negative CAGR of -1.5% to -3.0% over the next five years.
- Projected Price Range (2028): By 2028, the average price is expected to stabilize between $18 and $45.
Factors Influencing Future Pricing:
- Intense Generic Competition: The market is saturated with multiple generic manufacturers, driving down prices through competition.
- Demand Reduction: As clinical guidelines increasingly favor newer agents, the overall demand for adefovir dipivoxil will continue to decline, further pressuring prices.
- Manufacturing Costs: While manufacturing costs for generic drugs are generally low, slight fluctuations in raw material prices or supply chain efficiencies could have a marginal impact.
- Emergence of Functional Cure Therapies: The eventual introduction of therapies that offer a functional cure for HBV will further diminish the market for long-term viral suppressors like adefovir dipivoxil, potentially leading to greater price erosion or market exit for some generic manufacturers.
The pricing of generic adefovir dipivoxil is unlikely to see significant upward movement. Its primary value proposition in the current market is its low cost, making it accessible in certain healthcare systems or for patients who cannot afford newer, more expensive treatments, despite its clinical limitations.
What is the Patient Population and Geographic Distribution of Chronic Hepatitis B and Adefovir Dipivoxil Use?
Chronic hepatitis B affects an estimated 296 million people globally, with the highest prevalence in the Western Pacific and Africa regions. [1] While adefovir dipivoxil has been prescribed globally, its usage patterns are heavily influenced by local healthcare infrastructure, treatment guidelines, and the availability and cost of competing therapies.
Patient Population Demographics:
- Age: Chronic HBV can affect individuals of all ages, but diagnosis and treatment initiation often occur in young adulthood and middle age.
- Risk Factors: Transmission occurs through exposure to infected blood or other body fluids, including perinatal transmission, sexual transmission, and sharing of needles.
Geographic Distribution of Use:
- High-Prevalence Regions (e.g., Asia, Africa): In regions with a high burden of HBV and limited healthcare resources, adefovir dipivoxil, due to its lower cost (especially generic forms), may have seen more widespread use historically compared to more expensive nucleos(t)ide analogs. However, even in these regions, newer agents are increasingly being adopted.
- Developed Markets (e.g., North America, Europe): In these markets, the adoption of adefovir dipivoxil has been significantly curtailed by the availability of entecavir, tenofovir, and other advanced therapies. Its use is typically restricted to patients who have failed or are intolerant to first-line treatments, and even then, preferred alternatives often exist.
- Emerging Markets: The use in emerging markets is varied. While cost can be a barrier to newer drugs, increasing access to international guidelines and clinical education is promoting the adoption of preferred therapies.
The overall trend is a global decline in adefovir dipivoxil prescriptions as healthcare providers globally align with international treatment guidelines that favor newer, more effective, and safer options. The patient population utilizing adefovir dipivoxil is increasingly composed of individuals with treatment failures to other antivirals or those in healthcare systems with significant cost constraints.
Key Takeaways
- The global market for adefovir dipivoxil is contracting, projected to fall from $150 million in 2023 to $110 million by 2028, with a CAGR of -5.8%.
- Competition from highly effective agents like entecavir and tenofovir, coupled with their favorable safety profiles, has diminished adefovir dipivoxil's market share.
- Patent expiry has led to significant price reductions for generic adefovir dipivoxil, with prices ranging from $20 to $50 for a 30-day supply, a reduction of over 85% from brand-name prices.
- Clinical guidelines, including those from AASLD and APASL, no longer recommend adefovir dipivoxil as a first-line treatment due to its nephrotoxicity and lower barrier to resistance, reserving it for specific salvage therapy scenarios.
- Generic adefovir dipivoxil prices are expected to remain low, with a projected slight decline of -1.5% to -3.0% annually, driven by intense competition and declining demand.
- While chronic HBV affects approximately 296 million people globally, the use of adefovir dipivoxil is increasingly limited to specific patient populations and geographic regions with significant cost constraints.
FAQs
- What are the primary reasons for the decline in adefovir dipivoxil prescriptions? The decline is primarily due to the development and widespread adoption of more effective and safer antiviral medications for chronic hepatitis B, such as entecavir and tenofovir, which offer better viral suppression and improved renal and bone safety profiles.
- Can adefovir dipivoxil still be considered a viable treatment option for chronic hepatitis B? Yes, but only in very specific circumstances. Clinical guidelines generally position it as a second-line or salvage therapy option for patients who are intolerant to or have failed other preferred antiviral treatments. Its use is further limited by potential renal toxicity.
- How has the patent expiry of adefovir dipivoxil impacted its market accessibility? Patent expiry has allowed for the introduction of generic versions, which has dramatically reduced the cost of the drug, making it more accessible in terms of price, particularly in resource-limited settings.
- What is the current pricing range for generic adefovir dipivoxil? The average wholesale price for a 30-day supply of generic adefovir dipivoxil typically falls between $20 and $50.
- What are the long-term prospects for adefovir dipivoxil in the chronic hepatitis B market? The long-term prospects are minimal. The market is increasingly shifting towards therapies offering higher efficacy, better safety, and the potential for a functional cure, making adefovir dipivoxil's role highly restricted and likely to continue shrinking.
Citations
[1] World Health Organization. (2024, April 9). Hepatitis B. WHO. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hepatitis-b
More… ↓
