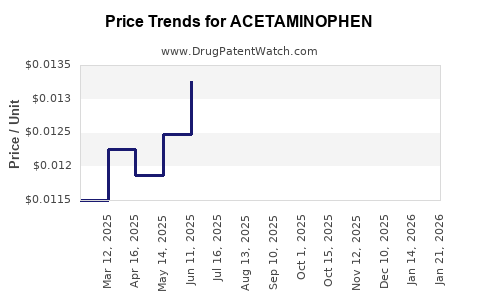
Last updated: February 19, 2026
Acetaminophen, a widely used over-the-counter (OTC) analgesic and antipyretic, exhibits a stable global market driven by consistent demand and broad accessibility. Patent expirations for key formulations have led to a fragmented generic landscape, impacting price dynamics and fostering competition. Future price trajectories are anticipated to be influenced by raw material costs, manufacturing efficiencies, and evolving regulatory environments.
What is the current global market size for acetaminophen?
The global acetaminophen market was valued at approximately USD 3.2 billion in 2023 [1]. This figure reflects the aggregate value of all acetaminophen-based products sold worldwide, encompassing various dosage forms and strengths. The market is characterized by its significant volume, underscoring acetaminophen's role as a cornerstone medication for pain and fever management.
Which factors drive demand for acetaminophen?
Several key factors underpin the sustained demand for acetaminophen:
- Ubiquitous Pain and Fever Management: Acetaminophen is the first-line recommendation for managing mild to moderate pain and fever across diverse patient populations, including children and pregnant women, where other analgesics may be contraindicated [2]. This broad applicability ensures consistent, high-volume sales.
- Accessibility and Affordability: As a widely available OTC medication, acetaminophen is easily accessible to consumers globally. Its generic status further contributes to its affordability, making it a cost-effective choice for individuals and healthcare systems.
- Established Safety Profile: Decades of clinical use have established acetaminophen's favorable safety profile when used at recommended dosages. This contributes to consumer confidence and physician recommendation [3].
- Growing and Aging Global Population: The general increase in global population and the demographic shift towards aging populations, which are often associated with higher incidences of chronic pain, contribute to a continuously expanding consumer base for analgesics like acetaminophen.
- Expansion of Healthcare Access: In emerging markets, increasing access to healthcare services and a growing awareness of pain management options are driving up demand for essential medications, including acetaminophen.
What is the competitive landscape for acetaminophen?
The competitive landscape for acetaminophen is largely dominated by generic manufacturers. The expiration of primary patents for acetaminophen formulations many years ago has opened the market to numerous producers, leading to intense price competition. Key characteristics include:
- Generic Dominance: The market is saturated with generic acetaminophen products from a multitude of pharmaceutical companies. This has suppressed price increases and focused competition on manufacturing efficiency and distribution networks.
- Key Players: While specific market share figures fluctuate and are often proprietary, major global pharmaceutical and contract manufacturing organizations involved in acetaminophen production include but are not limited to:
- GlaxoSmithKline (GSK) (Historically significant, though generics have fragmented market)
- Johnson & Johnson (Through its consumer health divisions)
- McNeil Consumer Healthcare (A subsidiary of J&J, historically a major producer)
- Bayer AG (Offers acetaminophen-containing products)
- Sanofi S.A. (Involved in various OTC analgesics)
- Numerous generic manufacturers in North America, Europe, and Asia, including Perrigo Company plc, Tevea Healthcare, and manufacturers in China and India which are significant sources of active pharmaceutical ingredients (APIs) and finished products.
- Product Differentiation: While the API is largely commoditized, differentiation occurs through branding of OTC products, formulation variations (e.g., extended-release, liquid suspensions, combination products with other active ingredients), packaging, and distribution channels.
- Price Sensitivity: The generic nature of the market makes it highly price-sensitive. Procurement decisions by large distributors and pharmacy chains often prioritize cost-effectiveness.
What are the key regions for acetaminophen consumption?
