Last updated: February 13, 2026
What is ABSORICA LD and How is it Positioned in the Market?
ABSORICA LD is a formulation of isotretinoin, marketed by Sun Pharmaceutical Industries. It targets severe acne patients who require systemic retinoid therapy. The drug is designed to improve absorption and reduce the gastrointestinal side effects associated with traditional isotretinoin formulations.
Major competitors include Accutane (established brand), Claravis (AbbVie), and Amnesteem (Mayne Pharma), with generics increasingly gaining market share.
What Are the Recent Sales and Market Trends for ABSORICA LD?
Market data indicates rising demand for retinoids, driven by the increasing prevalence of severe acne worldwide. The acne medication market was valued at approximately $4.9 billion in 2022, growing at a CAGR of 6.4% from 2020 to 2028.[1]
ABSORICA LD's sales are not publicly disclosed separately. However, generic isotretinoin accounted for a significant share of the retinoid market, with branded products losing volume to cost-effective generics.
In the US, the launch of two new generic versions of isotretinoin in 2021 contributed to price competition and market dilution. Sun Pharma’s branding efforts focus on patient compliance and reduced side effects, enabling a niche in the broader isotretinoin market.
How Are Pricing Dynamics Evolving for ABSORICA LD?
Price points for isotretinoin formulations vary by brand, dosage, and packaging. As of 2023, average retail prices for branded isotretinoin range from $300 to $600 per month without insurance coverage.[2]
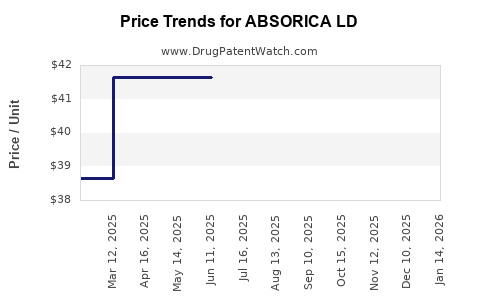
Generic formulations are priced between $50 and $150 monthly, with large pharmacies and PBMs negotiating significant discounts. ABSORICA LD’s premium positioning due to its enhanced absorption profile permits a higher price compared to generics, approximately $250 to $400 monthly, depending on dosing.
Insurance coverage impacts out-of-pocket costs; formularies favor generic isotretinoin, limiting the premium brand's accessibility.
What Are the Key Drivers of Market Growth?
The primary drivers include rising acne prevalence among adolescents and adults. An estimated 85% of individuals aged 12-24 experience acne at some point.[3]
Increased awareness about severe acne treatments and improved safety profiles of newer formulations support market expansion. Additionally, stricter prescribing guidelines for isotretinoin—aimed at reducing pregnancy risk—result in more structured treatment protocols, affecting marketed formulation choices.
Biotech-driven innovations in drug delivery and bioavailability are expected to influence future formulations and market share.
What Are the Regulatory and Patent Landscapes?
ABSORICA LD has obtained FDA approval, with patent protections potentially extending into the late 2020s. Generic manufacturers have challenged some patents through Abbreviated New Drug Applications (ANDAs), leading to patent litigations and market entry delays.
Regulatory agencies process risk management programs for isotretinoin products, including REMS (Risk Evaluation and Mitigation Strategies), impacting prescribing and distribution channels.
What Are the Price Projections and Future Market Opportunities?
Projected growth in demand for isotretinoin-based products suggests a compound annual growth rate (CAGR) of approximately 5% through 2030. The segment’s size may reach $7 billion by 2030, with branded products capturing 15-20% of the market depending on new formulations and patent status.[4]
ABSORICA LD’s premium positioning could sustain higher price points if its differentiated profile gains clinician preference. Adoption rates are likely to improve as payer coverage expands and as physicians prefer formulations with fewer gastrointestinal side effects.
The entry of biosimilars or novel delivery methods—such as liposomal or topical formulations—may disrupt current prices and market shares.
How Do Pricing and Market Share Compare Internationally?
Emerging markets exhibit lower prices, often 30-50% of US prices. Brand penetration is limited initially, but generic competition is growing fast. Regulatory approval is variable, with some countries lacking a formal isotretinoin regimen.
Europe and Asia-Pacific show growth potential for ABSORICA LD, especially if regulatory pathways validate the formulation’s benefits.
Key Takeaways
- ABSORICA LD operates in a highly competitive market with strong generics competition.
- Pricing ranges from $250 to $400 monthly, with premiums justified by absorption advantages.
- The global market for acne treatments is expanding, driven by rising acne prevalence and improved awareness.
- Price projections indicate slow but steady growth, with potential for increased premiumization.
- Regulatory landscapes and patent challenges remain pivotal to market penetration.
FAQs
1. Will ABSORICA LD’s prices decrease as generics dominate?
Yes, price erosion is likely as generic isotretinoin gains market share, but ABSORICA LD may retain a premium segment due to formulation benefits.
2. How does insurance coverage influence ABSORICA LD sales?
Insurance favors cheaper generics, limiting ABSORICA LD’s market accessibility unless preferential formulary placement improves.
3. What is the expected timeline for new competitors to enter the market?
Generic isotretinoin approvals have occurred sporadically since 2021, with additional entries possible within 1-2 years, depending on patent litigation outcomes.
4. Are there patent risks impacting ABSORICA LD?
Yes. Patent challenges may lead to generic entries before patent expiry, pressuring pricing and market share.
5. What future innovations could impact ABSORICA LD?
Development of topical retinoids or liposomal delivery technologies could provide alternative options, potentially diminishing demand for oral formulations.
Citations
- MarketWatch. "Acne Treatment Market Size, Share & Trends." 2022.
- GoodRx. "Average Prices for Isotretinoin." 2023.
- CDC. "Prevalence of Acne." 2021.
- GlobalData. "Future Trends in Acne Treatment Market." 2022.