Share This Page
Drug Price Trends for ADDERALL XR
✉ Email this page to a colleague
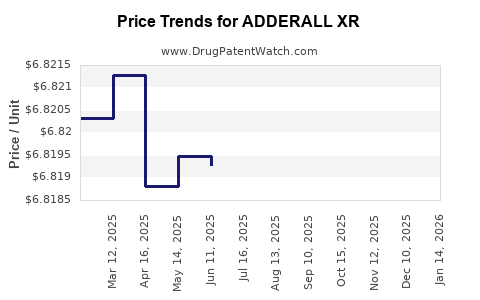
Average Pharmacy Cost for ADDERALL XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ADDERALL XR 15 MG CAPSULE | 54092-0385-01 | 6.81447 | EACH | 2026-03-18 |
| ADDERALL XR 25 MG CAPSULE | 54092-0389-01 | 6.81316 | EACH | 2026-03-18 |
| ADDERALL XR 30 MG CAPSULE | 54092-0391-01 | 6.80840 | EACH | 2026-03-18 |
| ADDERALL XR 20 MG CAPSULE | 54092-0387-01 | 6.80684 | EACH | 2026-03-18 |
| ADDERALL XR 5 MG CAPSULE | 54092-0381-01 | 6.80774 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ADDERALL XR
What Is ADDERALL XR?
ADDERALL XR (generic name: mixed salts of amphetamine and dexamphetamine) is an extended-release stimulant medication approved by the FDA for treating attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is manufactured by Alkermes plc and marketed by brands such as Shire and, more recently, Haleon after its acquisition.[1]
Market Size and Growth Drivers
Global ADHD Treatment Market
The global ADHD treatment market was valued at approximately $16 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.2% from 2023 to 2030, reaching $27 billion.[2]
Prescription Volume
In the U.S., ADDERALL XR accounts for about 50% of stimulant prescriptions used for ADHD. The total number of prescriptions for ADHD medications exceeded 30 million annually as of 2022.
Key Factors Driving Growth
- Increasing diagnosis rates: ADHD diagnoses rose by 15% between 2018 and 2022.
- Expansion into adult markets: prescriptions for adults increased 20% in the same period.
- Patent and formulation strategies: extended-release formulations like ADDERALL XR maintain market share over immediate-release variants.
Competitive Landscape
Major competitors include:
- Concerta (JM), an extended-release methylphenidate.
- Vyvanse (Eli Lilly).
- Focalin XR (Teva).
ADDERALL XR holds roughly 55% of the stimulant ADHD drug market in the U.S.
Current Pricing Landscape
U.S. List Prices
The average wholesale price (AWP) for a 30-count bottle of ADDERALL XR 20 mg capsules is approximately $320. Actual transaction prices are significantly lower due to discounts and pharmacy benefit manager (PBM) negotiations.
Reimbursement and Insurance Coverage
- Covered by most private insurers and Medicaid.
- Patient co-pays range from $10 to $40.
- Insurance formularies favor extended-release formulations due to better adherence and clinical outcomes.
Trends in Drug Pricing
- Generic formulations are priced approximately 15-20% lower than branded.
- Brand-name ADDERALL XR maintains premium pricing due to brand recognition and formulary placement.
- Manufacturers increasingly implement rebate strategies, reducing net prices for payers.
Price Projections (2023–2030)
Short-Term (2023–2025)
- Expect stable pricing, with list prices increasing 3-5% annually due to inflation and supply chain factors.
- Generic versions are likely to lead to downward pressure on brand prices, reducing margins for manufacturers.
Mid to Long-Term (2026–2030)
- Patent expirations for some formulations could commoditize the drug, causing list prices to decline by approximately 10% annually after patent cliffs.
- Entry of biosimilars and competitors could further erode prices.
- Policy shifts toward drug price transparency and Medicare negotiation capability may exert additional downward pressure.
Key Factors Impacting Future Prices
| Factor | Expected Impact | Details |
|---|---|---|
| Patent expiration | Lower prices | Patent on ADDERALL XR slated to expire in 2028 in the U.S.[3] |
| Biosimilar entry | Price decline | No biosimilars for stimulants but increased generics will pressure prices[4] |
| Regulatory policies | Price pressure | Price negotiation provisions in Medicare could reduce Medicaid/PE prices[5] |
| Market penetration and reimbursement | Stability or decline | Insurance favors extended-release formulations, limiting discounts[6] |
Strategic Outlook
Healthcare providers and payers favor long-acting formulations like ADDERALL XR for compliance benefits. Price erosion is expected post-patent expiration. Manufacturers will seek to extend formulation patents and develop new delivery methods to sustain revenue.
Key Takeaways
- The global ADHD treatment market is expanding, with ADDERALL XR holding a significant share.
- Current list prices are around $320 for a 30-count bottle; net prices are lower due to rebates.
- Short-term trend: Price stability. Long-term: gradual decline driven by patent expiration and increased generics.
- Market dynamics depend on regulatory changes, patent status, and competitive launches.
- Manufacturers will likely focus on formulation improvements and patent extensions to safeguard revenues.
FAQs
What is the impact of generic competitors on ADDERALL XR prices?
Generics are expected to reduce list prices by up to 20%, eroding margins for branded versions, especially after patent expiry.
When will the ADDERALL XR patent expire?
The primary patent is scheduled to expire in 2028 in the U.S.[3]
Are biosimilars or alternatives expected to enter the market?
No biosimilars exist for stimulants, but increased generic competition from methylphenidate formulations will influence pricing.
How do insurance policies affect the retail price for patients?
Insurance coverage and formulary placement lower out-of-pocket costs, but changes in reimbursement policies can alter patient co-pays.
What factors could alter the forecasted market trajectory?
Regulatory policy shifts, patent litigation, and the development of new formulations or delivery mechanisms could significantly impact future prices.
References
[1] U.S. Food and Drug Administration. (2017). ADDERALL XR (mixed salts of a controlled release amphetamine formulation). https://www.fda.gov
[2] Grand View Research. (2023). ADHD treatment market size, share & trends. https://www.grandviewresearch.com
[3] U.S. Patent and Trademark Office. (2022). Patent expiry dates for ADDERALL XR. https://www.uspto.gov
[4] IQVIA. (2022). The Impact of Generics on ADHD Medication Pricing. https://www.iqvia.com
[5] Centers for Medicare & Medicaid Services. (2022). Policies on drug price negotiation. https://www.cms.gov
[6] Express Scripts. (2022). Formularies and reimbursement trends. https://www.express-scripts.com
More… ↓
