Share This Page
Drug Price Trends for ACETYLCYSTEINE
✉ Email this page to a colleague
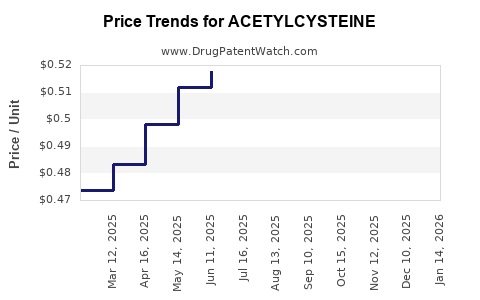
Average Pharmacy Cost for ACETYLCYSTEINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ACETYLCYSTEINE 10% VIAL (NOT FOR INJECTION) | 42494-0435-25 | 2.55690 | ML | 2026-05-20 |
| ACETYLCYSTEINE 10% VIAL (NOT FOR INJECTION) | 00517-7504-25 | 2.55690 | ML | 2026-05-20 |
| ACETYLCYSTEINE 20% VIAL (NOT FOR INJECTION) | 70069-0020-03 | 0.70085 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ACETYLCYSTEINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ACETADOTE 20% SOLN,INHL,30ML | Cumberland Pharmaceuticals, Inc. | 66220-0207-30 | 4X30ML | 150.75 | 2023-02-15 - 2028-02-14 | FSS | ||
| ACETADOTE 20% SOLN,INHL,30ML | Cumberland Pharmaceuticals, Inc. | 66220-0207-30 | 4X30ML | 150.75 | 2024-01-01 - 2028-02-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Acetylcysteine Market Analysis and Price Projections
Acetylcysteine (NAC) is a mucolytic agent and antioxidant used to treat acetaminophen overdose and as a pulmonary medication for respiratory conditions. Its therapeutic applications, coupled with increasing demand in niche markets, drive current and future market dynamics. Global market revenue for NAC was approximately $2.0 billion in 2023, with projections indicating a compound annual growth rate (CAGR) of 4.5% through 2030.
What is the current market size and key drivers for acetylcysteine?
The global acetylcysteine market reached an estimated $2.0 billion in 2023. Key drivers include:
- Growing prevalence of respiratory diseases: Chronic obstructive pulmonary disease (COPD), bronchitis, and cystic fibrosis contribute significantly to NAC demand for airway clearance and mucus reduction. The World Health Organization (WHO) estimates COPD affects 251 million people globally [1].
- Increasing incidence of acetaminophen overdose: Acetaminophen is a common over-the-counter pain reliever, and its misuse or accidental overdose leads to significant demand for NAC as an antidote. Liver damage from acetaminophen overdose is a leading cause of acute liver failure in many developed nations.
- Expanding research and development in new indications: NAC's antioxidant properties are being investigated for various conditions, including psychiatric disorders, neurodegenerative diseases, and certain viral infections, potentially broadening its therapeutic scope.
- Rising demand in emerging economies: Increased healthcare spending, improved access to medical facilities, and growing awareness of NAC's benefits in countries in Asia-Pacific and Latin America are contributing to market expansion.
- Availability of generics: The presence of multiple generic manufacturers leads to competitive pricing and wider accessibility.
Which therapeutic areas represent the largest demand for acetylcysteine?
The primary therapeutic areas driving demand for acetylcysteine are:
- Pulmonary medicine: This segment constitutes the largest share of the NAC market. It encompasses treatments for:
- Chronic Obstructive Pulmonary Disease (COPD): NAC helps to reduce the viscosity of mucus and protect lung tissue from oxidative damage in COPD patients.
- Bronchitis: Both acute and chronic bronchitis benefit from NAC's mucolytic action.
- Cystic Fibrosis (CF): NAC is a crucial component in managing CF by thinning mucus secretions in the lungs, improving airway patency.
- Pneumonia: It can be used as an adjunct therapy to help clear secretions.
- Hepatology (Acetaminophen Overdose): This is a critical indication where NAC acts as a life-saving antidote. The demand is directly linked to the incidence of acetaminophen toxicity, particularly in regions with high over-the-counter analgesic use.
- Ophthalmology: NAC eye drops are used to treat dry eye syndrome and other ocular surface conditions due to their antioxidant and tear-stimulating properties.
- Psychiatry and Neurology: Emerging research is exploring NAC's potential in treating conditions such as obsessive-compulsive disorder (OCD), depression, schizophrenia, and Parkinson's disease, driven by its role in modulating glutamate and antioxidant pathways.
What is the competitive landscape and key players in the acetylcysteine market?