The consumption of acetaminophen is distributed globally, with significant demand concentrated in the following regions:
- North America (United States and Canada): This region represents one of the largest markets due to its high disposable income, established healthcare infrastructure, and widespread consumer familiarity with OTC medications.
- Europe: Western European countries, with their aging populations and robust healthcare systems, are major consumers. Eastern European markets are also showing increasing demand as healthcare access improves.
- Asia Pacific: This region is experiencing rapid growth, driven by large populations, increasing healthcare awareness, and rising disposable incomes, particularly in countries like China, India, and Southeast Asian nations.
- Latin America: Demand is growing due to increasing population, improving economic conditions, and greater accessibility to pharmacies and basic healthcare.
- Middle East and Africa: While currently a smaller market share, this region presents significant future growth potential driven by population expansion and developing healthcare infrastructure.
What are the primary cost drivers for acetaminophen manufacturing?
The manufacturing costs for acetaminophen are influenced by several factors:
- Raw Material Costs: The primary raw material for acetaminophen synthesis is para-aminophenol (PAP), which is derived from phenol. Fluctuations in the price of phenol, driven by petrochemical market dynamics, directly impact PAP and, consequently, acetaminophen production costs [4]. Other reagents and solvents also contribute to raw material expenses.
- Energy Costs: Chemical synthesis is an energy-intensive process. Global energy price volatility (electricity, natural gas) significantly affects manufacturing overhead.
- Labor Costs: Labor expenses, including skilled chemical engineers, technicians, and production line workers, vary by geographic location of manufacturing facilities.
- Regulatory Compliance: Adhering to Good Manufacturing Practices (GMP), environmental regulations, and quality control standards incurs significant costs related to facility maintenance, testing, documentation, and personnel training.
- Capital Investment: Maintaining and upgrading manufacturing facilities, including specialized reactors, purification equipment, and packaging lines, requires substantial capital expenditure.
- Supply Chain and Logistics: Efficient and cost-effective sourcing of raw materials and distribution of finished products are critical. Disruptions in the global supply chain, transportation costs, and tariffs can impact overall manufacturing expenses.
What is the outlook for acetaminophen API pricing?
Acetaminophen Active Pharmaceutical Ingredient (API) pricing is expected to remain relatively stable, with incremental increases driven by a combination of factors. The market is highly competitive, with major production concentrated in regions offering cost advantages.
Current API Price Range (Indicative): USD $2.50 - $4.50 per kilogram for bulk acetaminophen API. (This range can vary significantly based on purity, volume, supplier, and contract terms.)
Projected Price Movement (Next 3-5 Years):
- Low to Moderate Growth (1-3% annually): The primary upward pressure on API prices will stem from gradual increases in raw material costs, particularly phenol and its derivatives. Energy price fluctuations and tightening environmental regulations in key manufacturing regions may also contribute to modest cost inflation.
- Competitive Pressure: The presence of numerous large-scale API manufacturers, especially in Asia, will continue to exert downward pressure on prices, limiting significant price hikes. Overcapacity in certain segments of the API market may also arise.
- Manufacturing Efficiencies: Continuous improvements in synthesis processes and economies of scale achieved by major producers can help offset some cost increases, thereby stabilizing or slightly moderating price rises.
- Geopolitical and Supply Chain Factors: Unexpected geopolitical events, trade disputes, or significant supply chain disruptions could lead to temporary price spikes. However, the diverse manufacturing base for acetaminophen API generally provides resilience against widespread, prolonged shortages.
What is the outlook for finished acetaminophen product pricing?
Pricing for finished acetaminophen products, particularly in the OTC segment, is expected to follow a pattern of stability with localized fluctuations influenced by brand positioning, competition, and promotional activities.