The acetylcysteine market is characterized by a mix of established pharmaceutical companies and specialized generic manufacturers. Key players include:
- Mylan N.V. (now Viatris Inc.): A significant supplier of generic NAC formulations.
- Teva Pharmaceutical Industries Ltd.: Offers a range of generic NAC products.
- Sanofi S.A.: Holds a strong presence with its branded formulations, particularly for respiratory indications.
- Grünenthal GmbH: A key player with a focus on pain management and related therapies, including NAC.
- CVS Health Corporation: A major retailer and healthcare provider that also manufactures and distributes generic pharmaceuticals, including NAC.
- Merck & Co., Inc.: While not a primary NAC manufacturer, its portfolio of respiratory and liver-related treatments influences the broader therapeutic landscape.
- Hikma Pharmaceuticals PLC: Active in the generics market across multiple geographies.
Competition is largely driven by pricing, product availability, and the development of differentiated formulations (e.g., oral powders, intravenous solutions, nebulizer solutions). The patent expiry of originator products has led to a highly competitive generics market.
What are the price trends and projections for acetylcysteine?
Acetylcysteine pricing varies significantly based on formulation, dosage, region, and whether it is a branded or generic product.
- Intravenous (IV) NAC: Used primarily for acetaminophen overdose, IV NAC is typically the most expensive form. Prices can range from $50 to $250 per 200 mg/mL vial, depending on the supplier and quantity. Bulk hospital purchasing may reduce per-unit costs.
- Oral NAC (Powders and Capsules): Used for pulmonary conditions and as a dietary supplement.
- Powders (e.g., 600 mg sachets): Prices can range from $0.50 to $3.00 per sachet, with bulk purchases offering lower per-unit costs.
- Capsules (e.g., 500 mg, 600 mg): A bottle of 100 capsules can cost between $10 and $40, averaging $0.10 to $0.40 per capsule.
- Nebulizer Solutions: For respiratory administration, these typically cost between $2.00 and $10.00 per 3 mL ampule.
Price Projections:
- Stable to Moderate Decline in Generics: The generic market, which dominates NAC supply, is expected to see stable to a modest price decline (0-2% annually) due to ongoing competition and manufacturing efficiencies.
- Potential Price Increases for Specialized Formulations: Any new, patented formulations or novel delivery systems for emerging indications could command premium pricing. However, these represent a small fraction of the current market.
- Regional Price Disparities: Prices will continue to vary across regions, with higher costs in North America and Europe compared to Asia-Pacific and Latin America, reflecting differences in regulatory environments, healthcare infrastructure, and market maturity.
- Impact of Raw Material Costs: Fluctuations in the cost of raw materials, such as cysteine derivatives, could introduce minor price volatility, though this is generally a less significant factor for high-volume generics like NAC.
- Increased Demand in Specific Geographies: As demand grows in emerging markets, initial pricing may be higher due to supply chain development and import costs, potentially converging with global averages over time.
The overall projected trend for the bulk of the acetylcysteine market (generics) is stable pricing with potential minor downward pressure, while niche or novel applications might see higher price points.
What is the regulatory landscape and its impact on the acetylcysteine market?
The regulatory landscape for acetylcysteine is governed by national health authorities like the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others globally. Key aspects include:
- Drug Approval Processes: NAC products for specific therapeutic indications require stringent clinical trials and regulatory review for safety and efficacy. Generic drug approval relies on demonstrating bioequivalence to the reference product.
- Pharmacopeial Standards: NAC must meet the purity and quality standards set by official pharmacopeias (e.g., United States Pharmacopeia - USP, European Pharmacopoeia - Ph. Eur.).
- Manufacturing Standards (GMP): Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure product quality, consistency, and safety. Regulatory inspections ensure compliance.
- Labeling and Indication Restrictions: Approved indications and labeling are strictly regulated. Off-label use, while common in clinical practice, is not promoted by manufacturers.
- Dietary Supplement vs. Pharmaceutical: In some regions, NAC is also available as a dietary supplement. The regulatory framework for supplements is generally less stringent than for pharmaceuticals, impacting quality control and marketing claims.
- Intellectual Property: The patent landscape for original NAC formulations has largely expired, leading to widespread generic competition. Any new patents would likely relate to novel delivery methods, specific combination therapies, or new therapeutic uses, which could temporarily impact pricing for those specific applications.
The regulatory environment ensures product safety and efficacy, but also creates barriers to entry for new manufacturers. The generic nature of most NAC products means that regulatory hurdles primarily concern manufacturing quality and bioequivalence, rather than novel drug development for core indications.
What are the future growth opportunities and challenges for acetylcysteine?