Current OTC Product Pricing (Indicative):
- Generic Tablets (e.g., 500mg, 100-200 count bottle): USD $0.05 - $0.15 per tablet
- Branded Products (e.g., Tylenol): USD $0.10 - $0.25 per tablet (Premium pricing for brand recognition and marketing)
- Specialty Formulations (e.g., extended-release, combination products): Higher price points, often 2-3 times that of standard generic tablets.
Projected Price Movement (Next 3-5 Years):
- Stable to Slightly Increasing (0-2% annually for generics): Generic finished product pricing will largely be dictated by the stable API costs and intense competition among generic manufacturers and private label brands. Incremental increases will be tied to manufacturing, packaging, and distribution costs.
- Branded Product Pricing: Branded products may see slightly higher, though still modest, price increases (1-3% annually) due to brand equity, marketing investments, and potential for product line extensions or improved formulations.
- Retailer Influence: Large retail chains and pharmacy benefit programs will continue to exert significant pressure on pricing through private label programs and preferred supplier agreements, keeping generic prices highly competitive.
- Combination Products: Products combining acetaminophen with other APIs (e.g., decongestants, antihistamines, opioids) will be subject to the pricing dynamics of all active ingredients and may see more varied price movements based on the availability and cost of these other components.
- E-commerce Impact: The growth of e-commerce for pharmaceuticals may introduce new pricing strategies and increased price transparency, potentially moderating overall price increases for widely available products.
What are potential future challenges for the acetaminophen market?
Despite its strong position, the acetaminophen market faces several potential challenges:
- Regulatory Scrutiny and Safety Concerns: While generally safe, ongoing vigilance regarding acetaminophen's potential for liver toxicity when taken in excess of recommended doses or in combination with alcohol remains a subject of regulatory review and public health campaigns. Stricter labeling requirements or dosage restrictions could impact market dynamics [5].
- Raw Material Supply Chain Volatility: Dependence on specific chemical precursors and manufacturing hubs for APIs creates vulnerability to disruptions. Trade policies, environmental crackdowns in key producing nations, or global health crises can affect the consistent supply and cost of raw materials.
- Competition from Alternative Analgesics: While acetaminophen holds a dominant position for its specific applications, advancements in non-opioid pain management and increased use of other OTC or prescription alternatives for specific types of pain could gradually erode market share in niche areas.
- Counterfeit Products: The global nature of pharmaceutical supply chains, particularly for high-volume OTC drugs, presents a persistent risk of counterfeit products entering the market. This poses a significant threat to patient safety and brand integrity.
- Environmental Sustainability: Increasing pressure on pharmaceutical manufacturers to adopt more sustainable manufacturing processes and reduce their environmental footprint could necessitate investments in new technologies and practices, potentially impacting production costs.
Key Takeaways
Acetaminophen remains a stable and essential global pharmaceutical commodity. Its market is characterized by high volume, broad accessibility, and a competitive generic landscape. Price projections indicate sustained stability for both API and finished products, with modest upward pressures from raw material and operational costs. Regulatory oversight and supply chain resilience are critical factors influencing future market dynamics.
Frequently Asked Questions
What is the primary active ingredient in acetaminophen?
The primary active ingredient is acetaminophen (also known as paracetamol).
Are there any significant patent cliffs remaining for standard acetaminophen formulations?
No, patents for basic acetaminophen formulations expired decades ago, leading to the current generic market.
How does acetaminophen compare to ibuprofen in terms of market share?
While both are leading OTC analgesics, acetaminophen generally holds a larger market share globally due to its broader applicability across age groups and during pregnancy, as well as its established safety profile for individuals with certain gastrointestinal conditions.
What is the typical shelf life of acetaminophen products?
The typical shelf life for acetaminophen tablets and capsules is between 2 to 5 years from the date of manufacture, depending on the specific formulation and packaging. Liquid suspensions may have shorter shelf lives.
What are the main risks associated with taking too much acetaminophen?
The primary risk associated with exceeding the recommended dosage of acetaminophen is severe liver damage, which can be fatal. Other potential side effects, though less common at therapeutic doses, can include kidney problems and skin reactions.