Growth Opportunities:
- Expansion into Novel Indications: Further clinical validation and regulatory approval for NAC's use in psychiatric disorders (e.g., addiction, depression, bipolar disorder), neurodegenerative diseases (e.g., Parkinson's, Alzheimer's), and as an adjunct in treating viral infections (e.g., influenza, COVID-19) could significantly expand the market.
- Development of Advanced Delivery Systems: Innovative formulations such as sustained-release oral preparations, targeted lung delivery systems, or improved nasal sprays could enhance patient compliance and therapeutic outcomes, creating new market segments.
- Growth in Animal Health: NAC's antioxidant and mucolytic properties are being explored for veterinary applications, potentially opening a new market vertical.
- Increased Awareness and Preventative Healthcare: As public awareness of the benefits of antioxidants and preventative health measures grows, NAC's use as a supplement could see incremental growth.
- Emerging Market Penetration: Continued economic development and healthcare infrastructure improvements in Asia-Pacific, Latin America, and Africa will drive demand for essential medicines like NAC.
Challenges:
- Intense Generic Competition: The highly competitive nature of the generic NAC market exerts continuous downward pressure on prices, limiting profit margins for manufacturers.
- Limited Differentiation for Core Indications: For established uses like acetaminophen overdose and basic mucolytic therapy, differentiation between products is minimal, leading to price-based competition.
- Regulatory Hurdles for New Indications: Pursuing regulatory approval for novel therapeutic uses requires substantial investment in clinical trials, with no guarantee of success.
- Stringent Quality Control Requirements: Maintaining consistent quality and adhering to GMP standards across global supply chains can be challenging and costly.
- Alternative Treatments: The development of superior or more convenient treatment options for respiratory diseases or liver conditions could pose a threat to NAC's market share.
- Public Perception and Misinformation: As a widely available supplement, NAC can be subject to misinformation regarding its efficacy and safety, potentially impacting consumer choices.
Key Takeaways
- The acetylcysteine market is valued at approximately $2.0 billion, driven by pulmonary diseases and acetaminophen overdose treatment.
- Growth is projected at 4.5% CAGR through 2030, fueled by increasing respiratory illness prevalence and R&D into new applications.
- Pulmonary medicine represents the largest demand segment, followed by hepatology.
- The market is characterized by strong generic competition from companies like Viatris and Teva.
- Pricing varies significantly by formulation, with IV NAC being the most expensive. Stable to moderate price declines are anticipated for generics.
- Regulatory approval for new indications and advanced delivery systems presents significant growth opportunities.
- Intense price competition and regulatory hurdles for novel uses are key challenges.
FAQs
-
What is the primary difference in regulatory oversight between pharmaceutical-grade acetylcysteine and NAC sold as a dietary supplement? Pharmaceutical-grade NAC requires rigorous clinical trials for safety and efficacy, adheres to strict pharmacopeial standards, and is manufactured under Good Manufacturing Practices (GMP). NAC sold as a dietary supplement typically faces less stringent regulatory requirements, focusing primarily on ingredient safety and labeling accuracy rather than proven therapeutic claims or clinical efficacy for specific diseases.
-
How does the patent expiry of originator acetylcysteine products impact current market pricing? The expiration of patents for originator NAC products has led to the entry of numerous generic manufacturers. This increased competition significantly lowers prices, making NAC widely accessible and affordable, and shifting market focus to manufacturing efficiency and cost reduction rather than product innovation for established indications.
-
What are the key cost components influencing the price of acetylcysteine manufacturing? The primary cost drivers include the procurement of raw materials, such as L-cysteine derivatives, energy costs for synthesis and purification, labor, adherence to Good Manufacturing Practices (GMP) for quality control and facility maintenance, packaging materials, and regulatory compliance expenses, including drug master file submissions and periodic inspections.
-
Can acetylcysteine's potential use in treating psychiatric disorders lead to significant price increases for the drug? If NAC receives regulatory approval for new psychiatric indications, companies developing and marketing these specific formulations could introduce premium-priced products. However, the overall market price of NAC for existing indications, particularly generics, is unlikely to be significantly affected by these niche, higher-priced applications.
-
What is the projected impact of emerging market growth on global acetylcysteine demand and pricing? The growing healthcare infrastructure and increased purchasing power in emerging economies are expected to drive higher volume demand for acetylcysteine. Initially, pricing in these markets might reflect import costs and localized distribution challenges. Over time, as supply chains mature and local manufacturing increases, prices are likely to converge towards global averages, potentially contributing to overall market value growth without necessarily driving up average global prices significantly due to the dominant role of generics.
Citations
[1] World Health Organization. (2023). Chronic obstructive pulmonary disease (COPD). Retrieved from https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
More… ↓
